Aspirin
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /əˌsiːtəlˌsælɪˈsɪlɪk/ |
| Trade names | Bayer Aspirin, others |
| Other names |
|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682878 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral, rectal |
| Drug class | Nonsteroidal anti-inflammatory drug (NSAID) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80–100%[6] |
| Protein binding | 80–90%[7] |
| Metabolism | Liver (CYP2C19 and possibly CYP3A), some is also hydrolysed to salicylate in the gut wall.[7] |
| Elimination half-life | Dose-dependent; 2–3 h for low doses (100 mg or less), 15–30 h for larger doses.[7] |
| Excretion | Urine (80–100%), sweat, saliva, feces[6] |
| Identifiers | |
| |
JSmol) | |
| Density | 1.40 g/cm3 |
| Melting point | 135 °C (275 °F) [9] |
| Boiling point | 140 °C (284 °F) (decomposes) |
| Solubility in water | 3 g/L |
SMILES
| |
| |
| (verify) | |
Aspirin (/ˈæsp(ə)rɪn/[10]) is the genericized trademark for acetylsalicylic acid (ASA), a nonsteroidal anti-inflammatory drug (NSAID) used to reduce pain, fever, and inflammation, and as an antithrombotic.[11] Specific inflammatory conditions that aspirin is used to treat include Kawasaki disease, pericarditis, and rheumatic fever.[11]
Aspirin is also used long-term to help prevent further
One common
A
Aspirin is available
Brand vs. generic name
In 1897, scientists at the Bayer company began studying acetylsalicylic acid as a less-irritating replacement medication for common salicylate medicines.[14]: 69–75 [21] By 1899, Bayer had named it "Aspirin" and was selling it around the world.[16]
Aspirin's popularity grew over the first half of the 20th century, leading to competition between many brands and formulations.[22] The word Aspirin was Bayer's brand name; however, its rights to the trademark were lost or sold in many countries.[22] The name is ultimately a blend of the prefix a(cetyl) + spir Spiraea, the meadowsweet plant genus from which the acetylsalicylic acid was originally derived at Bayer + -in, the common chemical suffix.[citation needed]
Chemical properties
Aspirin decomposes rapidly in solutions of
Like
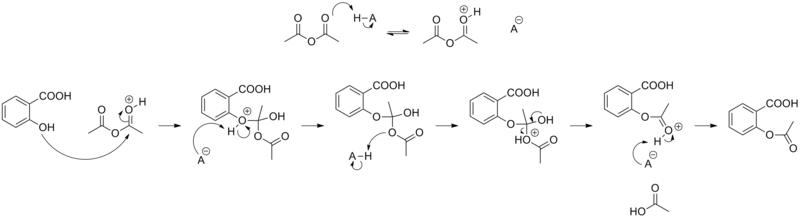
Synthesis
The synthesis of aspirin is classified as an

Reaction between acetic acid and salicylic acid can also form aspirin but this esterification reaction is reversible and the presence of water can lead to hydrolysis of the aspirin. So, an anhydrous reagent is preferred.[27]
- Reaction mechanism
Formulations containing high concentrations of aspirin often smell like vinegar[28] because aspirin can decompose through hydrolysis in moist conditions, yielding salicylic and acetic acids.[29]
Physical properties
Aspirin, an
Polymorphism
Until 2005, there was only one proven polymorph of aspirin (Form I), though the existence of another polymorph was debated since the 1960s, and one report from 1981 reported that when crystallized in the presence of aspirin anhydride, the
Form II was reported in 2005,[33][34] found after attempted co-crystallization of aspirin and levetiracetam from hot acetonitrile.
In form I, pairs of aspirin molecules form centrosymmetric
Pure Form II aspirin could be prepared by seeding the batch with aspirin anhydrate in 15% weight.[32]
Form III was reported in 2015 by compressing form I above 2 GPa, but it reverts back to Form I when pressure is removed.[36] Form IV was reported in 2017. It is stable at ambient conditions.[37]
Mechanism of action
Discovery of the mechanism
In 1971, British
Prostaglandins and thromboxanes
Aspirin's ability to suppress the production of prostaglandins and thromboxanes is due to its irreversible inactivation of the cyclooxygenase (COX; officially known as prostaglandin-endoperoxide synthase, PTGS) enzyme required for prostaglandin and thromboxane synthesis. Aspirin acts as an acetylating agent where an acetyl group is covalently attached to a serine residue in the active site of the COX enzyme (Suicide inhibition). This makes aspirin different from other NSAIDs (such as diclofenac and ibuprofen), which are reversible inhibitors.
Low-dose aspirin use irreversibly blocks the formation of thromboxane A2 in platelets, producing an inhibitory effect on platelet aggregation during the lifetime of the affected platelet (8–9 days). This antithrombotic property makes aspirin useful for reducing the incidence of heart attacks in people who have had a heart attack, unstable angina, ischemic stroke or transient ischemic attack.[41] 40 mg of aspirin a day is able to inhibit a large proportion of maximum thromboxane A2 release provoked acutely, with the prostaglandin I2 synthesis being little affected; however, higher doses of aspirin are required to attain further inhibition.[42]
Prostaglandins, local
COX-1 and COX-2 inhibition
At least two different types of
Several COX-2 inhibitors, such as
Furthermore, aspirin, while inhibiting the ability of COX-2 to form pro-inflammatory products such as the
Additional mechanisms
Aspirin has been shown to have at least three additional modes of action. It uncouples oxidative phosphorylation in cartilaginous (and hepatic) mitochondria, by diffusing from the inner membrane space as a proton carrier back into the mitochondrial matrix, where it ionizes once again to release protons.[51] Aspirin buffers and transports the protons. When high doses are given, it may actually cause fever, owing to the heat released from the electron transport chain, as opposed to the antipyretic action of aspirin seen with lower doses. In addition, aspirin induces the formation of NO-radicals in the body, which have been shown in mice to have an independent mechanism of reducing inflammation. This reduced leukocyte adhesion is an important step in the immune response to infection; however, evidence is insufficient to show aspirin helps to fight infection.[52] More recent data also suggest salicylic acid and its derivatives modulate signalling through NF-κB.[53] NF-κB, a transcription factor complex, plays a central role in many biological processes, including inflammation.[54][55][56]
Aspirin is readily broken down in the body to salicylic acid, which itself has anti-inflammatory, antipyretic, and analgesic effects. In 2012, salicylic acid was found to activate AMP-activated protein kinase, which has been suggested as a possible explanation for some of the effects of both salicylic acid and aspirin.[57][58] The acetyl portion of the aspirin molecule has its own targets. Acetylation of cellular proteins is a well-established phenomenon in the regulation of protein function at the post-translational level. Aspirin is able to acetylate several other targets in addition to COX isoenzymes.[59][60] These acetylation reactions may explain many hitherto unexplained effects of aspirin.[61]
Formulations
This section needs expansion. You can help by adding to it. (January 2023) |
Aspirin is produced in many formulations, with some differences in effect. In particular, aspirin can cause gastrointestinal bleeding, and formulations are sought which deliver the benefits of aspirin while mitigating harmful bleeding. Formulations may be combined (e.g., buffered + vitamin C).
