Potassium dichromate

| |

| |
| Names | |
|---|---|
| IUPAC name
Potassium dichromate(VI)
| |
| Other names | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.029.005 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3288 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| K2Cr2O7 | |
| Molar mass | 294.185 g/mol |
| Appearance | red-orange crystalline solid |
| Odor | odorless |
| Density | 2.676 g/cm3, solid |
| Melting point | 398 °C (748 °F; 671 K) |
| Boiling point | 500 °C (932 °F; 773 K) decomposes |
| 4.9 g/100 mL (0 °C) 13 g/100 mL (20 °C) 102 g/100 mL (100 °C) | |
| Solubility | insoluble in alcohol, acetone. |
Refractive index (nD)
|
1.738 |
| Structure | |
Triclinic (α-form, <241.6 °C)
| |
Tetrahedral (for Cr)
| |
| Thermochemistry | |
Heat capacity (C)
|
219 J/mol[2] |
Std molar
entropy (S⦵298) |
291.2 J/(K·mol) |
Std enthalpy of (ΔfH⦵298)formation |
−2033 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
carcinogenic,[4] corrosive |
| GHS labelling: | |
     [3] [3]
| |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
25 mg/kg (oral, rat)[5] |
| Safety data sheet (SDS) | ICSC 1371 |
| Related compounds | |
Other anions
|
Potassium chromate Potassium molybdate Potassium tungstate |
Other cations
|
Ammonium dichromate Sodium dichromate |
Related compounds
|
Potassium permanganate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium dichromate,
Chemistry
Production
Potassium dichromate is usually prepared by the reaction of potassium chloride on sodium dichromate. Alternatively, it can be also obtained from potassium chromate by roasting chromite ore with potassium hydroxide. It is soluble in water and in the dissolution process it ionizes:
- K2Cr2O7 → 2 K+ + Cr
2O2−
7 - Cr
2O2−
7 + H2O ⇌ 2 CrO2−
4 + 2 H+
Reaction
Potassium dichromate is an oxidising agent in organic chemistry, and is milder than potassium permanganate. It is used to oxidize alcohols. It converts primary alcohols into aldehydes and, under more forcing conditions, into carboxylic acids. In contrast, potassium permanganate tends to give carboxylic acids as the sole products. Secondary alcohols are converted into ketones. For example, menthone may be prepared by oxidation of menthol with acidified dichromate.[7] Tertiary alcohols cannot be oxidized.
In an aqueous solution the color change exhibited can be used to test for distinguishing aldehydes from ketones. Aldehydes reduce dichromate from the +6 to the +3 oxidation state, changing color from orange to green. This color change arises because the aldehyde can be oxidized to the corresponding carboxylic acid. A ketone will show no such change because it cannot be oxidized further, and so the solution will remain orange.
When heated strongly, it decomposes with the evolution of oxygen.
When an alkali is added to an orange-red solution containing dichromate ions, a yellow solution is obtained due to the formation of chromate ions (CrO2−4). For example, potassium chromate is produced industrially using potash:
- K2Cr2O7 + K2CO3 → 2 K2CrO4 + CO2
The reaction is reversible.
Treatment with cold
- K2Cr2O7 + 2 H2SO4 → 2 CrO3 + 2 KHSO4 + H2O
On heating with concentrated acid, oxygen is evolved:
Uses
Potassium dichromate has few major applications, as the sodium salt is dominant industrially. The main use is as a precursor to
Cleaning
Like other
Construction
It is used as an ingredient in
Photography and printing
In 1839,
These discoveries soon led to the
Dichromated colloids were also used as photoresists in various industrial applications, most widely in the creation of metal printing plates for use in photomechanical printing processes.
Chromium intensification or Photochromos uses potassium dichromate together with equal parts of concentrated
A potassium dichromate solution in sulfuric acid can be used to produce a reversal negative (that is, a positive transparency from a negative film). This is effected by developing a black and white film but allowing the development to proceed more or less to the end point. The development is then stopped by copious washing and the film then treated in the acid dichromate solution. This converts the silver metal to silver sulfate, a compound that is insensitive to light. After thorough washing and exposure to actinic light, the film is developed again allowing the previously unexposed silver halide to be reduced to silver metal. The results obtained can be unpredictable, but sometimes excellent results are obtained producing images that would otherwise be unobtainable. This process can be coupled with
In
Analytical reagent
Because it is non-hygroscopic, potassium dichromate is a common reagent in classical "wet tests" in analytical chemistry.
Ethanol determination

