Cancer stem cell
Cancer stem cells (CSCs) are
Existing cancer treatments have mostly been developed based on
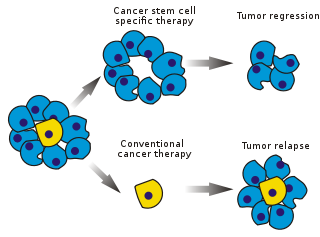
The efficacy of cancer treatments is, in the initial stages of testing, often measured by the ablation fraction of tumor mass (fractional kill). As CSCs form a small proportion of the tumor, this may not necessarily select for drugs that act specifically on the stem cells. The theory suggests that conventional chemotherapies kill differentiated or differentiating cells, which form the bulk of the tumor but do not generate new cells. A population of CSCs, which gave rise to it, could remain untouched and cause relapse.
Cancer stem cells were first identified by
Tumor propagation models
In different

The cancer stem cell model
The cancer stem cell model, also known as the Hierarchical Model proposes that tumors are hierarchically organized (CSCs lying at the apex[6] (Fig. 3).) Within the cancer population of the tumors there are cancer stem cells (CSC) that are tumorigenic cells and are biologically distinct from other subpopulations[7] They have two defining features: their long-term ability to self-renew and their capacity to differentiate into progeny that is non-tumorigenic but still contributes to the growth of the tumor. This model suggests that only certain subpopulations of cancer stem cells have the ability to drive the progression of cancer, meaning that there are specific (intrinsic) characteristics that can be identified and then targeted to destroy a tumor long-term without the need to battle the whole tumor.[8]
Stochastic model
In order for a cell to become cancerous it must undergo a significant number of alterations to its DNA sequence. This cell model suggests these mutations could occur to any cell in the body resulting in a cancer. Essentially this theory proposes that all cells have the ability to be tumorigenic making all tumor cells equipotent with the ability to self-renew or differentiate, leading to tumor heterogeneity while others can differentiate into non-CSCs
These mutations could progressively accumulate and enhance the resistance and fitness of cells that allow them to outcompete other tumor cells, better known as the

[12] These two models are not mutually exclusive, as CSCs themselves undergo clonal evolution. Thus, the secondary more dominant CSCs may emerge, if a mutation confers more aggressive properties[13]
Tying CSC and stochastic models together
A study in 2014 argues the gap between these two controversial models can be bridged by providing an alternative explanation of tumor heterogeneity. They demonstrate a model that includes aspects of both the "Dreamy" and BULL CSC models.[9] They examined cancer stem cell plasticity in which cancer stem cells can transition between non-cancer stem cells (Non-CSC) and CSC via in situ supporting a more Stochastic model.[9][14] But the existence of both biologically distinct non-CSC and CSC populations supports a more CSC model, proposing that both models may play a vital role in tumor heterogeneity.[9]

The cancer stem cell immunology model
This model suggests that immunological properties may be important for understanding tumorigenesis and heterogeneity. As such, CSCs can be very rare in some tumors,[15] but some researchers found that a large proportion of tumor cells can initiate tumors if transplanted into severely immunocompromised mice,[16] and thus questioned the relevance of rare CSCs. However, both stem cells[17] and CSCs[18] possess unique immunological properties which render them highly resistant towards immunosurveillance. Thus, only CSCs may be able to seed tumors in patients with functional immunosurveillance, and immune privilege may be a key criterion for identifying CSCs.[19] Furthermore, the model suggests that CSCs may initially be dependent on stem cell niches, and CSCs may function there as a reservoir in which mutations can accumulate over decades unrestricted by the immune system. Clinically overt tumors may grow if: A) CSCs lose their dependence on niche factors (less differentiated tumors), B) their offspring of highly proliferative, yet initially immunogenic normal tumor cells evolve means to escape immunosurveillance or C) the immune system may lose its tumorsuppressive capacity, e.g. due to ageing.[19]
Debate
The existence of CSCs is under debate, because many studies found no cells with their specific characteristics.
