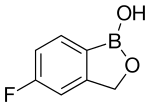
Tavaborole
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Kerydin |
| Other names | AN2690 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614049 |
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
Tavaborole, sold under the brand name Kerydin, is a
Medical uses
Tavaborole is used in the treatment of onychomycosis. In clinical trials, tavaborole was more effective than the vehicle (ethyl acetate and propylene glycol) alone in curing onychomycosis. In two studies, fungal infection was eliminated using tavaborole in 6.5% of the cases vs. 0.5% using the vehicle alone, and 27.5% vs. 14.6% using the vehicle alone.[1]
Side effects
Application site exfoliation, erythema (rash), and irritation are possible side effects, and may occur in less than 5% of individuals.[1]
Pharmacology
Tavaborole acts as an inhibitor of fungal leucyl-tRNA synthetase.[3]
Pharmacokinetics
Tavaborole, when prepared with a 1:1 mixture of ethyl acetate and propylene glycol, has the ability to fully penetrate through the human nail.[citation needed] In studies with cadaver fingernails, a 5% solution of tavaborole penetrated the nail an average of 524.7 mcg/cm2 after two weeks of daily use.[1]
Tavaborole is detectable in the blood at a level of 3.54 ng/mL after a single use of 0.2 mL of the 5% solution. Tavaborole has an
Society and culture
Economics
Tavaborole began phase III clinical trials in December 2010[4] and was approved by the US FDA in July 2014.[5]
Originally developed by
See also
- Crisaborole – a structurally related topical medication used for the treatment of mild-to-moderate atopic dermatitis (eczema)
References
- ^ PMID 25956661.
- ^ "FDA Approves Anacor Pharmaceuticals' KERYDIN™ (Tavaborole) Topical Solution, 5% for the Treatment of Onychomycosis of the Toenails". Market Watch. July 8, 2014.
- PMID 29547036.
- ^ Clinical trial number NCT01270971 for "Efficacy and Safety Evaluation of AN2690 Topical Solution to Treat Onychomycosis of the Toenail" at ClinicalTrials.gov
- ^ "FDA Approves Anacor Pharmaceuticals' KERYDIN™ (Tavaborole) Topical Solution, 5% for the Treatment of Onychomycosis of the Toenails". Market Watch. July 8, 2014.
- ^ "Anacor's Kerydin to Be Commercialized by Sandoz in the U.S." Zacks.com. 22 July 2014. Retrieved 7 October 2015.
- ^ "Kerydin". Retrieved 12 August 2015.
External links
- "Tavaborole". Drug Information Portal. U.S. National Library of Medicine.
