Apoptosis regulator BAX
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) |
| ||||||||
| RefSeq (protein) |
| ||||||||
| Location (UCSC) | Chr 19: 48.95 – 48.96 Mb | Chr 7: 45.11 – 45.12 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Apoptosis regulator BAX, also known as bcl-2-like protein 4, is a protein that in humans is encoded by the BAX gene.[5] BAX is a member of the Bcl-2 gene family. BCL2 family members form hetero- or homodimers and act as anti- or pro-apoptotic regulators that are involved in a wide variety of cellular activities. This protein forms a heterodimer with BCL2, and functions as an apoptotic activator. This protein is reported to interact with, and increase the opening of, the mitochondrial voltage-dependent anion channel (VDAC), which leads to the loss in membrane potential and the release of cytochrome c. The expression of this gene is regulated by the tumor suppressor P53 and has been shown to be involved in P53-mediated apoptosis.[6]
Structure
The BAX gene was the first identified pro-apoptotic member of the Bcl-2 protein family.[7] Bcl-2 family members share one or more of the four characteristic domains of homology entitled the Bcl-2 homology (BH) domains (named BH1, BH2, BH3 and BH4), and can form hetero- or homodimers.[7][8] These domains are composed of nine α-helices, with a hydrophobic α-helix core surrounded by amphipathic helices and a transmembrane C-terminal α-helix anchored to the mitochondrial outer membrane (MOM). A hydrophobic groove formed along the C-terminal of α2 to the N-terminal of α5, and some residues from α8, binds the BH3 domain of other BAX or BCL-2 proteins in its active form. In the protein's inactive form, the groove binds its transmembrane domain, transitioning it from a membrane-bound to a cytosolic protein. A smaller hydrophobic groove formed by the α1 and α6 helices is located on the opposite side of the protein from the major groove, and may serve as a BAX activation site.[9]
Function
In healthy mammalian cells, the majority of BAX is found in the cytosol, but upon initiation of apoptotic signaling, Bax undergoes a conformational shift. Upon induction of apoptosis, BAX becomes organelle membrane-associated, and in particular, mitochondrial membrane associated.[11][12][13][14][15]
BAX is believed to interact with, and induce the opening of the mitochondrial voltage-dependent anion channel,
Clinical significance
The expression of BAX is upregulated by the tumor suppressor protein p53, and BAX has been shown to be involved in p53-mediated apoptosis. The p53 protein is a transcription factor that, when activated as part of the cell's response to stress, regulates many downstream target genes, including BAX. Wild-type p53 has been demonstrated to upregulate the transcription of a chimeric reporter plasmid utilizing the consensus promoter sequence of BAX approximately 50-fold over mutant p53. Thus it is likely that p53 promotes BAX's apoptotic faculties in vivo as a primary transcription factor. However, p53 also has a transcription-independent role in apoptosis. In particular, p53 interacts with BAX, promoting its activation as well as its insertion into the mitochondrial membrane.[20][21][22]
Drugs that activate BAX, such as ABT-737, a BH3 mimetic, hold promise as anticancer treatments by inducing apoptosis in cancer cells.[9] For instance, binding of HA-BAD to BCL-xL and concomitant disruption of BAX:BCL-xL interaction was found to partly reverse paclitaxel resistance in human ovarian cancer cells.[23] Meanwhile, excessive apoptosis in such conditions as ischemia reperfusion injury and amyotrophic lateral sclerosis may benefit from drug inhibitors of BAX.[9]
Interactions
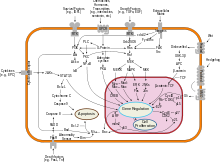
Bcl-2-associated X protein has been shown to interact with:
- Bcl-2,[7][8][24][25][26]
- BCL2A1[8][29]
- SH3GLB1,[14][30]
- SLC25A4,[31]
- VDAC1,[16][19]
- TCTP,[32]
- YWHAQ,[33]
- Bid,[9]
- Bim,[9]
- Puma,[9]
- Noxa,[9]
- Mfn2,[34]
- cholesterol,[35] and
- cardiolipin.[35]
See also
- Apoptosis
- Apoptosome
- Bcl-2
- BH3 interacting domain death agonist(BID)
- Caspases
- Cytochrome c
- Noxa
- Mitochondrion
- p53 upregulated modulator of apoptosis (PUMA)
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000087088 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000003873 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "UniProt". www.uniprot.org. Retrieved 30 April 2023.
- ^ "Entrez Gene: BCL2-associated X protein".
- ^ S2CID 31151334.
- ^ PMID 7644501.
- ^ PMID 24162660.
- ^ "OrthoMaM phylogenetic marker: BAX coding sequence". Archived from the original on 24 September 2015. Retrieved 20 December 2009.
- PMID 9670005.
- PMID 9108035.
- PMID 10228148.
- ^ PMID 11161816.
- PMID 9382873.
- ^ PMID 12767928.
- PMID 16874066.
- PMID 29472455.
- ^ PMID 15637055.
- PMID 8183579.
- PMID 8183578.
- S2CID 14495370.
- ^ S2CID 44240363.
- PMID 15231068.
- S2CID 17808479.
- S2CID 52847351.
- PMID 12137781.
- PMID 12853473.
- PMID 10753914.
- PMID 11259440.
- PMID 9748162.
- PMID 18274553.
- PMID 12426317.
- PMID 21255726.
- ^ PMID 24874738.
External links
- Human BAX genome location and BAX gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: Q07812 (Human Apoptosis regulator BAX) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: Q07813 (Mouse Apoptosis regulator BAX) at the PDBe-KB.