- Tablets, typically of about 75–100 mg and 300–320 mg of immediate-release aspirin (IR-ASA).
- Dispersible tablets.
- Enteric-coated tablets.
- Buffered formulations containing aspirin with one of many buffering agents.
- Formulations of aspirin with vitamin C (ASA-VitC)
- A phospholipid-aspirin complex liquid formulation, PL-ASA. As of 2023[update] the phospholipid coating was being trialled to determine if it caused less gastrointestinal damage.[62]
Pharmacokinetics
Acetylsalicylic acid is a
About 50–80% of salicylate in the blood is bound to human serum albumin, while the rest remains in the active, ionized state; protein binding is concentration-dependent. Saturation of binding sites leads to more free salicylate and increased toxicity. The volume of distribution is 0.1–0.2 L/kg. Acidosis increases the volume of distribution because of enhancement of tissue penetration of salicylates.[65]
As much as 80% of therapeutic doses of salicylic acid is metabolized in the liver. Conjugation with glycine forms salicyluric acid, and with glucuronic acid to form two different glucuronide esters. The conjugate with the acetyl group intact is referred to as the acyl glucuronide; the deacetylated conjugate is the phenolic glucuronide. These metabolic pathways have only a limited capacity. Small amounts of salicylic acid are also hydroxylated to gentisic acid. With large salicylate doses, the kinetics switch from first-order to zero-order, as metabolic pathways become saturated and renal excretion becomes increasingly important.[65]
Salicylates are excreted mainly by the kidneys as salicyluric acid (75%), free salicylic acid (10%), salicylic phenol (10%), and acyl glucuronides (5%), gentisic acid (< 1%), and 2,3-dihydroxybenzoic acid.[66] When small doses (less than 250 mg in an adult) are ingested, all pathways proceed by first-order kinetics, with an elimination half-life of about 2.0 h to 4.5 h.[67][68] When higher doses of salicylate are ingested (more than 4 g), the half-life becomes much longer (15 h to 30 h),[69] because the biotransformation pathways concerned with the formation of salicyluric acid and salicyl phenolic glucuronide become saturated.[70] Renal excretion of salicylic acid becomes increasingly important as the metabolic pathways become saturated, because it is extremely sensitive to changes in urinary pH. A 10- to 20-fold increase in renal clearance occurs when urine pH is increased from 5 to 8. The use of urinary alkalinization exploits this particular aspect of salicylate elimination.[71] It was found that short-term aspirin use in therapeutic doses might precipitate reversible acute kidney injury when the patient was ill with glomerulonephritis or cirrhosis.[72] Aspirin for some patients with chronic kidney disease and some children with congestive heart failure was contraindicated.[72]
History

Medicines made from

In 1853, chemist Charles Frédéric Gerhardt treated sodium salicylate with acetyl chloride to produce acetylsalicylic acid for the first time;[14]: 46–48 in the second half of the 19th century, other academic chemists established the compound's chemical structure and devised more efficient methods of synthesis. In 1897, scientists at the drug and dye firm Bayer began investigating acetylsalicylic acid as a less-irritating replacement for standard common salicylate medicines, and identified a new way to synthesize it.[14]: 69–75 That year, Felix Hoffmann (or Arthur Eichengrün) of Bayer was the first to produce acetylsalicylic acid in a pure, stable form.[15]
Salicylic acid had been extracted in 1838 from the herb meadowsweet, whose German name, Spirsäure, was the basis for naming the newly synthesized drug, which, by 1899, Bayer was selling globally.[14]: 46–55 [16]: 27 The word Aspirin was Bayer's brand name, rather than the generic name of the drug; however, Bayer's rights to the trademark were lost or sold in many countries. Aspirin's popularity grew over the first half of the 20th century, leading to fierce competition with the proliferation of aspirin brands and products.[22]
Aspirin's popularity declined after the development of
Aspirin sales revived considerably in the last decades of the 20th century, and remain strong in the 21st century with widespread use as a preventive treatment for
: 267–269Trademark
Bayer lost its trademark for Aspirin in the United States and some other countries in actions taken between 1918 and 1921 because it had failed to use the name for its own product correctly and had for years allowed the use of "Aspirin" by other manufacturers without defending the intellectual property rights.[78] Today, aspirin is a generic trademark in many countries.[79][80] Aspirin, with a capital "A", remains a registered trademark of Bayer in Germany, Canada, Mexico, and in over 80 other countries, for acetylsalicylic acid in all markets, but using different packaging and physical aspects for each.[81][82]
Compendial status
Medical use
Aspirin is used in the treatment of a number of conditions, including fever, pain, rheumatic fever, and inflammatory conditions, such as rheumatoid arthritis, pericarditis, and Kawasaki disease.[11] Lower doses of aspirin have also been shown to reduce the risk of death from a heart attack, or the risk of stroke in people who are at high risk or who have cardiovascular disease, but not in elderly people who are otherwise healthy.[85][86][87][88][89] There is evidence that aspirin is effective at preventing colorectal cancer, though the mechanisms of this effect are unclear.[90]
Pain
Aspirin is an effective analgesic for acute pain, although it is generally considered inferior to
Aspirin, either by itself or in a combined formulation, effectively treats certain types of a headache, but its efficacy may be questionable for others. Secondary headaches, meaning those caused by another disorder or trauma, should be promptly treated by a medical provider. Among primary headaches, the International Classification of Headache Disorders distinguishes between tension headache (the most common), migraine, and cluster headache. Aspirin or other over-the-counter analgesics are widely recognized as effective for the treatment of tension headaches.[97] Aspirin, especially as a component of an aspirin/paracetamol/caffeine combination, is considered a first-line therapy in the treatment of migraine, and comparable to lower doses of sumatriptan. It is most effective at stopping migraines when they are first beginning.[98]
Fever
Like its ability to control pain, aspirin's ability to control
