Potassium chlorate
| |||

| |||
| Names | |||
|---|---|---|---|
| Other names
Potassium chlorate(V), Potcrate, Berthollet salt
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.021.173 | ||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1485 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
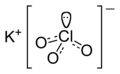
| KClO3 | |||
| Molar mass | 122.55 g mol−1 | ||
| Appearance | white crystals or powder | ||
| Density | 2.32 g/cm3 | ||
| Melting point | 356 °C (673 °F; 629 K) | ||
| Boiling point | 400 °C (752 °F; 673 K) decomposes[1] | ||
| 3.13 g/100 mL (0 °C) 4.46 g/100 mL (10 °C) 8.15 g/100 mL (25 °C) 13.21 g/100 mL (40 °C) 53.51 g/100 mL (100 °C) 183 g/100 g (190 °C) 2930 g/100 g (330 °C)[2] | |||
| Solubility | soluble in glycerol negligible in acetone and liquid ammonia[1] | ||
| Solubility in glycerol | 1 g/100 g (20 °C)[1] | ||
| −42.8·10−6 cm3/mol | |||
Refractive index (nD)
|
1.40835 | ||
| Structure | |||
| monoclinic | |||
| Thermochemistry | |||
Heat capacity (C)
|
100.25 J/mol·K[1] | ||
Std molar
entropy (S⦵298) |
142.97 J/mol·K[3][1] | ||
Std enthalpy of (ΔfH⦵298)formation |
−391.2 kJ/mol[3][1] | ||
Gibbs free energy (ΔfG⦵)
|
-289.9 kJ/mol[1] | ||
| Hazards | |||
| GHS labelling: | |||
   [4] [4]
| |||
| Danger | |||
| H271, H302, H332, H411[4] | |||
| P220, P273[4] | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
1870 mg/kg (oral, rat)[5] | ||
| Safety data sheet (SDS) | ICSC 0548 | ||
| Related compounds | |||
Other anions
|
Potassium bromate Potassium iodate Potassium nitrate | ||
Other cations
|
Ammonium chlorate Sodium chlorate Barium chlorate | ||
Related compounds
|
Potassium chloride Potassium hypochlorite Potassium chlorite Potassium perchlorate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Potassium chlorate is a compound containing potassium, chlorine and oxygen, with the molecular formula KClO3. In its pure form, it is a white crystalline substance. After sodium chlorate, it is the second most common chlorate in industrial use. It is a strong oxidizing agent and its most important application is in safety matches.[6] In other applications it is mostly obsolete and has been replaced by safer alternatives in recent decades. It has been used
- in explosives,
- to prepare oxygen, both in the lab and in chemical oxygen generators,
- as a disinfectant, for example in medical mouthwashes,
- in agriculture as a herbicide.
Production
On the industrial scale, potassium chlorate is produced by the salt metathesis reaction of sodium chlorate and potassium chloride:
The reaction is driven by the low solubility of potassium chlorate in water. The equilibrium of the reaction is shifted to the right hand side by the continuous precipitation of the product (
The direct electrolysis of
Potassium chlorate can be produced in small amounts by disproportionation in a sodium hypochlorite solution followed by metathesis reaction with potassium chloride:[7]
It can also be produced by passing chlorine gas into a hot solution of caustic potash:[8]
Uses

