Discovery and development of gastrointestinal lipase inhibitors
| Lipase inhibitor | |
|---|---|
pancreatic lipase | |
| Legal status | |
| In Wikidata |
Discovery of lipase inhibitors and their development
Pancreatic lipase inhibitor was originally discovered and isolated from
Orlistat has a few
Mechanism of action
The lipase inhibitors lipstatin and orlistat act locally in the intestinal tract. They are minimally absorbed in the circulation because of their lipophilicity.[7] Hence, they do not affect systemic lipases.[11]
The mechanism of lipase inhibitors in fat digestion is shown in figure 1. These inhibitors bind
Cetilistat has a
Drug target
Lipases in the gastrointestinal tract play a critical role in fat digestion. More than 95% of fat in food consists of
Gastric lipase
Gastric- and lingual lipases are the two acidic lipolytic enzymes that origin preduodenal but the gastric lipase is in much higher levels in humans. Gastric lipase is synthesized and
but has maximum activity at pH 3-6.[20] Fat digestion begins when gastric lipase hydrolyses dietary triglycerides, by cleaving only one long-, medium- or short-Gastric lipase is composed of 379
Pancreatic lipase
Pancreatic lipase is the most important lipolytic enzyme in the gastrointestinal tract[21] and is essential for fat digestion.[23] Pancreatic lipase is secreted from
The pancreatic lipase consists of 465 amino acids. Schematic picture of pancreatic lipase is shown in figure 2. Pancreatic and gastric lipases share little homology but have the same hydrophobic region at the active site, which is important for the lipolytic activity. The hydrophobic region has the hexapeptide sequence Val-Gly-His-Ser-Gln-Gly and is at Ser153 in pancreatic lipases but Ser152 in gastric lipases.[21]
Chemistry of lipase inhibitors
β-lactone class
The
Drugs of this class include:
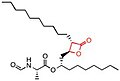
- Lipstatin, the first known lipase inhibitor,alkyl chains located at the α- (C6) and β-site (C13) of the compound. It contains N-formyl-L-leucine amino acid connected to the β-alkyl chain via ester-bond.[5] The structure of Lipstatin is shown in the table below.[28]
- Orlistat is a semi-synthetic compound, which has a similar structure to lipstatin. They differ only in the saturation of the β-alkyl chain, where orlistat is saturated while lipstatin has two
Structure-activity relationship (SAR)
Most natural lipase inhibitors differ only in the structure of the
| Lipstatin | Orlistat | Esterastin | Valilactone | Panclicin D | Ebelactone | Vibralactone | |
|---|---|---|---|---|---|---|---|
| Structure |  |
 |
 |
 |
 |
 | |
| IC50 value | 6.9 × 10−2 μg/ml[1] | 1.2 × 10−1 μg/ml[1] | 2.0 × 10−1 μg/ml[1] | 1.4 × 10−4 μg/ml[1] | 3.9 × 10−1 μg/ml[1] | 1.0 × 10−3 μg/ml[1] | 4.0 × 10−1 μg/ml[1] |
Synthetic lipase inhibitor: cetilistat
| Cetilistat | |
|---|---|
| Structure | |
| IUPAC | 2-hexadecoxy-6-methyl-3,1-benzoxazin-4-one[32] |
| Chemical formula | C25H39NO3[32] |
| Molar mass (g/mol) | 401.6[32] |
| IC50 | 5.95 nmol/l = 2.39 × 10-3 μg/mL (human pancreatic lipase)[33] |
Cetilistat is a synthetic lipase inhibitor. Instead of having a β-lactone structure like most of the lipase inhibitors,[16] it has a bicyclic benzoxazinone ring. It is also a lipophilic compound but differs in the hydro- and lipophilic side chain.[14] The structure and more information about Cetilistat is shown in the table on the right.[32]
Other lipase inhibitors
Other lipase inhibitors have been recognized, e.g. from different plant products. These include
Lipase inhibitors from microbial source can be divided into two classes based on their structure. Those who have a β-lactone ring are lipstatin, valilactone, percyquinin, panclicin A-E, ebelactone A and B, vibralactone and esterastin. Those who do not have a β-lactone ring are (E)-4-amino styryl acetate, ε–polylysine and caulerpenyne.[8]
Lipase inhibitors have also been made synthetically, e.g. cetilistat, based on the structure of triglycerides and other natural lipase substrates.[8] However, the synthetic lipase inhibitors differ in structure and some of them lack the β-lactone ring.[4]
Additional activities
Potential for cancer treatment
As further discussed, orlistat is a pancreatic and gastric lipase inhibitor. Orlistat is also a
Orlistat works locally in the intestines as a lipase inhibitor, and therefore suffers from several limitations in its development as a systemic drug. Its poor bioavailability and solubility are the main reasons to develop a new anticancer analogue to overcome these limitations.[6][34]
References
- ^ a b c d e f g h i j Schaefer B (2015). Natural Products in the Chemical Industry. Berlin Heidelberg: Springer.
