Bisphenol A

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
4,4′-(Propane-2,2-diyl)diphenol | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.001.133 |
| EC Number |
|
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 2430 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H16O2 | |
| Molar mass | 228.291 g·mol−1 |
| Appearance | White solid |
| Odor | Phenolic, medical |
| Density | 1.217 g/cm3[1] |
| Melting point | 155 °C (311 °F; 428 K)[5] |
| Boiling point | 250–252 °C (482–486 °F; 523–525 K)[5] at 13 torrs (0.017 atm) |
| 0.3 g/L (25 °C)[2] | |
| log P | 3.41[3] |
| Vapor pressure | 5×10−6 Pa (25 °C)[4] |
| Hazards[6] | |
| GHS labelling: | |
   
| |
| Danger | |
| H317, H318, H335, H360, H411[6] | |
| P201, P202, P261, P273, P302+P352, P304+P340, P305+P351+P338, P308+P313, P333+P313, P363, P403+P233[6] | |
| NFPA 704 (fire diamond) | |
| Flash point | 227 °C (441 °F; 500 K)[6] |
| 510 °C (950 °F; 783 K)[6] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
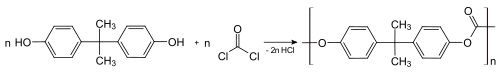
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water.[2][7] BPA is produced on an industrial scale by the condensation reaction of phenol and acetone. Global production in 2022 was estimated to be in the region of 10 million tonnes.[8]
BPA's largest single application is as a
although it is often wrongly labelled as such.The health effects of BPA have been the subject of prolonged public and scientific debate.
History
Bisphenol A was first reported in 1891 by the Russian chemist Aleksandr Dianin.[26]
In 1934, workers at
The British biochemist Edward
Production
The synthesis of BPA still follows Dianin's general method, with the fundamentals changing little in 130 years. The
Usually, the addition of acetone takes place at the para position on both phenols, however minor amounts of the ortho-para (up to 3%) and ortho-ortho isomers are also produced, along with several other minor by‑products.[35] These are not always removed and are known impurities in commercial samples of BPA.[36][35]
Properties
BPA has a fairly high melting point but can be easily dissolved in a broad range of organic solvents including
Uses and applications

Main uses
Polycarbonates
About 65–70% of all bisphenol A is used to make
Epoxy and vinyl ester resins
About 25–30% of all BPA is used in the manufacture of
Some of this is further reacted with
Minor uses
The remaining 5% of BPA is used in a wide range of applications, many of which involve plastic.[49] BPA is a main component of several high-performance plastics, the production of these is low compared to other plastics but still equals several thousand tons a year. Comparatively minor amounts of BPA are also used as additives or modifiers in some commodity plastics. These materials are much more common but their BPA content will be low.
Plastics
- As a major component
- Polycyanurates can be produced from BPA by way of its diprinted circuit boards.
- Polyetherimides such as Ultem can be produced from BPA via a nitro-displacement of appropriate bisnitroimides.[51][52] These thermoplastic polyimide plastics have exceptional resistance to mechanical, thermal and chemical damage. They are used in medical devices and other high performance instrumentation.
- Polybenzoxazines may be produced from a number of biphenols, including BPA.[53][54]
- Bisphenol-A formaldehyde resins are a subset of high-pressure laminates[49]
- As a minor component
- Polyurethane can incorporate BPA and its derivatives as hard segment chain extenders, particularly in memory foams.[56][57]
- heat stabilizers. Historically 5–10% by weight of BPA was included in barium-cadmium types, although these have largely been phased out due health concerns surrounding the cadmium. BPA diglycidyl ether (BADGE) is used as an acid scavenger, particularly in PVC dispersions, such as organosols or plastisols,[59][60] which are used as coatings for the inside of food cans, as well as embossed clothes designs produced using heat transfer vinyl or screen printing machines.[19]
- BPA is used to form a number of phosphorus oxychloride and phenol forms bisphenol-A bis(diphenyl phosphate) (BADP), which is used as a liquid flame retarder in some high performance polymer blends such as polycarbonate/ABS mixtures.[63]
Other applications
- BPA is used as an antioxidant in several fields, particularly in brake fluids.[64]
- BPA is used as a developing agent in thermal paper (shop receipts).[20] Recycled paper products can also contain BPA,[65] although this can depend strongly on how it is recycled. Deinking can remove 95% of BPA,[9] with the pulp produced used to make newsprint, toilet paper and facial tissues. If deinking is not performed then the BPA remains in the fibers, paper recycled this way is usually made into corrugated fiberboard.[9]
- Ethoxylated BPA finds minor use as a 'levelling agent' in tin electroplating.
- Several drug candidates have also been developed from bisphenol A, including ralaniten, ralaniten acetate, and EPI-001.
BPA substitutes
Concerns about the health effects of BPA have led some manufacturers replacing it with other bisphenols, such as bisphenol S and bisphenol F. These are produced in a similar manner to BPA, by replacing acetone with other ketones, which undergo analogous condensation reactions.[7] Thus, in bisphenol F, the F signifies formaldehyde. Health concerns have also been raised about these substitutes.[66][24] Alternative polymers, such as tritan copolyester have been developed to give the same properties as polycarbonate (durable, clear) without using BPA or its analogues.
| Structural formula | Name | CAS
|
Reactants
| |
|---|---|---|---|---|
 |
Bisphenol AF | 1478-61-1 | Phenol | Hexafluoroacetone |
| Bisphenol F | 620-92-8 | Phenol | Formaldehyde | |
 |
Bisphenol S | 80-09-1 | Phenol | Sulfur trioxide |
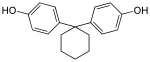 |
Bisphenol Z | 843-55-0 | Phenol | Cyclohexanone |
 |
Tetramethyl bisphenol F | 5384-21-4 | 2,6-xylenol |
Formaldehyde |
Human safety
Exposure

