Discovery and development of triptans
History
Search for a new anti-migraine drug started at
Mechanism
Triptans are specific and selective agonists for the 5-HT1 receptors. Sumatriptan[4] binds to 5-HT1D receptors, zolmitriptan,[5] rizatriptan,[6] naratriptan,[7] almotriptan,[8] and frovatriptan[9] binds to 5-HT1B/1D and eletriptan[10] binds to 5-HT1B/1D/1F receptors. Triptans are believed to exert their effects through vasoconstriction, leading to reduced carotid arterial circulation without affecting cerebral blood flow, peripheral neuronal inhibition, or inhibition of transmission through second order neurons of the trigeminocervical complex. [1]
Receptors
5-HT receptors are all G-protein coupled receptors (GPCR) except for 5-HT3 which is a ligand gated ion channel. The receptors that have been found to be involved in migraine are 5-HT1B, 5-HT1D and 5-HT1F receptors. 5-HT1B are found in meningeal arteries, agonism of 5-HT1B causes vasoconstriction in cranial nerves. The 5-HT1D receptors are located primarily in the trigeminal nerve in the central nervous system (CNS). They are also found in vascular smooth muscles, mediating contraction. Agonism of 5-HT1D receptors subdues the release of inflammatory inducing nerve stimulants. The amino acids contributing to the binding of ligands to the receptor are aspartic acid (Asp), phenylalanine (Phe), serine (Ser), threonine (Thr), tryptophan (Trp) and tyrosine (Tyr). It has been shown that both 5-HT1B and 5-HT1D receptors in humans have a very similar amino acids structures which demonstrates the similarities in binding properties.[11][12][13]
Design
All triptans have an indole structure identical to the neurotransmitter 5-HT. Classic triptan structure contain side chain on the indole ring, and a basic nitrogen in a similar distance from the indole structure. The main structural difference of the triptans is the position of the sulfonamide and the side chain attached to it (see figure 1 and table 1). Rizatriptan and zolmitriptan have instead of a sulfonamide a triazole and 2-oxazolidone respectively. Another exception to the classic structure is seen on eletriptan where the nitrogen-alkyl chain connected to the indole ring is replaced with a dimethyl-pyrrolidine, and in naratriptan where the nitrogen-alkyl chain is replaced with a 1-methyl-piperidine ring.
One of the frovatriptan side chains forms an additional ring with the indole, resulting in a carbazole ring system.
Structures of the triptans
| Triptan | R1 | R2 | Triptan | R1 | R2 |
|---|---|---|---|---|---|
| Sumatriptan | 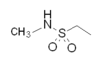
|

|
Eletriptan | 
|

|
| Rizatriptan | 
|

|
Naratriptan | 
|

|
| Almotriptan | 
|

|
Frovatriptan | 
|

|
| Zolmitriptan | 
|

|
– | – | – |
The 5-HT1B/D pharmacophore
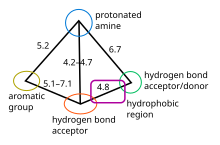

5-HT1B and 5-HT1D receptors are considered very similar, they share amino acid
Relevant structural features of triptans and binding to the receptor
Triptan structures were designed from the structure of 5-HT to attain affinity to 5-HT receptors, hence the identical indole structure. The hydroxyl group (-OH) on the hexane of the indole core and the alkyl-amine side chain on position C3 on 5-HT have been replaced with other compounds, such as sulfonamides or azol-ring structured derivatives and different amine-alkyl side chains. An electro-negative group can form a hydrogen bond with Thr in the pocket of the receptor. Sulfonamide derivatives attached to the hexane ring of the indole structure have electro-negative properties, as well as the triazole and 2-oxazolidone on rizatriptan and zolmitriptan respectively. This can increase binding ability of the compound and the efficacy, especially with the 5-HT1D receptor.[11]