Inflammation
Aspirin is used as an
Heart attacks and strokes
Aspirin is an important part of the treatment of those who have had a heart attack.[105] It is generally not recommended for routine use by people with no other health problems, including those over the age of 70.[106]
The 2009 Antithrombotic Trialists' Collaboration published in Lancet evaluated the efficacy and safety of low dose aspirin in secondary prevention. In those with prior ischaemic stroke or acute myocardial infarction, daily low dose aspirin was associated with a 19% relative risk reduction of serious cardiovascular events (non-fatal myocardial infarction, non-fatal stroke, or vascular death). This did come at the expense of a 0.19% absolute risk increase in gastrointestinal bleeding; however, the benefits outweigh the hazard risk in this case.[citation needed] Data from previous trials have suggested that weight-based dosing of aspirin has greater benefits in primary prevention of cardiovascular outcomes.[107] However, more recent trials were not able to replicate similar outcomes using low dose aspirin in low body weight (<70 kg) in specific subset of population studied i.e. elderly and diabetic population, and more evidence is required to study the effect of high dose aspirin in high body weight (≥70 kg).[108][109][110]
After
The status of the use of aspirin for the primary prevention in cardiovascular disease is conflicting and inconsistent, with recent changes from previously recommending it widely decades ago, and that some referenced newer trials in clinical guidelines show less of benefit of adding aspirin alongside other anti-hypertensive and cholesterol lowering therapies.[106][118] The ASCEND study demonstrated that in high-bleeding risk diabetics with no prior cardiovascular disease, there is no overall clinical benefit (12% decrease in risk of ischaemic events v/s 29% increase in GI bleeding) of low dose aspirin in preventing the serious vascular events over a period of 7.4 years. Similarly, the results of the ARRIVE study also showed no benefit of same dose of aspirin in reducing the time to first cardiovascular outcome in patients with moderate risk of cardiovascular disease over a period of five years. Aspirin has also been suggested as a component of a polypill for prevention of cardiovascular disease.[119][120] Complicating the use of aspirin for prevention is the phenomenon of aspirin resistance.[121][122] For people who are resistant, aspirin's efficacy is reduced.[123] Some authors have suggested testing regimens to identify people who are resistant to aspirin.[124]
As of April 2022[update], the
Cancer prevention
Aspirin may reduce the overall risk of both getting cancer and dying from cancer.[128] There is substantial evidence for lowering the risk of colorectal cancer (CRC),[90][129][130][131] but aspirin must be taken for at least 10–20 years to see this benefit.[132] It may also slightly reduce the risk of endometrial cancer[133] and prostate cancer.[134]
Some conclude the benefits are greater than the risks due to bleeding in those at average risk.[128] Others are unclear if the benefits are greater than the risk.[135][136] Given this uncertainty, the 2007 United States Preventive Services Task Force (USPSTF) guidelines on this topic recommended against the use of aspirin for prevention of CRC in people with average risk.[137] Nine years later however, the USPSTF issued a grade B recommendation for the use of low-dose aspirin (75 to 100 mg/day) "for the primary prevention of CVD [cardiovascular disease] and CRC in adults 50 to 59 years of age who have a 10% or greater 10-year CVD risk, are not at increased risk for bleeding, have a life expectancy of at least 10 years, and are willing to take low-dose aspirin daily for at least 10 years".[138]
A meta-analysis through 2019 said that there was an association between taking aspirin and lower risk of cancer of the colorectum, esophagus, and stomach.[139]
In 2021, the U.S. Preventive services Task Force raised questions about the use of aspirin in cancer prevention. It notes the results of the 2018 ASPREE (Aspirin in Reducing Events in the Elderly) Trial, in which the risk of cancer-related death was higher in the aspirin-treated group than in the placebo group.[140]
In 2025, a group of scientists at the
Psychiatry
Bipolar disorder
Aspirin, along with several other agents with anti-inflammatory properties, has been repurposed as an add-on treatment for depressive episodes in subjects with bipolar disorder in light of the possible role of inflammation in the pathogenesis of severe mental disorders.[145] A 2022 systematic review concluded that aspirin exposure reduced the risk of depression in a pooled cohort of three studies (HR 0.624, 95% CI: 0.0503, 1.198, P=0.033). However, further high-quality, longer-duration, double-blind randomized controlled trials (RCTs) are needed to determine whether aspirin is an effective add-on treatment for bipolar depression.[146][147][148] Thus, notwithstanding the biological rationale, the clinical perspectives of aspirin and anti-inflammatory agents in the treatment of bipolar depression remain uncertain.[145]
Dementia
Although cohort and longitudinal studies have shown low-dose aspirin has a greater likelihood of reducing the incidence of dementia, numerous randomized controlled trials have not validated this.[149][150]
Schizophrenia
Some researchers have speculated the anti-inflammatory effects of aspirin may be beneficial for schizophrenia. Small trials have been conducted but evidence remains lacking.[151][152]
Other uses
Aspirin is a first-line treatment for the fever and joint-pain symptoms of acute rheumatic fever. The therapy often lasts for one to two weeks, and is rarely indicated for longer periods. After fever and pain have subsided, the aspirin is no longer necessary, since it does not decrease the incidence of heart complications and residual rheumatic heart disease.[153][154] Naproxen has been shown to be as effective as aspirin and less toxic, but due to the limited clinical experience, naproxen is recommended only as a second-line treatment.[153][155]
Along with rheumatic fever, Kawasaki disease remains one of the few indications for aspirin use in children[156] in spite of a lack of high quality evidence for its effectiveness.[157]
Low-dose aspirin supplementation has moderate benefits when used for prevention of pre-eclampsia.[158][159] This benefit is greater when started in early pregnancy.[160]
