Discovery and development of gliflozins
Three drugs have been accepted by the Food and Drug Administration (FDA) in the United States; dapagliflozin, canagliflozin and empagliflozin. Canagliflozin was the first SGLT-2 inhibitor that was approved by the FDA, being accepted in March 2013. Dapagliflozin and empagliflozin were accepted in 2014.
Introduction
Role of kidneys in glucose homeostasis
There are at least four members of SLC-5 gene family, which are secondary active glucose transporters. The sodium glucose transporters proteins
SGLT-2 protein
Sodium/glucose co-transporter (
SGLT-2 is a member of the glucose transporter family and is a low-affinity, high-capacity glucose transporter. SGLT-2 is mainly expressed in the S-1 and S-2 segments of the
In diabetes, extracellular glucose concentration increases and this high glucose level leads to
Drugs that inhibit sodium/glucose cotransporter 2 inhibit renal glucose reabsorption which leads to enhanced urinary glucose excretion and lower glucose in blood. They work independently of insulin and can reduce glucose levels without causing hypoglycemia or weight gain.[1][7]
Discovery
Medieval physicians routinely tasted urine and wrote discourses on their observations. Which physician originally thought that
In the previous decade[
Phlorizin

Phlorizin is a compound that has been known for over a century. It is a naturally occurring botanical glucoside that produces renal glucosuria and blocks intestinal glucose absorption through inhibition of the sodium/glucose symporters located in the proximal renal tubule and mucosa of the small intestine. Phlorizin was first isolated in 1835 and was subsequently found to be a potent but rather non-selective inhibitor of both SGLT-1 and SGLT-2 proteins.[10][11][12]
Phlorizin seemed to have very interesting properties and the results in animal studies were encouraging, it improved insulin sensitivity and in diabetic
Although phlorizin was not suitable for further
Inhibition of SGLT-2 results as better control of glucose level, lower insulin, lower blood pressure and uric acid levels and augments calorie wasting. Some data supports the hypothesis that SGLT-2 inhibition may have direct renoprotective effects. This includes actions to attenuate tubular hypertrophy and hyperfiltration associated with diabetes and to reduce the tubular toxicity of glucose. Inhibition of SGLT-2 following treatment with dapagliflozin reduces the capacity for tubular glucose reabsorption by approximately 30–50%.[8]
Drug development
Phlorizin consists of glucose
Most of the reported SGLT-2 inhibitors are
T-1095

Because Phlorizin is a nonselective inhibitor with poor oral bioavailability, a phlorizin derivative was synthesised and called T-1095. T-1095 is a
ISIS 388626
According to preliminary findings of a novel method of SGLT-2 inhibition, the
Activity of SGLT-2 inhibitors in glycemic control
Michael Nauck recounts that
The results, summed up, were that 10 mg of dapagliflozin showed more effect than placebo in the control of glucose, when given for 24 weeks. However, no inferior efficacy of 10 mg dapagliflozin was shown when used as an add-on therapy to metformin, compared with
The results from meta-analysis when canagliflozin was examined, showed that compared to a placebo, canagliflozin affects HbA1c. Meta-analysis studies also showed that 10 mg and 25 mg of empagliflozin, improved HbA1c compared with a placebo.[4]
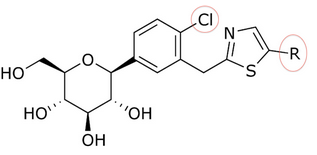
Structure-activity relationship (SAR)
The
The discovery of T-1095 led to an investigation[
Song and his partners did preparate thiazole compound by starting with carboxyl acid. Working with that, it took them three steps to get a compound like dapagliflozin with a thiazole ring. Inhibitory effects on SGLT-2 of the compounds were tested by Song and his partners. In tables 1, 2, and 3, the IC50 value changes depending on what compound is in the ring position, in the C-4 region of the proximal phenyl ring, and how the thiazole ring relates.[3]
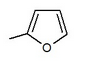
Many compounds gave different IC50 value in the ring position in an in vitro activity. For an example there was a big difference if there was an n-pentyl group (IC50 = 13,3 nM), n-butyl (IC50 = 119 nM), phenyl with 2-furyl (IC50 = 0,720) or 3-thiophenyl (IC50 = 0,772). As seen in table 1, the in vitro activity increases depending on what compound is bonded to the distal ring (given that in the C-4 region of the proximal phenyl ring is a Cl atom).[3]
Table 1: Differences in in vitro activity depending on which compound is bonded to the distal ring.
| R | IC50 (nM)[3] | Activity[3] |
|---|---|---|
 |
0.720 | in vitro activity improved* |
 |
1.14 | in vitro activity improved* |
| 13.3 | As the number of carbons increases, the IC-50 value fluctuates | |
| 19.6 | in vitro activity decreased* | |
 |
21.2 | in vitro activity decreased* |
*comparator to ethyl group (IC50 = 16,7)
In table 2, the in vitro activity changes depending on the compound in the C-4 region of the proximal phenyl ring (X). Small methyl groups or other halogen atoms in the C-4 position gave IC50 ranging from 0.72–36.7 (given that the phenyl with 2-furyl is in the ring position).[3]
Table 2: Differences in in vitro activity depending on what compound is in the C-4 region of the proximal phenyl ring.
| X[3] | IC50 (nM)[3] |
|---|---|
| Cl | 0.720 |
| Me | 1.43 |
| F | 6.11 |
| H | 22.6 |
| CN | 36.7 |
Table 3: Difference in the IC50 value depending on how the thiazole ring relates (nothing else is changed in the structure (X = Cl, R = phenyl with 2-furyl).
| Compound | IC50 (nM)[3] |
|---|---|
 |
0.720 |
 |
1.11 |
See also
- Sodium-glucose transport proteins
- SLC5A2
- SGLT1
- SGLT2
- Dapagliflozin
- Empagliflozin
- Canagliflozin
- Ipragliflozin
References
- ^ S2CID 5040096.
- S2CID 44399123.
- ^ PMID 24900297.
- ^ PMID 25246775.
- PMID 23210765.
- PMID 25311718.
- PMID 22923998.
- ^ PMID 25126408.
- ^ S2CID 45835248.
- ^ S2CID 37909306.
- ^ S2CID 21788174.
- ^ PMID 24729157.
- ^ PMID 22262072.
- ^ PMID 21940664.
- PMID 21398124.
- S2CID 22590286.
- . Retrieved 10 June 2025.
- ^ "Isis Initiates Phase 1 Clinical Trial of SGLT2 Antisense Drug".
- PMID 28596840.
- PMID 21193308.