As a result of the presence of BPA in plastics and other commonplace materials, most people are frequently exposed to trace levels of BPA.
Health effects and regulation
The health effects of BPA have been the subject of prolonged public and scientific debate,[12][13][14] with PubMed listing more than 18,000 scientific papers as of 2024.[75] Concern is mostly related to its estrogen-like activity, although it can interact with other receptor systems as an endocrine-disrupting chemical.[76] These interactions are all very weak, but exposure to BPA is effectively lifelong, leading to concern over possible cumulative effects. Studying this sort of long‑term, low‑dose interaction is difficult, and although there have been numerous studies, there are considerable discrepancies in their conclusions regarding the nature of the effects observed as well as the levels at which they occur.[12] A common criticism is that industry-sponsored trials tend to show BPA as being safer than studies performed by academic or government laboratories,[14][77] although this has also been explained in terms of industry studies being better designed.[13][78]
Public health agencies in the EU,
BPA exhibits very low
Pharmacology

BPA has been found to interact with a diverse range of
Bisphenol A's interacts with the
Environmental safety
Distribution and degradation
BPA has been detectable in the natural environment since the 1990s and is now widely distributed.[95] It is primarily a river pollutant,[96] but has also been observed in the marine environment,[97] in soils,[98] and lower levels can also be detected in air.[99] The solubility of BPA in water is low (~300 g per ton of water)[2] but this is still sufficient to make it a significant means of distribution into the environment.[98] Many of the largest sources of BPA pollution are water-based, particularly wastewater from industrial facilities using BPA. Paper recycling can be a major source of release when this includes thermal paper,[9][100] leaching from PVC items may also be a significant source,[96] as can landfill leachate.[101]
In all cases, wastewater treatment can be highly effective at removing BPA, giving reductions of 91–98%.[102] Regardless, the remaining 2–9% of BPA will continue through to the environment, with low levels of BPA commonly observed in surface water and sediment in the U.S. and Europe.[103]
Once in the environment BPA is aerobically biodegraded by a wide a variety of organisms.
Environmental effects
BPA is an environmental
See also
- Structurally related
- 4,4'-Dihydroxybenzophenone - used as a UV stabilizer in cosmetics and plastics
- Dinitrobisphenol A - a proposed metabolite of BPA, which may show increased endocrine disrupting character
- HPTE - a metabolite of the synthetic insecticide methoxychlor
- Others
- 2,2,4,4-Tetramethyl-1,3-cyclobutanediol - next generation BPA replacement
- 4-tert-Butylphenol - used as a chain-length regulator in the production of polycarbonates and epoxy resins, it has also been studied as a potential endocrine disruptor
References
- S2CID 97284173.
- ^ .
- S2CID 13528747.
- ^ "Chemical Fact Sheet – Cas #80057 CASRN 80-05-7". speclab.com. 1 April 2012. Archived from the original on 12 February 2012. Retrieved 14 June 2012.
- ^ S2CID 98036316.
- ^ a b c d e Sigma-Aldrich Co., Bisphenol A.
- ^ ISBN 978-3527306732.
- ^ S2CID 208186123.
- ^ ISBN 9789279175428.
- ^ PMID 34323825.
- ISBN 3527306730.
- ^ ISBN 978-92-4-156427-4. Retrieved 23 March 2022.
- ^ PMID 21438738.
- ^ PMID 19337501.
- ISSN 0021-1753.
- ^ PMID 10746941.
- ^ PMID 22889897.