Aspirin has also demonstrated
Resistance
For some people, aspirin does not have as strong an effect on platelets as for others, an effect known as aspirin-resistance or insensitivity. One study has suggested women are more likely to be resistant than men,[162] and a different, aggregate study of 2,930 people found 28% were resistant.[163] A study in 100 Italian people found, of the apparent 31% aspirin-resistant subjects, only 5% were truly resistant, and the others were
Meta-analysis and systematic reviews have concluded that laboratory confirmed aspirin resistance confers increased rates of poorer outcomes in cardiovascular and neurovascular diseases.[166][163][167][168][169][170] Although the majority of research conducted has surrounded cardiovascular and neurovascular, there is emerging research into the risk of aspirin resistance after orthopaedic surgery where aspirin is used for venous thromboembolism prophylaxis.[171] Aspirin resistance in orthopaedic surgery, specifically after total hip and knee arthroplasties, is of interest as risk factors for aspirin resistance are also risk factors for venous thromboembolisms and osteoarthritis; the sequelae of requiring a total hip or knee arthroplasty. Some of these risk factors include obesity, advancing age, diabetes mellitus, dyslipidemia and inflammatory diseases.[171]
Dosages
Adult aspirin tablets are produced in standardised sizes, which vary slightly from country to country, for example 300 mg in Britain and 325 mg in the United States. Smaller doses are based on these standards, e.g., 75 mg and 81 mg tablets. The 81 mg tablets are commonly called "baby aspirin" or "baby-strength", because they were originally – but no longer – intended to be administered to infants and children.[172] No medical significance occurs due to the slight difference in dosage between the 75 mg and the 81 mg tablets. The dose required for benefit appears to depend on a person's weight.[107] For those weighing less than 70 kilograms (154 lb), low dose is effective for preventing cardiovascular disease; for patients above this weight, higher doses are required.[107]
In general, for adults, doses are taken four times a day for fever or arthritis,[173] with doses near the maximal daily dose used historically for the treatment of rheumatic fever.[174] For the prevention of myocardial infarction (MI) in someone with documented or suspected coronary artery disease, much lower doses are taken once daily.[173]
March 2009 recommendations from the USPSTF on the use of aspirin for the primary prevention of coronary heart disease encourage men aged 45–79 and women aged 55–79 to use aspirin when the potential benefit of a reduction in MI for men or stroke for women outweighs the potential harm of an increase in gastrointestinal hemorrhage.[175][176][needs update] The WHI study of postmenopausal women found that aspirin resulted in a 25% lower risk of death from cardiovascular disease and a 14% lower risk of death from any cause, though there was no significant difference between 81 mg and 325 mg aspirin doses.[177] The 2021 ADAPTABLE study also showed no significant difference in cardiovascular events or major bleeding between 81 mg and 325 mg doses of aspirin in patients (both men and women) with established cardiovascular disease.[178]
Low-dose aspirin use was also associated with a trend toward lower risk of cardiovascular events, and lower aspirin doses (75 or 81 mg/day) may optimize efficacy and safety for people requiring aspirin for long-term prevention.[176]
In children with Kawasaki disease, aspirin is taken at dosages based on body weight, initially four times a day for up to two weeks and then at a lower dose once daily for a further six to eight weeks.[179]
Adverse effects
In October 2020, the US Food and Drug Administration (FDA) required the drug label to be updated for all nonsteroidal anti-inflammatory medications to describe the risk of kidney problems in unborn babies that result in low amniotic fluid.[180][181] They recommend avoiding NSAIDs in pregnant women at 20 weeks or later in pregnancy.[180][181] One exception to the recommendation is the use of low-dose 81 mg aspirin at any point in pregnancy under the direction of a health care professional.[181]
Contraindications
Aspirin should not be taken by people who are allergic to
Gastrointestinal
Aspirin increases the risk of upper gastrointestinal bleeding.[193] Enteric coating on aspirin may be used in manufacturing to prevent release of aspirin into the stomach to reduce gastric harm, but enteric coating does not reduce gastrointestinal bleeding risk.[193][194] Enteric-coated aspirin may not be as effective at reducing blood clot risk.[195][196] Combining aspirin with other NSAIDs has been shown to further increase the risk of gastrointestinal bleeding.[193] Using aspirin in combination with clopidogrel or warfarin also increases the risk of upper gastrointestinal bleeding.[197]
Blockade of COX-1 by aspirin apparently results in the upregulation of COX-2 as part of a gastric defense.[198] There is no clear evidence that simultaneous use of a COX-2 inhibitor with aspirin may increase the risk of gastrointestinal injury.[199]
"Buffering" is an additional method used with the intent to mitigate gastrointestinal bleeding, such as by preventing aspirin from concentrating in the walls of the stomach, although the benefits of buffered aspirin are disputed.[200] Almost any buffering agent used in antacids can be used; Bufferin, for example, uses magnesium oxide. Other preparations use calcium carbonate.[201] Gas-forming agents in effervescent tablet and powder formulations can also double as a buffering agent, one example being sodium bicarbonate, used in Alka-Seltzer.[202]
Taking vitamin C with aspirin has been investigated as a method of protecting the stomach lining. In trials vitamin C-releasing aspirin (ASA-VitC) or a buffered aspirin formulation containing vitamin C was found to cause less stomach damage than aspirin alone.[203][204]
Retinal vein occlusion
It is a widespread habit among eye specialists (ophthalmologists) to prescribe aspirin as an add-on medication for patients with retinal vein occlusion (RVO), such as central retinal vein occlusion (CRVO) and branch retinal vein occlusion (BRVO). The reason of this widespread use is the evidence of its proven effectiveness in major systemic venous thrombotic disorders, and it has been assumed that may be similarly beneficial in various types of retinal vein occlusion.