A schematic drawing of the binding of sumatriptan to 5-HT1D receptor can be seen in figure 4. One study[11] showed that sumatriptan fits better in the binding site of the receptor when the side chain with the protonated nitrogen atom is folded back over the indole structure. This alignment contributes to the hydrogen bonding between the nitrogen in the sulfonamine and the Ser138 in the binding site. It is also favorable to the formation of the hydrogen bond between the oxygen of the sulfonamine and Thr202. Other binding in the pocket of the binding site occurs with the nitrogen atom in the pentene ring of the indole structure of the triptan and the amino acid Ser352. This energetically favorable position of the agonist makes it possible for additional binding of the ligand to other Ser in the binding site, along with additional anchoring between Phe in the pocket of the binding site and the indole of the agonist. The binding of Phe and the triptan is caused by π stacking interactions of the indole and amino acid and an additional effect on this interaction is because of dispersive effect of amino acid leucine (Leu; not shown in figure 4). The amino acids Trp343 and Tyr346 both have electron rich π-systems in their aromatic structures. With their position in the binding site they create a sort of aromatic cage around the protonated nitrogen atom of the side chain on position C3 on the triptans (this nitrogen atom is protonated at physiological condition), and thereby stabilizes the ion bond the nitrogen atom has formed with a carboxylate on aspartic acid. Side chains of the surrounding amino acids can have an effect on the binding of the nitrogen atom, mainly three Phe can affect the methyl groups bound to the nitrogen atom (not shown in figure 4).[11][12][13]
Eletriptan has higher
By placing an electron-withdrawing group or large group on position C2 on the indole structure the 5-HT agonist is conversed into an antagonist. This is thought to be because the indole ring is unable to occupy the aromatic part of the binding site.[12]
Triptan drugs
Properties of formulations
Sumatriptan was the pioneer drug in this class. The second generation's triptans such as zolmitriptan, naratriptan, rizatriptan, almotriptan, eletriptan and frovatriptan soon became available.[18] Different triptans are available in different
| Generic | Formulations[19] | Doses (mg)[19] | Maximum daily dose (mg)[19] |
Onset of action (min)[20] |
Duration
of action[20] |
Affinity (pKI in nM) | Metabolism[21] | Excretion[20] |
|---|---|---|---|---|---|---|---|---|
| Sumatriptan |
Tablets |
25, 50, 100 |
200 |
Short | 7.9–8.5 | MAO-A |
Urine (57%), | |
| Zolmitriptan |
Tablets |
2.5, 5 |
10 |
45 |
Short | 9.2 |
CYP1A2 |
Urine (65%), |
| Naratriptan | Tablets | 1, 2.5 |
5 |
60–180 | Long | 8.3 |
CYPa |
Urine |
| Rizatriptan |
Tablets |
5, 10 |
30 |
30–120 |
Short | 7.7 | MAO-A | Urine |
| Almotriptan | Tablets | 6.25, 12.5 | 25 | 60–180 | Short | 7.8 |
MAO-A |
Urine (40%), |
| Eletriptan | Tablets | 20, 40 | 80 | <60[23] | – | 8.9 | CYP3A4 | – |
| Frovatriptan | Tablets | 2.5 | 7.5 | 60–120 | Long | 8.4 | CYP1A2 | Urine (40%) |
a Specific enzyme not yet reported.
The
Pharmacokinetics
Pharmacokinetic properties (see table 3) are important when new drugs are developed.[26]
Patients seek rapid onset of action to relief the headache. Relatively short tmax, good bioavailability and lipophilicity are pharmacokinetic properties that have been associated with rapid onset of action. It has been speculated that good ability to cross the blood brain barrier and relatively long terminal elimination half-life may result in a lower incidence of headache recurrence. Sumatriptan and rizatriptan undergo first pass hepatic metabolism and result in lower bioavailability.[18]
| Generic | Bioavailability (%)[26] | Lipophilicity[19] | Protein binding (%)[20] |
t1/2 (h)[26] | tmax (h)[22] | ClR (mL min-1)[27] |
Log DpH7.4[28] | VD[20] |
|---|---|---|---|---|---|---|---|---|
| Sumatriptan | 14 | Low | 10–21 | 2–2.5 | 2–2.5 | 260 | -1.5 | 2.4–3.3 L/kg |
| Zolmitriptan | 40 | Moderate | 25 | 3 | 2 | 193 | -1.0 | 7.0 L/kg |
| Naratriptan | 63(M) / 74(F) | High | 28–31 | 5–6 | 2–3 | 220 | -0.2 | 2.4 L/kg |
| Rizatriptan | 47 | Moderate | 14 | 2–2.5 | 1.3 | 414 | -0.7 | 140(M) / 110(F) L |
| Almotriptan | 69 | – | 35 | 3.6 | 1.4–3.8 | – | -2.1 | 180–200 L |
| Eletriptan | 50 | High | 85[10] | 4–5 | 1–2 | 597 | 0.5 | 138 L[10] |
| Frovatriptan | 24(M) / 30(F) | Low | 20–30 | 25[18][26] | 2–4 | 216(M) / 132(F)[9] | -1.0[29] | 4.2(M) / 3.0(F) L/kg |
t1/2 = Elimination half-life;
tmax = Time to reach peak plasma drug concentration;
M = Male; F = Female
Future research
Most triptans were developed and introduced in the 1990s. Further studies have not shown much promise regarding the development of new triptans with better duration of action, efficacy and safety profile. Therefore, it is unlikely that further variations will be developed and new anti-migraine drugs are likely to have another mechanism of action.[29]
References
- ^ S2CID 2368571, archived from the originalon 2012-12-17
- PMID 17141570
- ]
- ^ "Imigran Tablets 50mg Imigran Tablets 100mg". Archived from the original on 2007-12-18. Retrieved 2008-11-09.
- ^ "Zomig Tablets 2.5mg". Archived from the original on 2005-08-10. Retrieved 2008-11-09.
- ^ "Maxalt 5mg, 10mg Tablets, Maxalt Melt 10mg Oral Lypophilisates". Archived from the original on 2008-05-02. Retrieved 2008-11-09.
- ^ "Naramig Tablets 2.5mg". Archived from the original on 2007-04-30. Retrieved 2008-11-09.
- ^ "Axert". Retrieved 2008-11-09.
- ^ a b "Migard". Retrieved 2008-11-09.[permanent dead link]
- ^ a b c "Relpax – 20 mg and 40 mg". Archived from the original on 2004-06-20. Retrieved 2008-11-09.
- ^
- ^ PMID 17017971
- ^
- ^ PMID 7658443.
- ^ PMID 11262079.
- ^ S2CID 6478226.
- PMID 8496922.
- ^ PMID 16356805
- ^ PMID 12806521
- ^ a b c d e "Drug class review: Oral 5HT1 Receptor agonists" (PDF). U.S. Department of Veterans Affairs. Archived from the original (PDF) on 2009-01-14. Retrieved 2008-11-03.
- PMID 12444236
- ^ S2CID 22223822
- PMID 12807526
- ^ "press release - Treximet (sumatriptan and naproxen sodium) tablets approved by FDA for acute treatment of migraine". GlaxoSmithKline. Archived from the original on 2008-12-04. Retrieved 2008-11-09.
- S2CID 43314572, archived from the originalon 2012-12-18
- ^ S2CID 31937163
- PMC 3611827
- ]
- ^ ]