- PMID 21598963.
- ^ PMID 28368574.
- ^ PMID 28528315.
- PMID 28759120.
- ^ S2CID 49194096.
- PMID 32092919.
- ^ PMID 27143250.
- PMID 25475787.
- ^ See:
- А. Дианина (1891) "О продуктахъ конденсацiи кетоновъ съ фенолами" (On condensation products of ketones with phenols), Журнал Русского физико-химического общества (Journal of the Russian Physical Chemistry Society), 23 : 488-517, 523–546, 601–611; see especially pages 491-493 ("Диметилдифенолметань" (dimethyldiphenolmethane)).
- Reprinted in condensed form in: A. Dianin (1892) "Condensationsproducte aus Ketonen und Phenolen" (Condensation products of ketones and phenols), Berichte der Deutschen chemischen Gesellschaft zu Berlin, 25, part 3 : 334-337.
- ISBN 978-3527306732.
- ISBN 978-3527306732.
- ^ PMID 19890158.
- S2CID 4171635.
- .
- PMID 17559906.
- ISBN 978-0-8493-4954-6.
- .
- ^ PMID 15041297.
- S2CID 44172964.
- ISBN 9781498754293.)
{{cite book}}: CS1 maint: location missing publisher (link - ISBN 9780080347141.
- ^ "2,2-bis(4-Hydroxyphenyl)propane". www.ccdc.cam.ac.uk. The Cambridge Crystallographic Data Centre. Retrieved 29 June 2022.
- .
- .
- ^ "4,4'-isopropylidenediphenol". sdbs.db.aist.go.jp. Spectral Database for Organic Compounds (SDBS). Retrieved 29 June 2022.
- ISBN 978-3527306732.
- PMID 28106795.
- ISBN 978-0-471-52695-7.
- PMID 19627357.
- PMID 11950052.
- PMID 15889433.
- ^ PMID 21570349.
- ISBN 978-0-7514-0044-1.
- .
- ISBN 978-1-4557-3107-7.
- S2CID 94255723.
- .
- doi:10.1002/0471238961.0118151323080920.a01.)
{{cite journal}}: Cite journal requires|journal=(help - S2CID 224955435.
- ISBN 9789004161245.
- S2CID 98016356.
- S2CID 97146596.
- ISBN 978-3527306732.
- S2CID 198998658.
- doi:10.1002/pi.2290.
- S2CID 44140721.
- PMID 21939283.
- PMID 25775505.
- PMID 18197297.
- PMID 30090587.
- PMID 17825522.
- PMID 21664675.
- .
- S2CID 24467830.
- S2CID 233290894.
- ^ "Does using plastic bottles and containers cause cancer?". Cancer Research UK. 23 December 2021.
- ^ "bisphenol a - Search Results - PubMed". PubMed. Retrieved 26 January 2024.
- ^ S2CID 23088708.
- PMID 16079060.
- PMID 23867546.
- ^ "Bisphenol A - ECHA". echa.europa.eu. Archived from the original on 8 June 2022. Retrieved 28 March 2022.
- ISBN 9789291996421.
- hdl:2164/12119.
- ^ OCSPP US EPA (21 September 2015). "Risk Management for Bisphenol A (BPA)". www.epa.gov. Retrieved 28 March 2022.
- S2CID 240266384.
- ^ Health Canada (16 April 2013). "Bisphenol A (BPA)". www.canada.ca (Health Canada). Government of Canada. Retrieved 28 March 2022.
- FSANZ). Department of Health (Australia). Retrieved 28 March 2022.
- ISBN 9789279158698.
- ^ "Indirect Food Additives: Polymers". Federal Register. U.S. Government Publishing Office.77 FR 41899
- ^ Legislative Services Branch (1 July 2020). "Consolidated federal laws of canada, Canada Consumer Product Safety Act". laws-lois.justice.gc.ca.
- ^ "EUR-Lex - 32011L0008 - EN - EUR-Lex". EUR-Lex. European Union.
COMMISSION DIRECTIVE 2011/8/EU of 28 January 2011 amending Directive 2002/72/EC as regards the restriction of use of Bisphenol A in plastic infant feeding bottles
- ECHA). Retrieved 19 June 2017.
- ^ Fisher D (12 July 2019). "EU court confirms BPA as substance of 'very high concern'". Environmental Health News. Retrieved 21 July 2020.
- PMID 14605012.
- ^ PMID 17761695.
- PMID 24530924.
- ^ PMID 9566294.
- ^ PMID 26674671.
- S2CID 58536418.
- ^ S2CID 43509780.
- PMID 34323825.
- PMID 10834378.
- ^ .
- S2CID 12283834.
- PMID 19746705.
- PMID 16288945.
- S2CID 94353391.
- PMID 27401879.
- S2CID 207939269.
- ^ S2CID 247423338.
- ^ S2CID 218765595.
- ^ PMID 19528055.