The concentration of ethanol in a sample can be determined by
- CH3CH2OH + 2[O] → CH3COOH + H2O
Full reaction of converting ethanol to acetic acid:
- 3 C2H5OH + 2 K2Cr2O7 + 8 H2SO4 → 3 CH3COOH + 2 Cr2(SO4)3 + 2 K2SO4 + 11 H2O
The excess dichromate is determined by titration against
One major application for this
Silver test
When dissolved in an approximately 35%
Sulfur dioxide test
Potassium dichromate paper can be used to test for sulfur dioxide, as it turns distinctively from orange to green. This is typical of all redox reactions where hexavalent chromium is reduced to trivalent chromium. Therefore, it is not a conclusive test for sulfur dioxide. The final product formed is Cr2(SO4)3.
- SO2 + K2Cr2O7 + 3H2SO4 → K2SO4 + Cr2(SO4)3 + 3 H2O
Wood treatment
Potassium dichromate is used to stain certain types of wood by darkening the tannins in the wood. It produces deep, rich browns that cannot be achieved with modern color dyes. It is a particularly effective treatment on mahogany.[10]
Natural occurrence

Potassium dichromate occurs naturally as the rare
Safety
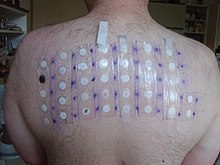
In 2005–06, potassium dichromate was the 11th-most-prevalent allergen in patch tests (4.8%).[12]
Potassium dichromate is one of the most common causes of chromium dermatitis;[13] chromium is highly likely to induce sensitization leading to dermatitis, especially of the hand and forearms, which is chronic and difficult to treat. Toxicological studies have further illustrated its highly toxic nature. With rabbits and rodents, concentrations as low as 14 mg/kg have shown a 50% fatality rate amongst test groups.[14] Aquatic organisms are especially vulnerable if exposed, and hence responsible disposal according to the local environmental regulations is advised.
As with other
References
- ^ "POTASSIUM DICHROMATE LISTING" (PDF). US EPA. 2015-07-23.
- ISBN 978-3-527-30524-7.
- ^ Sigma-Aldrich Co., Chromium(VI) oxide. Retrieved on 2014-06-15.
- ^ Like all compounds of hexavalent chromium, potassium dichromate is carcinogenic
- ^ Chambers, Michael. "ChemIDplus - 7778-50-9 - KMUONIBRACKNSN-UHFFFAOYSA-N - Potassium dichromate - Similar structures search, synonyms, formulas, resource links, and other chemical information".
- ^
- ^ L. T. Sandborn. "l-Menthone". Organic Syntheses; Collected Volumes, vol. 1, p. 340.
- S2CID 23159708.
- S2CID 27027304.
- ISBN 978-1-56158-154-2.
- ^ "Lópezite: Lópezite mineral information and data".
- ^ Zug KA, Warshaw EM, Fowler JF Jr, Maibach HI, Belsito DL, Pratt MD, Sasseville D, Storrs FJ, Taylor JS, Mathias CG, Deleo VA, Rietschel RL, Marks J. Patch-test results of the North American Contact Dermatitis Group 2005–2006. Dermatitis. 2009 May–Jun;20(3):149-60.
- ISBN 978-81-7021-136-5.
- ^ "Potassium dichromate MSDS". Sigma-Aldrich. Retrieved 2011-07-20.
- ISBN 978-92-832-0135-9. Retrieved 2020-01-05.
There is sufficient evidence in humans for the carcinogenicity of chromium (VI) compounds. Chromium (VI) compounds cause cancer of the lung. Also positive associations have been observed between exposure to Chromium (VI) compounds and cancer of the nose and nasal sinuses. There is sufficient evidence in experimental animals for the carcinogenicity of chromium (VI) compounds. Chromium (VI) compounds are carcinogenic to humans (Group 1).
- ^ "Potassium dichromate MSDS". JT Baker.
External links
- Potassium Dichromate at The Periodic Table of Videos(University of Nottingham)
- International Chemical Safety Card 1371
- National Pollutant Inventory – Chromium VI and compounds fact sheet
- NIOSH Pocket Guide to Chemical Hazards
- IARC Monograph "Chromium and Chromium compounds"
- Gold refining article listing color change when testing metals with Schwerter's Solution