However, a large-scale investigation based on data of nearly 700 patients showed "that aspirin or other antiplatelet aggregating agents or anticoagulants adversely influence the visual outcome in patients with CRVO and hemi-CRVO, without any evidence of protective or beneficial effect".[205] Several expert groups, including the Royal College of Ophthalmologists, recommended against the use of antithrombotic drugs (incl. aspirin) for patients with RVO.[206]
Central effects
Large doses of
Reye's syndrome
Reye's syndrome, a rare but severe illness characterized by acute
Skin
For a small number of people, taking aspirin can result in symptoms including hives, swelling, and headache.[210] Aspirin can exacerbate symptoms among those with chronic hives, or create acute symptoms of hives.[211] These responses can be due to allergic reactions to aspirin, or more often due to its effect of inhibiting the COX-1 enzyme.[211][212] Skin reactions may also tie to systemic contraindications, seen with NSAID-precipitated bronchospasm,[211][212] or those with atopy.[213]
Aspirin and other NSAIDs, such as ibuprofen, may delay the healing of skin wounds.[214] Earlier findings from two small, low-quality trials suggested a benefit with aspirin (alongside compression therapy) on venous leg ulcer healing time and leg ulcer size,[215][216][217] however larger, more recent studies of higher quality have been unable to corroborate these outcomes.[218][219] As such, further research is required to clarify the role of aspirin in this context.
Other adverse effects
Aspirin can induce swelling of skin tissues in some people. In one study, angioedema appeared one to six hours after ingesting aspirin in some of the people. However, when the aspirin was taken alone, it did not cause angioedema in these people; the aspirin had been taken in combination with another NSAID-induced drug when angioedema appeared.[220]
Aspirin causes an increased risk of cerebral microbleeds, having the appearance on
A study of a group with a mean dosage of aspirin of 270 mg per day estimated an average absolute risk increase in intracerebral hemorrhage (ICH) of 12 events per 10,000 persons.[223] In comparison, the estimated absolute risk reduction in myocardial infarction was 137 events per 10,000 persons, and a reduction of 39 events per 10,000 persons in ischemic stroke.[223] In cases where ICH already has occurred, aspirin use results in higher mortality, with a dose of about 250 mg per day resulting in a relative risk of death within three months after the ICH around 2.5 (95% confidence interval 1.3 to 4.6).[224]
Aspirin and other NSAIDs can cause
Use of low-dose aspirin before a surgical procedure has been associated with an increased risk of bleeding events in some patients, however, ceasing aspirin prior to surgery has also been associated with an increase in major adverse cardiac events. An analysis of multiple studies found a three-fold increase in adverse events such as myocardial infarction in patients who ceased aspirin prior to surgery. The analysis found that the risk is dependent on the type of surgery being performed and the patient indication for aspirin use.[226]
On 9 July 2015, the US
Overdose
Aspirin overdose can be acute or chronic. In acute poisoning, a single large dose is taken; in chronic poisoning, higher than normal doses are taken over a period of time. Acute overdose has a
Interactions
Aspirin is known to
Research
The ISIS-2 trial demonstrated that aspirin at doses of 160 mg daily for one month, decreased the mortality by 21% of participants with a suspected myocardial infarction in the first five weeks.[241] A single daily dose of 324 mg of aspirin for 12 weeks has a highly protective effect against acute myocardial infarction and death in men with unstable angina.[242]
Bipolar disorder
Aspirin has been repurposed as an add-on treatment for depressive episodes in subjects with bipolar disorder.[145] However, meta-analytic evidence is based on very few studies and does not suggest any efficacy of aspirin in the treatment of bipolar depression.[145] Thus, notwithstanding the biological rationale, the clinical perspectives of aspirin and anti-inflammatory agents in the treatment of bipolar depression remain uncertain.[145]
Infectious diseases
Several studies investigated the anti-infective properties of aspirin for bacterial, viral and parasitic infections. Aspirin was demonstrated to limit platelet activation induced by Staphylococcus aureus and Enterococcus faecalis and to reduce streptococcal adhesion to heart valves. In patients with tuberculous meningitis, the addition of aspirin reduced the risk of new cerebral infarction [RR = 0.52 (0.29-0.92)]. A role of aspirin on bacterial and fungal biofilm is also being supported by growing evidence.[243]
Cancer prevention
Evidence from observational studies was conflicting on the effect of aspirin in breast cancer prevention;[244] a randomized controlled trial showed that aspirin had no significant effect in reducing breast cancer,[245] thus further studies are needed to clarify the effect of aspirin in cancer prevention.
In gardening
There are anecdotal reports that aspirin can improve the growth and resistance of plants,[246][247] though most research has involved salicylic acid instead of aspirin.[248]
Veterinary medicine
Aspirin is sometimes used in veterinary medicine as an anticoagulant or to relieve pain associated with musculoskeletal inflammation or osteoarthritis. Aspirin should be given to animals only under the direct supervision of a veterinarian, as adverse effects—including gastrointestinal issues—are common. An aspirin overdose in any species may result in salicylate poisoning, characterized by hemorrhaging, seizures, coma, and even death.[249]
Dogs are better able to tolerate aspirin than cats are.[250] Cats metabolize aspirin slowly because they lack the glucuronide conjugates that aid in the excretion of aspirin, making it potentially toxic if dosing is not spaced out properly.[249][251] No clinical signs of toxicosis occurred when cats were given 25 mg/kg of aspirin every 48 hours for 4 weeks,[250] but the recommended dose for relief of pain and fever and for treating blood clotting diseases in cats is 10 mg/kg every 48 hours to allow for metabolization.[249][252]
References
- PMID 3311247.