Evidence
The first conclusive evidence for CSCs came in 1997. Bonnet and Dick isolated a subpopulation of leukemia cells that expressed surface marker
In cancer research experiments, tumor cells are sometimes injected into an experimental animal to establish a tumor. Disease progression is then followed in time and novel drugs can be tested for their efficacy. Tumor formation requires thousands or tens of thousands of cells to be introduced. Classically, this was explained by poor methodology (i.e., the tumor cells lose their viability during transfer) or the critical importance of the microenvironment, the particular biochemical surroundings of the injected cells. Supporters of the CSC paradigm argue that only a small fraction of the injected cells, the CSCs, have the potential to generate a tumor. In human acute myeloid leukemia the frequency of these cells is less than 1 in 10,000.[21]
Further evidence comes from
The existence of leukemia stem cells prompted research into other cancers. CSCs have recently been identified in several solid tumors, including:
- Brain[24]
- Breast[25]
- Colon[26]
- Ovary[27][28]
- Pancreas[29]
- Prostate[30][31]
- Melanoma[32][33][34][35]
- Multiple Myeloma[36][37]
- Non-melanoma skin cancer (basal cell carcinoma[38] and squamous cell carcinoma[39][40])
Mechanistic and mathematical models
Once the pathways to cancer are hypothesized, it is possible to develop predictive
Origin

The origin of CSCs is an active research area. The answer may depend on the tumor type and phenotype. So far the hypothesis that tumors originate from a single "cell of origin" has not been demonstrated using the cancer stem cell model. This is because cancer stem cells are not present in end-stage tumors.
Origin hypotheses include mutants in developing stem or progenitor cells, mutants in
Hypotheses
Stem cell mutation
The "mutation in
Adult stem cells
Another theory associates adult stem (ASC) with tumor formation. This is most often associated with tissues with a high rate of cell turnover (such as the
De-differentiation
De-differentiation of mutated cells may create stem cell-like characteristics, suggesting that any cell might become a cancer stem cell. In other words, fully differentiated cell undergoes mutations or extracellular signals that drive it back to a stem-like state. This concept has been demonstrated most recently in prostate cancer models, whereby cells undergoing androgen deprivation therapy appear to transiently alter their transcriptome to that of a neural crest stem-like cell, with the invasive and multipotent properties of this class of stem-like cells.[citation needed]
Hierarchy
The concept of tumor hierarchy claims that a tumor is a heterogeneous population of mutant cells, all of which share some mutations, but vary in specific phenotype. A tumor hosts several types of stem cells, one optimal to the specific environment and other less successful lines. These secondary lines may be more successful in other environments, allowing the tumor to adapt, including adaptation to therapeutic intervention. If correct, this concept impacts cancer stem cell-specific treatment regimes.[47] Such a hierarchy would complicate attempts to pinpoint the origin.
Identification
CSCs, now reported in most human tumors, are commonly identified and enriched using strategies for identifying normal stem cells that are similar across studies.
CSCs can also be identified by efflux of incorporated
Another approach is sphere-forming assays. Many normal
Recent years have seen an advent of genetic approaches to identify cancer stem cells in experimental rodents. In such studies, following the induction of cancer (usually through the application of mutagens), a genetic cassette is activated resulting in the expression of an easily identifiable marker, for instance green fluorescent protein (GFP). This overcomes the limitations of traditional approaches (e.g. the classic Bromodeoxyuridine (BrdU) labeling technique has been used to identify slow-cycling cells in animals) as genetic approaches are cell cycle independent and can be used for in vivo pulse-chase labeling to identify quiescent/slow-cycling cells.[52] This strategy, for instance, was instrumental for identifying the so-called Lgr5+ compartment as a cancer stem cell compartment in liver cancer and showing its potential as a viable therapeutic target.[53]
Heterogeneity (markers)
CSCs heterogeneity is a pool of differentiated and undifferentiated tumour cells that are replenished by cells possessing both tumour and stem cell like properties and having phenotypic and metabolic heterogeneity inside the single tumour mass. There are two theories to explain the phenotypic and metabolic heterogeneity of CSCs; clonal variation and cancer stem cell theory. While former theory dictates the role of genetic, epigenetic and micro environment where tumour cell resides to acquire undifferentiated tumorigenic traits. The latter theory focus more on the malignancy traits acquired by stem cells where these undifferentiated and highly tumorigenic stem cells repopulate the differentiated tumour mass.[54]
CSCs have been identified in various
CD133 (prominin 1) is a five-
EpCAM (epithelial cell adhesion molecule, ESA, TROP1) is hemophilic Ca2+-independent cell adhesion molecule expressed on the basolateral surface of most
CD90 (THY1) is a
CD44 (PGP1) is an adhesion molecule that has pleiotropic roles in cell signaling, migration and homing. It has multiple isoforms, including CD44H, which exhibits high affinity for hyaluronate and CD44V which has metastatic properties.
