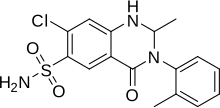
Metolazone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Zaroxolyn |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682345 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~65% |
| Metabolism | kidney (minimal) |
| Elimination half-life | 14 hours |
| Excretion | primarily urine |
| Identifiers | |
| |
JSmol) | |
| Melting point | 260 °C (500 °F) |
| |
| |
| | |
Metolazone is a
It was patented in 1966 and approved for medical use in 1974.[1]
Medical uses
One of the primary uses of metolazone is for treating
Metolazone may also be used in kidney disease, such as chronic kidney disease or the nephrotic syndrome. Chronic kidney disease causes excess fluid retention that is often treated with diet adjustments and diuretics.[2] Metolazone may be combined with other diuretics (typically loop diuretics) to treat diuretic resistance in congestive heart failure, chronic kidney disease, and nephrotic syndrome.[3] Metolazone and a loop diuretic will synergistically enhance diuresis over the use of either agent alone. Using this combination, diuretic effects will occur at two different segments of the nephron; namely, the loop diuretic will act at the loop of Henle, and metolazone will act at the distal convoluted tubule. Metolazone is frequently prescribed in addition to the loop diuretic. Metolazone may be used for edema caused by liver cirrhosis as well.
The other major use of metolazone is in treating hypertension (high blood pressure). Thiazide diuretics, though usually not metolazone, are very often used alone as first-line treatment for mild hypertension. They are also used in combination with other drugs for difficult-to-treat or more severe hypertension. "The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure" (JNC 7) recommends thiazide diuretics as the initial medication for treatment of hypertension. Hydrochlorothiazide is by far the most commonly used, as it is both better-studied and cheaper (about four times) than metolazone, although as mentioned above metolazone is used in patients with moderate chronic kidney disease.[4]
Toxicity
Since thiazide diuretics affect the transport of
Mechanism of action
The primary target of all thiazide diuretics, including metolazone, is the distal convoluted tubule, part of the nephron in the kidney, where they inhibit the sodium-chloride symporter.
In the kidney, blood is filtered into the
As with other regions, tubule cells in the distal convoluted tubule possess the
Metolazone and the other thiazide diuretics inhibit the function of the sodium-chloride symporter, preventing sodium and chloride, and therefore water too, from leaving the lumen to enter the tubule cell. As a result, water remains in the lumen and is excreted as urine, instead of being reabsorbed into the bloodstream. Since most of the sodium in the lumen has already been reabsorbed by the time the filtrate reaches the distal convoluted tubule, thiazide diuretics have limited effects on water balance and on
Pharmacokinetics
Metolazone is only available in oral preparations. Approximately 65% of the amount ingested becomes available in the bloodstream. Its half-life is approximately fourteen hours, similar to indapamide but considerably longer than hydrochlorothiazide. Metolazone is around ten times as potent as hydrochlorothiazide. The primary form of excretion is in the urine (around 80%); the remaining fifth is evenly split between biliary excretion and metabolism into inactive forms.[4]
Chemistry
The use of activated anthranilic acid derivatives facilitates the preparation of the amides in those cases where the amines are either unreactive or difficult to obtain.

Thus, reaction of (1) with
Structure and classification
Metolazone is a quinazoline, a derivative of the similar diuretic quinethazone, as well as a sulfonamide. It is related to analogs of 1,2,4-benzothiadizine-1,1-dioxide (benzothiadiazine). Such drugs are called benzothiadiazides, or thiazides for short; however, in terms of chemistry, metolazone is not a substituted benzothiadiazine, and therefore is not technically a thiazide. Since metolazone (as well as other drugs like indapamide) acts on the same target as thiazides and behave in a similar pharmacologic fashion, it is, however, considered a "thiazide-like diuretic." Therefore, metolazone and similar drugs are often categorized with thiazide diuretics despite not being thiazides themselves.[4]
History
Metolazone was developed in the 1970s. Its creator, Indian born chemist Dr. Bola Vithal Shetty has been active in helping the U.S. Food and Drug Administration review drug applications, and in the development of new medicines.[6] Metolazone quickly gained popularity due to its lower kidney toxicity compared to other diuretics (especially thiazides) in patients with chronic kidney disease.
References
- ISBN 9783527607495.
- ^ McGraw-Hill.
- S2CID 23704590.
- ^ McGraw-Hill.
- PMID 5458377.
- ^ Katague DB. "Chemistry Reviewer Still in Lab" (PDF). News Along the Pike. 2 (10). Food and Drug Administration' s Center for Drug Evaluation and Research.
