Solvation

Solvation describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the solute, including solubility, reactivity, and color, as well as influencing the properties of the solvent such as its viscosity and density.[1] If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. The surrounded solute particles then move away from the solid solute and out into the solution. Ions are surrounded by a concentric shell of solvent. Solvation is the process of reorganizing solvent and solute molecules into solvation complexes and involves bond formation, hydrogen bonding, and van der Waals forces. Solvation of a solute by water is called hydration.[2]
Solubility of solid compounds depends on a competition between lattice energy and solvation, including entropy effects related to changes in the solvent structure.[3]
Distinction from solubility
By an
Solvation is, in concept, distinct from solubility. Solvation or dissolution is a kinetic process and is quantified by its rate. Solubility quantifies the dynamic equilibrium state achieved when the rate of dissolution equals the rate of precipitation. The consideration of the units makes the distinction clearer. The typical unit for dissolution rate is mol/s. The units for solubility express a concentration: mass per volume (mg/mL), molarity (mol/L), etc.[citation needed]
Solvents and intermolecular interactions
Solvation involves different types of intermolecular interactions:

Solvent
Hydrogen bonding among solvent and solute molecules depends on the ability of each to accept H-bonds, donate H-bonds, or both. Solvents that can donate H-bonds are referred to as protic, while solvents that do not contain a polarized bond to a hydrogen atom and cannot donate a hydrogen bond are called aprotic. H-bond donor ability is classified on a scale (α).[6] Protic solvents can solvate solutes that can accept hydrogen bonds. Similarly, solvents that can accept a hydrogen bond can solvate H-bond-donating solutes. The hydrogen bond acceptor ability of a solvent is classified on a scale (β).[7] Solvents such as water can both donate and accept hydrogen bonds, making them excellent at solvating solutes that can donate or accept (or both) H-bonds.
Some chemical compounds experience solvatochromism, which is a change in color due to solvent polarity. This phenomenon illustrates how different solvents interact differently with the same solute. Other solvent effects include conformational or isomeric preferences and changes in the acidity of a solute.
Solvation energy and thermodynamic considerations
The solvation process will be thermodynamically favored only if the overall
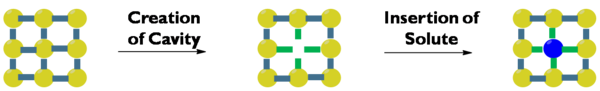
Solvation involves multiple steps with different energy consequences. First, a cavity must form in the solvent to make space for a solute. This is both entropically and enthalpically unfavorable, as solvent ordering increases and solvent-solvent interactions decrease. Stronger interactions among solvent molecules leads to a greater enthalpic penalty for cavity formation. Next, a particle of solute must separate from the bulk. This is enthalpically unfavorable since solute-solute interactions decrease, but when the solute particle enters the cavity, the resulting solvent-solute interactions are enthalpically favorable. Finally, as solute mixes into solvent, there is an entropy gain.[5]
The
Enthalpy of solvation can help explain why solvation occurs with some ionic lattices but not with others. The difference in energy between that which is necessary to release an ion from its lattice and the energy given off when it combines with a solvent molecule is called the
Although early thinking was that a higher ratio of a cation's ion charge to ionic radius, or the charge density, resulted in more solvation, this does not stand up to scrutiny for ions like iron(III) or lanthanides and actinides, which are readily hydrolyzed to form insoluble (hydrous) oxides. As these are solids, it is apparent that they are not solvated.
Strong solvent–solute interactions make the process of solvation more favorable. One way to compare how favorable the dissolution of a solute is in different solvents is to consider the free energy of transfer. The free energy of transfer quantifies the free energy difference between dilute solutions of a solute in two different solvents. This value essentially allows for comparison of solvation energies without including solute-solute interactions.[5]
In general, thermodynamic analysis of solutions is done by modeling them as reactions. For example, if you add sodium chloride to water, the salt will dissociate into the ions sodium(+aq) and chloride(-aq). The equilibrium constant for this dissociation can be predicted by the change in Gibbs energy of this reaction.
The Born equation is used to estimate Gibbs free energy of solvation of a gaseous ion.
Recent simulation studies have shown that the variation in solvation energy between the ions and the surrounding water molecules underlies the mechanism of the Hofmeister series.[9][1]
Macromolecules and assemblies
Solvation (specifically,
Solvation also affects host–guest complexation. Many host molecules have a hydrophobic pore that readily encapsulates a hydrophobic guest. These interactions can be used in applications such as drug delivery, such that a hydrophobic drug molecule can be delivered in a biological system without needing to covalently modify the drug in order to solubilize it. Binding constants for host–guest complexes depend on the polarity of the solvent.[12]
Hydration affects electronic and vibrational properties of biomolecules.[13][14]
Importance of solvation in computer simulations
Due to the importance of the effects of solvation on the structure of macromolecules, early computer simulations which attempted to model their behaviors without including the effects of solvent (in vacuo) could yield poor results when compared with experimental data obtained in solution. Small molecules may also adopt more compact conformations when simulated in vacuo; this is due to favorable van der Waals interactions and intramolecular electrostatic interactions which would be dampened in the presence of a solvent.
As computer power increased, it became possible to try and incorporate the effects of solvation within a simulation and the simplest way to do this is to surround the molecule being simulated with a "skin" of solvent molecules, akin to simulating the molecule within a drop of solvent if the skin is sufficiently deep.[15]
See also
- Saturated solution
- Solubility equilibrium
- Solvent models
- Born equation
- Supersaturation
- Water model
References
- ^ PMID 29611710.
- ISBN 978-0-13-201304-8.
- ISBN 978-0-08-037941-8.
- ^ ISBN 978-1-891389-31-3.
- .
- .
- .
- PMID 28816050.
- PMID 30384680.
- S2CID 20021399.
- ISBN 1118681509, 9781118681503.
- PMID 22443792.
- PMID 22594454.
- OCLC 45008511.
Further reading
- ISBN 0-444-42984-0(Chemistry)
- Jiang D., Urakawa A., Yulikov M., Mallat T., Jeschke G., Baiker A. (2009). "Size selectivity of a copper metal-organic framework and origin of catalytic activity in epoxide alcoholysis". Chemistry: A European Journal. 15 (45): 12255–62. PMID 19806616.) [One example of a solvated MOF, where partial dissolution is described.]
{{cite journal}}: CS1 maint: multiple names: authors list (link
External links
- Serafin, J.M. Transfer Free Energy and the Hydrophobic Effect. J. Chem. Educ. 2003, 80, 1194–1196 PDF.