- ^ "Aspirin Use During Pregnancy". Drugs.com. 2 April 2018. Retrieved 29 December 2019.
- ^ "OTC medicine monograph: Aspirin tablets for oral use". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 4 April 2023.
- ^ "Poisons Standard October 2022". Australian Government Federal Register of Legislation. 26 September 2022. Retrieved 9 January 2023.
- ^ "Aspirin Product information". Health Canada. 22 October 2009. Retrieved 20 August 2023.
- ^ a b "Zorprin, Bayer Buffered Aspirin (aspirin) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Archived from the original on 7 April 2014. Retrieved 3 April 2014.
- ^ a b c Brayfield A, ed. (14 January 2014). "Aspirin". Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 3 April 2014.
- ^ CID 2244 from PubChem
- ^ ISBN 1-4398-5511-0.
- ^ "aspirin". Random House Webster's Unabridged Dictionary.
- ^ a b c d e f g h i j k l m n "Aspirin". American Society of Health-System Pharmacists. 29 November 2021. Archived from the original on 25 April 2017 – via Drugs.com.
- ^ ISBN 978-0-470-09290-3.
- ISBN 978-3-527-32669-3.
- ^ ISBN 978-1-59691-816-0. Archived from the originalon 8 September 2017.: 46–48
- ^ a b "Felix Hoffmann". Science History Institute. Retrieved 3 October 2024.
- ^ ISBN 978-0-394-57894-1.
- ^ PMID 12374850.
- hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Aspirin Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ Dick B (2018). "Hard Work and Happenstance". Distillations. Vol. 4, no. 1. Science History Institute. pp. 44–45.
- ^ a b c d e "Aspirin". Chemical & Engineering News. Vol. 83, no. 25. 20 June 2005.
- ISBN 978-0-85369-160-0.
- ^ "Acetylsalicylic acid". NIOSH Pocket Guide to Chemical Hazards. U.S. National Institute for Occupational Safety and Health (NIOSH). 11 April 2016. Archived from the original on 11 May 2017.
- ^ "Appendix G: 1989 Air contaminants update project – Exposure limits NOT in effect". NIOSH pocket guide to chemical hazards. National Institute for Occupational Safety and Health. 13 February 2015. Archived from the original on 18 June 2017.
- ISBN 978-0-471-28250-1.
- ^ "Chemical of the Week -- Acetic Acid and Acetic Anhydride". www.eng.uwaterloo.ca. Archived from the original on 3 November 2022.
- ^ Barrans R (18 May 2008). "Aspirin aging". Newton BBS. Archived from the original on 18 May 2008.
- PMID 3379589.
- ISBN 978-0-313-33758-1. Archivedfrom the original on 10 June 2013.
- ^ "Acetylsalicylic acid". Jinno Laboratory, School of Materials Science, Toyohashi University of Technology. 4 March 1996. Archived from the original on 20 January 2012. Retrieved 12 April 2014.
- ^ PMID 26031248.
- PMID 16316223.
- PMID 17139692.
- ^ "Polytypism - Online Dictionary of Crystallography". reference.iucr.org.
- ISSN 0022-2860.
- OSTI 1373897.
- PMID 5284360.
- PMID 14592543.
- ^ "The Nobel Prize in Physiology or Medicine 1982". Nobelprize.org. Archived from the original on 27 June 2017.
- ^ "Aspirin in heart attack and stroke prevention". American Heart Association. Archived from the original on 31 March 2008. Retrieved 8 May 2008.
- PMID 1412574.
- PMID 19482214.
- ^ Goel A, Aggarwal S, Partap S, Saurabh A, Choudhary (2012). "Pharmacokinetic solubility and dissolution profile of antiarrythmic drugs". Int J Pharma Prof Res. 3 (1): 592–601.
- PMID 7568157.
- PMID 17691994.
- S2CID 39103383.
- PMID 25895638.
- PMID 23747022.
- PMID 26546723.
- S2CID 44832283.
- PMID 15238606.
- PMID 16880431.
- ISSN 1094-2912.
...there was postprandial reduction on the plasma concentration of IL-6 and IkBα preservation, followed by the lower activation of NFκB, considered the main transcription factor capable of inducing inflammatory response by stimulating the expression of proinflammatory cytokines, chemokines, and adhesion molecules.
- PMID 29467962.
- PMID 20457564.
- PMID 22517326.
- .
- PMID 19212664.
- PMID 21475861.
- PMID 21475861.
- ISSN 0735-1097.
- PMID 5468267.
- S2CID 29427204.
- ^ PMID 5044917.
- PMID 3342084.
- PMID 6606362.
- S2CID 245036862.
- PMID 17364628.
- PMID 6291695.
- PMID 11971828.
- ^ PMID 8669425.
- ^ Goldberg DR (Summer 2009). "Aspirin: Turn of the Century Miracle Drug". Chemical Heritage. Vol. 27, no. 2. pp. 26–30.
- ^ Jeffreys D (2004). Aspirin: the Remarkable Story of a Wonder Drug. Bloomsbury. p. 18-34.
- PMID 14523031.
- PMID 4816857.
- PMID 4593555.
- ^ Bayer Co. v. United Drug Co., 272 F. 505, p.512 (S.D.N.Y 1921).
- ^ "Has aspirin become a generic trademark?". genericides.org. 25 March 2020. Archived from the original on 5 March 2021. Retrieved 17 February 2021.
- ISBN 978-0-521-47154-1. Archivedfrom the original on 15 October 2015.