- PMID 9225172.
- ^ "All centralized human medicinal product by ATC code". Public Health. European Commission. Retrieved 1 October 2016.
- ^ PMID 26417311.
- ^ PMID 25239907.
- ^ PMID 16749788.
- ^ a b c Medeiros-Neto GA, Halpern A, Bouchard C (2003). "Chapter 9: Orlistat in the treatment of obesity". In Halpern A (ed.). Progress in Obesity Research. Food & Nutrition Press.
- ^ PMID 17933690.
- ^ PMID 10730683.
- .
- ^ a b c Wilding JP (2008). "Intestinal lipase inhibitors". In Wilding JP (ed.). Pharmacotherapy of Obesity. Basel: Birkhäuser.
- ^ a b c "Summary of Product Characteristics" (PDF). Xenical. European Medicines Agency. 19 December 2013. Retrieved 1 October 2016.
- ^ "Questions and Answers about FDA's Initiative Against Contaminated Weight Loss Products". U.S. Food And Drug Administration. U.S. Department of Health and Human Services. Retrieved 1 October 2016.
- ^ PMID 19220279.
- ^ Bray GA, Ryan D (2007). "Ectopic Fat and the Metabolic Syndrome". In Toledo FG, Kelley DE (eds.). Overweight and the Metabolic Syndrome: From Bench to Bedside. US: Springer.
- ^ PMID 18393108.
- PMID 24524093.
- ^ Vaclavik V, Christian EW (2007). Essentials of Food Science. New York: Springer.
- PMID 2328454.
- ^ a b c Müller G, Petry S (2006). "Physiology of Gastrointestinal Lipolysis and Therapeutical Use of Lipases and Digestive Lipase Inhibitors". Lipases and Phospholipases in Drug Development: From Biochemistry to Molecular Pharmacology. Wiley.
- ^ ISBN 978-1-893997-12-7.
- PMID 19883129.
- ^ a b Johnson LR (2013). Gastrointestinal Physiology. Digestion and absorption of nutrients. Elsevier Mosby.
- ^ Mansbach II CM, Tso P, Kuksis A (June 2011). Lowe ME (ed.). Intestinal lipid metabolism. Springer Science & Business Media.
- ^ Shahidi F (2006). "Structure Related Effects on Absorption and Metabolism of Nutraceutical and Specialty Lipids". Nutraceutical and Specialty Lipids and their Co-Products. CRC Press.
- ISBN 978-1-893997-12-7.
- .
- ^ "(-)-Lipstatin". PubChem Compound Database. National Center for Biotechnology Information. Retrieved 1 October 2016.
- ISBN 978-1-893997-12-7.
- ^ "Orlistat". PubChem Compound Database. National Center for Biotechnology Information. Retrieved 1 October 2016.
- doi:10.1071/CH03121.
- ^ a b c d "Cetilistat". PubChem Compound Database. National Center for Biotechnology Information. Retrieved 1 October 2016.
- PMID 18500680.
- ^ PMID 20373869.
- ^ PMID 18710210.
- PMID 25309810.
- PMID 22338595.

