Source: Wikipedia, the free encyclopedia.
Verbascoside
Names
IUPAC name
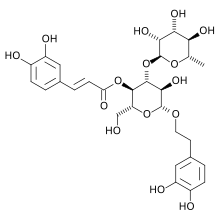
2-(3,4-Dihydroxyphenyl)ethyl α-L -rhamnopyranosyl-(1→4)-{5-O -[(2E )-3-(3,4-dihydroxymethyl)prop-2-enoyl]-β-D -glucopyranoside}
Systematic IUPAC name
(2R ,3R ,4R ,5R ,6R )-6-[2-(3,4-Dihydroxyphenyl)ethoxy]-5-hydroxy-2-(hydroxymethyl)-4-{[(2S ,3R ,4R ,5R ,6S )-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}oxan-3-yl (2E )-3-(3,4-dihydroxymethyl)prop-2-enoate
Other names
Acteoside
Identifiers
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard
100.112.547
UNII
InChI=1S/C29H36O15/c1-13-22(36)23(37)24(38)29(41-13)44-27-25(39)28(40-9-8-15-3-6-17(32)19(34)11-15)42-20(12-30)26(27)43-21(35)7-4-14-2-5-16(31)18(33)10-14/h2-7,10-11,13,20,22-34,36-39H,8-9,12H2,1H3/b7-4+/t13-,20+,22-,23+,24+,25+,26+,27+,28+,29-/m0/s1
N Key: FBSKJMQYURKNSU-ZLSOWSIRSA-N
N InChI=1/C29H36O15/c1-13-22(36)23(37)24(38)29(41-13)44-27-25(39)28(40-9-8-15-3-6-17(32)19(34)11-15)42-20(12-30)26(27)43-21(35)7-4-14-2-5-16(31)18(33)10-14/h2-7,10-11,13,20,22-34,36-39H,8-9,12H2,1H3/b7-4+/t13-,20+,22-,23+,24+,25+,26+,27+,28+,29-/m0/s1
Key: FBSKJMQYURKNSU-ZLSOWSIRBQ
Properties
C 29 H 36 O 15
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Verbascoside is a polyphenol glycoside[1] phenylpropanoid caffeic acid and the phenylethanoid hydroxytyrosol form an ester and an ether bond respectively, to the rhamnose part of a disaccharide, namely β-(3′,4′-dihydroxyphenyl)ethyl-O-α-L-rhamnopyranosyl(1→3)-β-D-(4-O-caffeoyl)-glucopyranoside.[2]
Occurrences Natural occurrences Verbascoside can be found in species in all the families of the order Lamiales (syn. Scrophulariales).[3] [4] Asterids .
in the Lamiales In the family
Pithecoctenium sp and
Tynanthus panurensis , in the family
Orobanchaceae , in
Cistanche sp and
Orobanche rapum-genistae ,
[2] in the
Plantaginaceae , in
Plantago lanceolata ,
[10] in
Verbenaceae , in
Verbena officinalis (common vervain),
[11] Aloysia citrodora (lemon verbena) and
Lantana camara ,
[12] in the
Oleaceae , in
Olea europaea (
olive ),
[13] in the
Lentibulariaceae , in the carnivorous plant
Pinguicula lusitanica ,
[4] and, in the Byblidaceae, in
Byblis liniflora .
[3] Derivatives Verbascoside derivatives can be found in the
In in vitro cultures It can also be produced in
Biological activity Verbascoside has an antimicrobial activity,[8] Staphylococcus aureus [9] anti-inflammatory properties.[7]
Although some
See also References
. ^ .
^ .^ .
. . ^ .
^ .
^ .
. . ^ .
. . . . . . .
Aglycones
Precursor Monohydroxycinnamic acids Dihydroxycinnamic acids Trihydroxycinnamic acids O -methylated formsothers
Esters
glycoside-likes
Esters of
Glycosides
Tartaric acid estersOther esters Caffeoyl phenylethanoid
Echinacoside Calceolarioside A , B , C , F Chiritoside A , B , C Cistanoside A , B , C , D , E , F , G , H Conandroside Myconoside Pauoifloside Plantainoside A Plantamajoside Tubuloside B Verbascoside (Isoverbascoside , 2′-Acetylverbascoside )
Oligomeric forms
Conjugates withcoenzyme A (CoA)