CD24 (HSA) is a glycosylated glycosylphosphatidylinositol-anchored adhesion molecule, which has co-stimulatory role in B and T cells.
CD200 (OX-2) is a type 1 membrane glycoprotein, which delivers an inhibitory signal to immune cells including T cells, natural killer cells and macrophages.
ALDH is a ubiquitous
The first solid malignancy from which CSCs were isolated and identified was
CSCs have been reported in many brain tumors. Stem-like tumor cells have been identified using cell surface markers including CD133,
CSCs were reported in human
Multiple CSCs have been reported in prostate,[72] lung and many other organs, including liver, pancreas, kidney or ovary.[73] In prostate cancer, the tumor-initiating cells have been identified in CD44+[74] cell subset as CD44+α2β1+,[75] TRA-1-60+CD151+CD166+ [76] or ALDH+ [77] cell populations. Putative markers for lung CSCs have been reported, including CD133+,[78] ALDH+,[79] CD44+ [80] and oncofetal protein 5T4+.[81]
Metastasis
Metastasis is the major cause of tumor lethality. However, not every tumor cell can metastasize.[82] This potential depends on factors that determine growth, angiogenesis, invasion and other basic processes.
Epithelial-mesenchymal transition
In epithelial tumors, the
EMT's important feature is the loss of membrane
Tumor cells undergoing an EMT may be precursors for metastatic cancer cells, or even metastatic CSCs.
Epithelial-mesenchymal transition requires iron, which is taken up in cells via CD44. These resulting mesenchymal cells have properties of cancer stem cells.[92]
Two-phase expression pattern
In breast cancer CD44+CD24−/low cells are detectable in metastatic pleural effusions.[25] By contrast, an increased number of CD24+ cells have been identified in distant metastases in breast cancer patients.[93] It is possible that CD44+CD24−/low cells initially metastasize and in the new site change their phenotype and undergo limited differentiation.[94] The two-phase expression pattern hypothesis proposes two forms of cancer stem cells - stationary (SCS) and mobile (MCS). SCS are embedded in tissue and persist in differentiated areas throughout tumor progression. MCS are located at the tumor-host interface. These cells are apparently derived from SCS through the acquisition of transient EMT (Figure 7).[95]

Implications
CSCs have implications for cancer therapy, including for disease identification, selective drug targets, prevention of metastasis and intervention strategies.
Treatment
CSCs are inherently more resistant to
- 1. Their niche protects them from coming into contact with large concentrations of anti-cancer drugs.
- 2. They express various transmembrane proteins, such as BCRP, that pump drugs out of the cytoplasm.
- 3. They divide slowly, like adult stem cells tend to do, and are thus not killed by chemotherapeutic agents that target rapidly replicating cells via damaging DNA or inhibiting mitosis.
- 4. They upregulate DNA damage repair proteins.
- 5. They are characterized by an overactivation of anti-apoptotic signaling pathways.
After chemotherapy treatment, surviving CSCs are able to repopulate the tumor and cause a relapse. Additional treatment targeted at removing CSCs in addition to cancerous somatic cells must be used to prevent this.