Potassium chlorate was one key ingredient in early
Chlorate-based propellants are more efficient than traditional gunpowder and are less susceptible to damage by water. However, they can be extremely unstable in the presence of sulfur or phosphorus and are much more expensive. Chlorate propellants must be used only in equipment designed for them; failure to follow this precaution is a common source of accidents. Potassium chlorate, often in combination with silver fulminate, is used in trick noise-makers known as "crackers", "snappers", "pop-its", "caps" or "bang-snaps", a popular type of novelty firework.
Another application of potassium chlorate is as the oxidizer in a smoke composition such as that used in smoke grenades. Since 2005, a cartridge with potassium chlorate mixed with lactose and rosin is used for generating the white smoke signaling the election of new pope by a papal conclave.[9]
High school and college laboratories often use potassium chlorate to generate oxygen gas. [
Heating it in the absence of a catalyst converts it into potassium perchlorate:[8]
With further heating, potassium perchlorate decomposes to potassium chloride and oxygen:
The safe performance of this reaction requires very pure reagents and careful temperature control. Molten potassium chlorate is an extremely powerful oxidizer and spontaneously reacts with many common materials such as sugar. Explosions have resulted from liquid chlorates spattering into the latex or PVC tubes of oxygen generators and from contact between chlorates and hydrocarbon sealing greases. Impurities in potassium chlorate itself can also cause problems. When working with a new batch of potassium chlorate, it is advisable to take a small sample (~1 gram) and heat it strongly on an open glass plate. Contamination may cause this small quantity to explode, indicating that the chlorate should be discarded.
Potassium chlorate is used in chemical oxygen generators (also called chlorate candles or oxygen candles), employed as oxygen-supply systems of e.g. aircraft, space stations, and submarines, and has been responsible for at least one plane crash. A fire on the space station Mir was traced to oxygen generation candles that use a similar lithium perchlorate. The decomposition of potassium chlorate was also used to provide the oxygen supply for limelights.
Potassium chlorate is used also as a pesticide. In Finland it was sold under trade name Fegabit.
Potassium chlorate can react with sulfuric acid to form a highly reactive solution of chloric acid and potassium sulfate:
The solution so produced is sufficiently reactive that it spontaneously ignites if combustible material (sugar, paper, etc.) is present.
In schools, molten potassium chlorate is used in screaming jelly babies, Gummy bear, Haribo, and Trolli candy demonstration where the candy is dropped into the molten salt.
In chemical labs it is used to oxidize HCl and release small amounts of gaseous chlorine.
Militant groups in
Potassium chlorate is used to force the blossoming stage of the longan tree, causing it to produce fruit in warmer climates.[11]
Safety
Potassium chlorate should be handled with care. It reacts vigorously, and in some cases spontaneously ignites or explodes, when mixed with many
See also
References
- ^ a b c d e f g "potassium chlorate". Retrieved 9 July 2015.
- ^ Seidell, Atherton; Linke, William F. (1952). Solubilities of Inorganic and Organic Compounds. Van Nostrand. Retrieved 2014-05-29.
- ^ ISBN 978-0-618-94690-7.
- ^ a b c "Potassium chlorate". Retrieved 14 February 2022.
- ^ Michael Chambers. "ChemIDplus - 3811-04-9 - VKJKEPKFPUWCAS-UHFFFAOYSA-M - Potassium chlorate - Similar structures search, synonyms, formulas, resource links, and other chemical information". Retrieved 9 July 2015.
- ^ ISBN 9783527303854.
- ^ Anne Marie Helmenstine, Ph.D. "Potassium Chlorate Synthesis (Substitute) Formula". About.com Education. Retrieved 9 July 2015.
- ^ ISBN 0-07-049439-8
- ^ Daniel J. Wakin and Alan Cowell (March 13, 2013). "New Round of Voting Fails to Name a Pope". The New York Times. Retrieved March 13, 2013.
- ^ "Afghan bomb makers shifting to new explosives for IEDs". USAToday.com. June 25, 2013. Retrieved 2013-06-25.
- doi:10.1016/j.scienta.2005.01.004. Retrieved November 28, 2010.)
{{cite journal}}: CS1 maint: location (link
- "Chlorate de potassium. Chlorate de sodium", Fiche toxicol. n° 217, Paris:Institut national de recherche et de sécurité, 2000. 4pp.
- Continuous process for the manufacture of potassium chlorate by coupling with a sodium chlorate production plant







![{\displaystyle {\ce {2 KClO3(s) ->[\Delta][MnO_2] 3 O2(g) + 2 KCl(s)}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8bce0d11cf57ee2802e42385013f05fe6f1c58e3)