- ^ "Aspirin: the versatile drug". CBC News. 28 May 2009. Archived from the original on 6 November 2016.
- PMID 17948100.
- Sigma Aldrich. Retrieved 24 January 2022.
- ^ "Index BP 2009" (PDF). British Pharmacopoeia. Archived from the original (PDF) on 11 April 2009. Retrieved 13 July 2009.
- ^
 This article incorporates text from this source, which is in the public domain: "Aspirin for reducing your risk of heart attack and stroke: know the facts". U.S. Food and Drug Administration (FDA). Archived from the original on 14 August 2012. Retrieved 26 July 2012.
This article incorporates text from this source, which is in the public domain: "Aspirin for reducing your risk of heart attack and stroke: know the facts". U.S. Food and Drug Administration (FDA). Archived from the original on 14 August 2012. Retrieved 26 July 2012.
- U.S. Preventive Services Task Force. Archived from the originalon 11 July 2012. Retrieved 26 July 2012.
- PMID 22231610.
- PMID 30221596.
- PMID 30221597.
- ^ PMID 22440112.
- PMID 15768621.
- PMID 14592563.
- PMID 25502052.
- PMID 10868553.
- S2CID 44459366.
- S2CID 10418793.
- PMID 18187725.
- PMID 21302868.
- PMID 20305990.
- S2CID 22361194.
- PMID 20000310.
- ^ "Medications Used to Treat Fever". American Academy of Pediatrics. 29 June 2012. Archived from the original on 18 February 2013. Retrieved 25 November 2012.
- ^ "51 FR 8180" (PDF). United States Federal Register. 51 (45). 7 March 1986. Archived from the original (PDF) on 19 August 2011.
- PMID 19597002.
- from the original on 31 December 2015.
- ^ PMID 30894318.
- ^ PMID 30017552.
- PMID 30146931.
- PMID 30221597.
- PMID 32223674.
- ^ National Guideline Clearinghouse (NGC). "2011 ACCF/AHA/SCAI guideline for percutaneous coronary artery intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions". United States Agency for Healthcare Research and Quality (AHRQ). Archived from the original on 13 August 2012. Retrieved 28 August 2012.
- S2CID 15459216.
- PMID 25718355.
- PMID 32795096.
- PMID 30522654.
- PMID 27036918.
- PMID 28886622.
- ^ PMID 34458905.
- PMID 16100022.
- PMID 16603580.
- PMID 16364973.
- PMID 20944898.
- PMID 21306212.
- PMID 19576352.
- ^
 This article incorporates text from this source, which is in the public domain: "Recommendation: Aspirin Use to Prevent Cardiovascular Disease: Preventive Medication". United States Preventive Services Taskforce. 23 November 2020. Retrieved 5 May 2022.
This article incorporates text from this source, which is in the public domain: "Recommendation: Aspirin Use to Prevent Cardiovascular Disease: Preventive Medication". United States Preventive Services Taskforce. 23 November 2020. Retrieved 5 May 2022.
- PMID 35471505.
- ^ Aubrey A, Stone W (26 April 2022). "Older adults shouldn't start a routine of daily aspirin, task force says". NPR.
- ^ PMID 25096604.
- PMID 22649288.
- PMID 22084361.
- S2CID 3332999.
- PMID 28577622.
- PMID 26701413.
- PMID 22517822.
- PMID 24074752.
- PMID 24714632.
- PMID 17339621.
- PMID 27064677.
- Lay summary in: "Who developed these recommendations?". Annals of Internal Medicine. 21 June 2016.
- PMID 32272209.
The present comprehensive meta-analysis supports and further quantifies the inverse association between regular aspirin use and the risk of colorectal and other digestive tract cancers, including some rare ones. The favorable effect of aspirin increases with longer duration of use, and, for colorectal cancer, with increasing dose.
- PMID 30221595.
- PMID 40044852.
- ^ "Scientists describe 'Eureka moment' over how aspirin prevents cancer spread". The Independent. 5 March 2025. Retrieved 5 March 2025.
- ^ "Scientists discover how aspirin could prevent some cancers from spreading". Medical Xpress. Retrieved 5 March 2025.
- ^ Cara E (5 March 2025). "A Surprising New Tool in the Fight Against Cancer: Aspirin". Gizmodo. Retrieved 5 March 2025.
- ^ S2CID 237485915.
- PMID 36678565.
- PMID 29362444.
- S2CID 56483705.
- PMID 33613260.
- PMID 28425093.
- PMID 31425623.
- PMID 32256401.
- ^ a b National Heart Foundation of Australia (RF/RHD guideline development working group) and the Cardiac Society of Australia and New Zealand (2006). "Diagnosis and management of acute rheumatic fever and rheumatic heart disease in Australia. An evidence-based review" (PDF). National Heart Foundation of Australia. pp. 33–37. Archived from the original (PDF) on 26 July 2008.
- PMID 18695275.
- PMID 14517527.
- PMID 20109049.
- PMID 17054199.
- PMID 31684684.
- S2CID 26372982.
- S2CID 3079979.
- PMID 36059148.
- S2CID 22245507.
- ^ PMID 18202034.
- S2CID 1776526.
- PMID 23212718.
- Lay summary in: Thomas K (4 December 2012). "Study Raises Questions on Coating of Aspirin". The New York Times.
- PMID 24201035.
- PMID 18242733.
- S2CID 38147668.
- S2CID 3485236.
- PMID 17698681.
- ^ S2CID 222234431.
- ^ "Invention of the safety cap". digitaldukemed.mc.duke.edu. Archived from the original on 4 March 2016. Retrieved 4 September 2015.
- ^ British Medical Journal and Royal Pharmaceutical Society of Great Britain. 2003.
- ^ "Aspirin monograph: dosages, etc". Medscape.com. Retrieved 11 May 2011.
- PMID 19293072.
- ^ a b "Aspirin: more evidence that low dose is all that is needed". Medscape. Medscape CME. Retrieved 11 May 2011.