Targeting
Selectively targeting CSCs may allow treatment of aggressive, non-resectable tumors, as well as prevent metastasis and relapse. The hypothesis suggests that upon CSC elimination, cancer could regress due to differentiation and/or cell death.[citation needed] The fraction of tumor cells that are CSCs and therefore need to be eliminated is unclear.[97]
Studies looked for specific markers[25] and for proteomic and genomic tumor signatures that distinguish CSCs from others.[98] In 2009, scientists identified the compound salinomycin, which selectively reduces the proportion of breast CSCs in mice by more than 100-fold relative to Paclitaxel, a commonly used chemotherapeutic agent.[99] It was later shown in 2017 that salinomycin kills cancer stem cells by sequestering iron in lysosomes and killing the cells by exploiting their vulnerability to ferroptosis.[100] Some types of cancer cells can survive treatment with salinomycin through autophagy,[101] whereby cells use acidic organelles such as lysosomes to degrade and recycle certain types of proteins. The use of autophagy inhibitors can kill cancer stem cells that survive by autophagy.[102]
The cell surface receptor interleukin-3 receptor-alpha (CD123) is overexpressed on CD34+CD38- leukemic stem cells (LSCs) in acute myelogenous leukemia (AML) but not on normal CD34+CD38- bone marrow cells.[103] Treating AML-engrafted NOD/SCID mice with a CD123-specific monoclonal antibody impaired LSCs homing to the bone marrow and reduced overall AML cell repopulation including the proportion of LSCs in secondary mouse recipients.[104]
A 2015 study packaged nanoparticles with miR-34a and ammonium bicarbonate and delivered them to prostate CSCs in a mouse model. Then they irradiated the area with near-infrared laser light. This caused the nanoparticles to swell three times or more in size bursting the endosomes and dispersing the RNA in the cell. miR-34a can lower the levels of CD44.[105][106]
In 2017 it was demonstrated that cancer stem cells have more iron and that this is an Achille's heel of these cells. Targeting iron, particularly by sequestering it in lysosomes, can selectively kill this cell population.[107]
A 2018 study identified inhibitors of the ALDH1A family of enzymes and showed that they could selectively deplete putative cancer stem cells in several ovarian cancer cell lines.[108]
A novel small molecule inhibitor, called compound 974 specifically targets cancer stem cells and inhibits cancer stem cell pathways and genes. This inhibitor also reduced the cancer stem cell frequency in mice. This 2022 study also identified a novel relation between ALDH1A1 and senescence. Blocking senescence by compound 974, inhibited cancer stemness in ovarian cancer cells.[55]
It has also been found that CSCs have the ability to exacerbate drug resistance through overexpression of ABC transporter proteins that can pump hydrophobic compounds.[109]
Pathways
The design of new drugs for targeting CSCs requires understanding the cellular mechanisms that regulate cell proliferation. The first advances in this area were made with hematopoietic stem cells (HSCs) and their transformed counterparts in leukemia, the disease for which the origin of CSCs is best understood. Stem cells of many organs share the same cellular pathways as leukemia-derived HSCs.
A normal
BMI-1
The
Notch
The
A branch of the Notch signaling pathway that involves the transcription factor Hes3 regulates a number of cultured cells with CSC characteristics obtained from glioblastoma patients.[116]
Sonic hedgehog and Wnt
These developmental pathways are SC regulators.
Sonic hedgehog blockers are available, such as cyclopamine. A water-soluble cyclopamine may be more effective in cancer treatment. DMAPT, a water-soluble derivative of parthenolide, induces oxidative stress and inhibits NF-κB signaling[121] for AML (leukemia) and possibly myeloma and prostate cancer. Telomerase is a study subject in CSC physiology.[122] GRN163L (Imetelstat) was recently started in trials to target myeloma stem cells.
Wnt signaling can become independent of regular stimuli, through mutations in downstream oncogenes and tumor suppressor genes that become permanently activated even though the normal receptor has not received a signal. β-catenin binds to transcription factors such as the protein TCF4 and in combination the molecules activate the necessary genes. LF3 strongly inhibits this binding in vitro, in cell lines and reduced tumor growth in mouse models. It prevented replication and reduced their ability to migrate, all without affecting healthy cells. No cancer stem cells remained after treatment. The discovery was the product of "rational drug design", involving AlphaScreens and ELISA technologies.[123]
References
- ^ S2CID 4039860.
- ^ Mukherjee, Siddhartha (2010-10-29). "The Cancer Sleeper Cell". New York Times. Retrieved 15 July 2014.
- S2CID 4343326.
- S2CID 2769686.
- PMID 23266557.
- S2CID 205381050.
