Total dissolved solids

Total dissolved solids (TDS) is a measure of the
Generally, the operational definition is that the solids must be small enough to survive filtration through a filter with 2-micrometer (nominal size, or smaller) pores. Total dissolved solids are normally discussed only for
Primary sources for TDS in receiving waters are
Total dissolved solids are differentiated from total suspended solids (TSS), in that the latter cannot pass through a sieve of 2 micrometers and yet are indefinitely suspended in solution. The term settleable solids refers to material of any size that will not remain suspended or dissolved in a holding tank not subject to motion, and excludes both TDS and TSS.[2] Settleable solids may include larger particulate matter or insoluble molecules.
Total dissolved solids include both volatile and non-volatile solids. Volatile solids are ones that can easily go from a solid to a gaseous state. Non-volatile solids must be heated to a high temperature, typically 550 °C, in order to achieve this state change. Examples of non-volatile substances include salts and sugars.[3]
Measurement
This section needs additional citations for verification. (February 2016) |

The two principal methods of measuring total dissolved solids are gravimetric analysis and conductivity.[4] Gravimetric methods are the most accurate and involve evaporating the liquid solvent and measuring the mass of residues left. This method is generally the best, although it is time-consuming. If inorganic salts comprise the great majority of TDS, conductivity-based methods are appropriate.
Conductivity of water is directly related to the concentration of dissolved ionized solids. These ions allow the water to conduct
The relationship of TDS and specific conductance of groundwater can be approximated by the following equation:
- TDS = keEC
where TDS is expressed in mg/L and EC is the electrical conductivity in microsiemens per centimeter at 25 °C. The conversion factor ke varies between 0.55 and 0.8.[5]
Some TDS meters use an electrical conductivity measurement to the
Hydrological simulation

Hydrologic transport models are used to mathematically analyze movement of TDS within river systems. The most common models address surface runoff, allowing variation in
Basin models are used to more comprehensively evaluate total dissolved solids within a
Practical implications

When measuring water treated with
In the case of hydroponics and aquaculture, TDS is often monitored to create a water quality environment favorable for organism productivity. For freshwater, oysters, trouts, and other high-value seafood, the highest productivity and economic returns are achieved by mimicking the TDS and pH levels of each species' native environment. For hydroponic uses, total dissolved solids are considered one of the best indices of nutrient availability for the aquatic plants being grown.
Because the threshold of acceptable
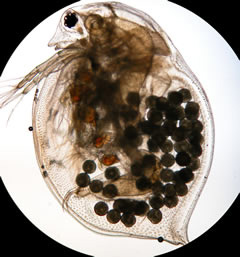
The fathead minnow (Pimephales promelas), for example, realizes an LD50 concentration of 5,600 ppm based upon a 96-hour exposure. LD50 is the concentration required to produce a lethal effect on 50 percent of the exposed population. Daphnia magna, a good example of a primary member of the food chain, is a small planktonic crustacean, about 0.5 mm (0.020 in) in length, having an LD50 of about 10,000 ppm TDS for a 96-hour exposure.[11]
For terrestrial animals, poultry typically possess a safe upper limit of TDS exposure of approximately 2,900 mg/L, whereas dairy cattle are measured to have a safe upper limit of about 7,100 mg/L. Research has shown that exposure to TDS is compounded in toxicity when other
In countries with often unsafe/unclean tap water supplies, technicians frequently check the TDS of drinking water to gauge how effectively their RO/Water Filtration devices are working. While TDS readings will not provide the number of microorganisms present in a sample of water, they can indicate how efficient the filter is by the presence of TDS.
Water classification
[14] Water can be classified by the level of total dissolved solids (TDS) in the water:
- Fresh water: TDS is less than 1,000 ppm.
- Brackish water: TDS = 1,000 to 10,000 ppm.
- Saline water: TDS = 10,000 to 35,000 ppm.
- Hypersaline: TDS greater than 35,000 ppm.
Drinking water generally has a TDS below 500 ppm. Higher TDS Fresh Water is drinkable but taste may be objectionable.
See also
- Acid rain
- Surface runoff
- Regarding meters;
- EC meter
- pH meter
- Salinometer
References
- ^ "What Is The Acceptable Total Dissolved Solids (TDS) Level In Drinking Water?". The Berkey. Archived from the original on 2020-02-22. Retrieved 2020-02-22.
- ISBN 0-471-28789-X.
- ^ Wetzel, R. G. (2001). Limnology: Lake and river ecosystems. San Diego: Academic Press.
- ^ "Total Dissolved Solids (TDS): EPA Method 160.1 (Gravimetric, Dried at 180 deg. C)". Washington, D.C.: U.S. Environmental Protection Agency (EPA). 1999-11-16. Archived from the original on 2016-02-23.
- doi:10.1016/j.jappgeo.2004.08.003. Archived from the original(PDF) on 1 August 2014. Retrieved 15 February 2016.
- ^ "Frequently Asked Questions". Archived from the original on 2017-06-18. Retrieved 23 May 2017.
- ^ a b C.M. Hogan, Marc Papineau et al. Development of a dynamic water quality simulation model for the Truckee River, Earth Metrics Inc., Environmental Protection Agency Technology Series, Washington D.C. (1987)
- ^ EPA. "Guidance for Water Quality-Based Decisions: The TMDL Process." Archived 2017-07-06 at the Wayback Machine Doc. No. EPA 440/4-91-001. April 1991.
- ^ W. Adam Sigler, Jim Bauder. "TDS Fact Sheet". Montana State University. Archived from the original on 2015-04-29. Retrieved 23 January 2015.
- ISBN 0-7923-7853-9.
- ^ Position Paper on Total Dissolved Solids, State of Iowa, IAC 567 61.3 (2)g et sequitur updated March 27, 2003
- ^ Kaiser Engineers, California, Final Report to the State of California, San Francisco Bay-Delta Water Quality Control Program, State of California, Sacramento, CA (1969)
- ^ Hogan, C. Michael; Patmore, Leda C.; Seidman, Harry (August 1973). "Statistical Prediction of Dynamic Thermal Equilibrium Temperatures using Standard Meteorological Data Bases". EPA. Archived from the original on 2016-02-23. Retrieved 2016-02-15.
{{cite journal}}: Cite journal requires|journal=(help) Environmental Protection Technology Series. Document No. EPA-660/2-73-003. - ^ "Saline Water and Salinity | U.S. Geological Survey". 13 November 2018. Archived from the original on 2020-08-18. Retrieved 2020-02-12.
External links
 Media related to Total dissolved solids at Wikimedia Commons
Media related to Total dissolved solids at Wikimedia Commons