- PMID 20031819.
- S2CID 237214969.
- British Medical Journal and Royal Pharmaceutical Society. 2006.
- ^ a b
 This article incorporates text from this source, which is in the public domain: "FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S. Food and Drug Administration (FDA) (Press release). 15 October 2020. Retrieved 15 October 2020.
This article incorporates text from this source, which is in the public domain: "FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S. Food and Drug Administration (FDA) (Press release). 15 October 2020. Retrieved 15 October 2020.
- ^ a b c
 This article incorporates text from this source, which is in the public domain: "NSAIDs may cause rare kidney problems in unborn babies". U.S. Food and Drug Administration. 21 July 2017. Retrieved 15 October 2020.
This article incorporates text from this source, which is in the public domain: "NSAIDs may cause rare kidney problems in unborn babies". U.S. Food and Drug Administration. 21 July 2017. Retrieved 15 October 2020.
- ^ a b c d e "Aspirin information from Drugs.com". Drugs.com. Archived from the original on 9 May 2008. Retrieved 8 May 2008.
- ^ a b c "Oral Aspirin information". First DataBank. Archived from the original on 18 September 2000. Retrieved 8 May 2008.
- (PDF) from the original on 9 April 2011.
- PMID 8566739.
- ^ a b "PDR guide to over the counter (OTC) drugs". Archived from the original on 10 April 2008. Retrieved 28 April 2008.
- ISBN 978-0-19-503634-3.
- ^ "Dengue and dengue hemorrhagic fever: information for health care practitioners". Archived from the original on 17 March 2008. Retrieved 28 April 2008.
- PMID 23345599.
- PMID 35430328.
- S2CID 253194139.
- ^ PMID 12411346.
- ^ PMID 11007221.
- PMID 34466020.
- ^ Torborg L (4 December 2018). "Mayo Clinic Q and A: Coated aspirin may not be as effective at reducing blood clot risk". Mayo Clinic News Network.
- S2CID 8034371.
- PMID 17698822.
- S2CID 448875.
- PMID 17556027.
- PMID 37103016.
- ^ "General chemistry online: FAQ: Acids and bases: What is the buffer system in buffered aspirin?". Antoine.frostburg.edu. Archived from the original on 14 April 2011. Retrieved 11 May 2011.
- PMID 13883982.
- S2CID 22688422.
- PMID 17218764.
- PMID 24769221.
- PMID 26780742.
- PMID 12736364.
- ^ PMID 10228187.
- ^ "Reye's syndrome". Health A to Z. National Health Service. 14 September 2023. Retrieved 24 August 2024.
- ^ "Are You Sensitive to Aspirin? Here are Some Reasons Why". Health Essentials from Cleveland Clinic. 5 February 2015. Archived from the original on 25 October 2020. Retrieved 5 March 2020.
- ^ PMID 30420763.
- ^ S2CID 52276808.
- PMID 10674573.
- PMID 9777971.
- S2CID 912169.
- PMID 22437068.
- PMID 26889740.
- PMID 29175902.
- PMID 30325305.
- S2CID 29909460.
- PMID 19364926.
- PMID 19506128.
- ^ S2CID 22997730.
- PMID 16322483.
- ^ Medical knowledge self-assessment program for students 4, By American College of Physicians, Clerkship Directors in Internal Medicine, Nephrology 227, Item 29
- PMID 17053008.
- ^ a b "FDA strengthens warning of heart attack and stroke risk for non-steroidal anti-inflammatory drugs". U.S. Food and Drug Administration (FDA). 9 July 2015. Archived from the original on 11 July 2015. Retrieved 9 July 2015.
- ^ Kreplick LW (2001). "Salicylate toxicity in emergency medicine". Medscape. Archived from the original on 31 August 2012.
- S2CID 12738659. (primary source)
- ISBN 978-0-323-02845-5.
- S2CID 9843820.
- ISBN 978-0-9626523-8-7.
- ^ S2CID 48361086.
- PMID 18359332.
- ^ Baxter K, Preston CL (eds.). "Aspirin and Spironolactone". MedicinesComplete: Stockley's Drug Interactions. Royal Pharmaceutical Society.
- PMID 785608.
- ISBN 978-0-8385-0565-6.
- PMID 4490672.
- PMID 6811490.
- PMID 6818974.
- S2CID 21071664.
- PMID 6135989.
- PMID 35203473.
- PMID 32957311.
- PMID 15998890.
- ^ "Aspirin Water Helps Plants". cybercemetery.unt.edu.
- ^ "Gardens: drug therapy for plants | Gardening advice". The Guardian. 31 January 2016.
- ^ "Priming Plant Defenses with Aspirin-like Compound : USDA ARS". www.ars.usda.gov.
- ^ a b c Edwards SH. "Nonsteroidal Anti-inflammatory Drugs: Aspirin". Merck Veterinary Manual. Archived from the original on 18 December 2016. Retrieved 20 January 2018.
- ^ a b "Analgesics (toxicity)". Merck. Archived from the original on 11 April 2015. Retrieved 19 January 2018.
- ISBN 978-1-56053-461-7.
- ^ "Plants poisonous to livestock". Cornell University Department of Animal Science. Archived from the original on 16 August 2015. Retrieved 3 March 2016.
Further reading
- Desborough MJ, Keeling DM (June 2017). "The aspirin story - from willow to wonder drug". British Journal of Haematology. 177 (5): 674–683. S2CID 46794541.
- McTavish JR (1987). "What's in a name? Aspirin and the American Medical Association". Bulletin of the History of Medicine. 61 (3): 343–66. PMID 3311247.
- Ling G (2005). "Aspirin". How Products Are Made. Vol. 1. Thomson Gale.
- Jeffreys D (2004). Aspirin: the Remarkable Story of a Wonder Drug. Bloomsbury.
External links
 Media related to Aspirin at Wikimedia Commons
Media related to Aspirin at Wikimedia Commons