- ^ S2CID 11882727
- S2CID 2615068.
- ^ PMID 24416258
- PMID 24607403
- S2CID 34417642.
- S2CID 38445059.
- S2CID 4301092.
- PMID 25621103
- ^ S2CID 14167044. [permanent dead link]>
- PMID 19052619.
- PMID 23756938.
- PMID 24333096.
- ^ PMID 25120546.
- PMID 14315085.
- ^ S2CID 205381050.
- S2CID 37236481.
- S2CID 2039638.
- PMID 14522905.
- ^ PMID 12629218.
- ^ S2CID 4419499.
- PMID 18519691.
- PMID 19158483.
- PMID 17283135.
- PMID 18539965.
- PMID 19040209.
- PMID 18202660.
- PMID 20596026.
- PMID 21282657.
- PMID 21393506.
- PMID 14630803.
- PMID 18172311.
- PMID 23292936.
- PMID 22011906.
- ^ S2CID 254124217.
- ISBN 978-1-58488-361-6.
- PMID 16426418.
- PMID 17531079.
- PMID 19477430.
- PMID 26097879.
- PMID 26090957.
- PMID 16990346.
- PMID 21295271.
- ^ S2CID 23736581.
- PMID 21549325.
- S2CID 25401054.
- PMID 28716722.
- PMID 32327656.
- PMID 32670883.
- ^ PMID 35884498.
- S2CID 90912166.
- S2CID 40382183.
- PMID 15381773.
- PMID 18371393.
- S2CID 5547104.
- PMID 20422001.
- PMID 20484027.
- S2CID 4430962.
- PMID 19427293.
- ^ PMID 20858720.
- PMID 21156287.
- ^ PMID 17548814.
- PMID 21652540.
- PMID 18757407.
- PMID 19336570.
- PMID 20068153.
- PMID 21240262.
- PMID 17510412.
- S2CID 28427525.
- PMID 19116269.
- PMID 21245843.
- PMID 20010854.
- S2CID 2484961.
- PMID 21118965.
- PMID 21124918.
- PMID 21540235.
- ^ PMID 32974184.
- S2CID 5236443.
- PMID 10441547.
- PMID 17537911.
- PMID 14623871.
- PMID 21703345.
- PMID 26938687.
- PMID 16341007.
- PMID 18371365.
- PMID 18242515.
- PMID 32747755.
- PMID 17349583.
- PMID 18497886.
- S2CID 20197701.
- PMID 26899500.
- S2CID 205056717.
- PMID 21793190.
- PMID 19682730.
- PMID 28937680.
- S2CID 9298118.
- PMID 25023646.
- S2CID 9467715.
- PMID 19570512.
- ^ "'Nanobombs' that blow up cancer cells | KurzweilAI". www.kurzweilai.net. 2015-12-07. Retrieved 2016-02-20.
- S2CID 205264763.
- PMID 28937680.
- PMID 30221940.
- ^ Shweta, Bisht. "Cancer Stem Cells: From an Insight into the Basics to Recent Advances and Therapeutic Targeting". Hindawi.
- PMID 8414519.
- S2CID 4403711.
- PMID 14574365.
- PMID 14645703.
- PMID 15535842.
- S2CID 2533675.
- PMID 23393614.
- ^ Milosevic, V. et al. Wnt/IL-1β/IL-8 autocrine circuitries control chemoresistancein mesothelioma initiating cells by inducing ABCB5.Int. J. Cancer, https://doi.org/10.1002/ijc.32419
- S2CID 4428056.
- S2CID 31467958.
- PMID 16299030.
- PMID 20712949.
- S2CID 205478338.
- ^ Hodge R (2016-01-25). "Hacking the programs of cancer stem cells". medicalxpress.com. Medical Express. Retrieved 2016-02-12.
Further reading
- Rajasekhar VK, ed. (2014). Cancer Stem Cells. Hoboken, New Jersey: Wiley. ISBN 9781118356166.
- Milosevic V, et al. (January 2020). "Wnt/IL-1β/IL-8 autocrine circuitries control chemoresistance in mesothelioma initiating cells by inducing ABCB5". Int. J. Cancer. 146 (1): 192–207. S2CID 160014053.
