Human betaherpesvirus 5
| Human cytomegalovirus | |
|---|---|
Infectious disease | |
| Causes | Human betaherpesvirus 5 |
| Human betaherpesvirus 5 | |
|---|---|
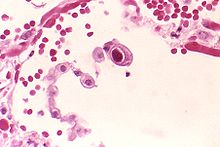
| |
| CMV infection of a human lung pneumocyte
| |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Duplodnaviria |
| Kingdom: | Heunggongvirae
|
| Phylum: | Peploviricota |
| Class: | Herviviricetes |
| Order: | Herpesvirales |
| Family: | Orthoherpesviridae
|
| Genus: | Cytomegalovirus |
| Species: | Human betaherpesvirus 5
|
| Synonyms[1] | |
| |
Human betaherpesvirus 5, also called human cytomegalovirus (HCMV,HHV-5),[2] is a species of virus in the genus Cytomegalovirus, which in turn is a member of the viral family known as Herpesviridae or herpesviruses. It is also commonly called CMV.[3] Within Herpesviridae, HCMV belongs to the Betaherpesvirinae subfamily, which also includes cytomegaloviruses from other mammals.[4] CMV is a double-stranded DNA virus.[5]
Although they may be found throughout the body, HCMV infections are frequently associated with the
HCMV is found in all geographic locations and all socioeconomic groups, and infects between 60% and 70% of adults in the
Signs and symptoms
Human betaherpesvirus 5 infection has a classic triad of symptoms:
Virology
Transmission
The mode of HCMV transmission from person to person is unknown, but is presumed to occur through bodily fluids, including
Replication
HCMV replicates within infected
In disseminated cytomegalovirus infections, as may be seen in the context of an immunosuppressed host, the virus is readily transmitted between polymorphonuclear leukocytes (PM-NLs) and endothelial cells. Infected endothelial cells produce cytokines that attract PM-NLs, which then adhere to the endothelium by interactions between their
Synthesis of the viral double-stranded DNA genome occurs at the host cell nucleus within specialized viral replication compartments.[26]
Nearly 75% of the genes encoded by HCMV strain AD169 can be deleted and still result in the production of infectious virus.
At-risk populations
CMV infections are most significant in the
Pregnancy and congenital infection
HCMV is one of the
Up to 5 of every 1,000 live births are infected. Five percent develop multiple handicaps, and develop
However, infants born preterm and infected with HCMV after birth may experience cognitive and motor impairments later in life.[32]
Immunocompromised adults
CMV infection or reactivation in people whose immune systems are compromised—for example people who have received transplants or are significantly burned—causes illness and increases the risk of death.[33][34]
CMV reactivation is commonly seen in people with severe colitis.[35]
Specific disease entities recognized in those people are
- CMV hepatitis, which may cause fulminant liver failure
- cytomegalovirus retinitis (inflammation of the retina, characterised by a "pizza pie appearance" on ophthalmoscopy)
- large bowel)
- CMV pneumonitis
- CMV esophagitis[36]
- polyradiculopathy, transverse myelitis, and subacute encephalitis
People without CMV infection who are given organ transplants from CMV-infected donors require prophylactic treatment with valganciclovir (ideally) or ganciclovir, and regular serological monitoring to detect a rising CMV titre; if treated early establishment of a potentially life-threatening infection can be prevented.[37]
Immunocompetent adults
CMV infections can still be of clinical significance in adult immunocompetent populations.[38]
- CMV mononucleosis (some sources reserve "mononucleosis" for Epstein–Barr virus only). However, the mononucleosis syndrome associated with CMV typically lacks signs of enlarged cervical lymph nodes and splenomegaly.[39][18]
- CMV has also been associated with Guillain–Barré syndrome,[40] type 1 diabetes,[41] and type 2 diabetes.[42]
The question of whether latent CMV infection has any negative effects on people who are otherwise healthy has been debated; as of 2016 the answer was not clear, but discussions had focused on whether latent CMV might increase the risk of some cardiovascular diseases and cancers.[33]
Pathogenesis

Most healthy people who are infected by HCMV after birth have no symptoms.
Infectious CMV may be shed in the bodily fluids of any infected person, and can be found in urine, saliva, blood, tears, semen, and breast milk. The shedding of virus can occur intermittently, without any detectable signs or symptoms.
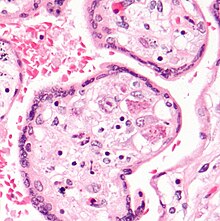
CMV infection can be demonstrated microscopically by the detection of intranuclear
HCMV infection is important to certain high-risk groups.
Lytically replicating viruses disrupt the cytoskeleton, causing massive cell enlargement, which is the source of the virus' name.
A study published in 2009 links infection with CMV to high blood pressure in mice, and suggests that the result of CMV infection of blood vessel endothelium in humans is a major cause of atherosclerosis.[47] Researchers also found that when the cells were infected with CMV, they created renin, a protein known to contribute to high blood pressure.
Human CMV causes cellular senescence, which could contribute to chronic inflammation (inflammaging).[48] Human CMV is also linked to age-associated T cell dysfunction, contributing to immunosenescence.[48] In persons infected with CMV about 10% of memory T cells are CMV-specific, but these may expand to as much as 30% in the elderly,[49] 50% or more of CD8+ memory T-cells.[50] COVID-19 symptom severity is associated with CMV, although the exact mechanism has not been elucidated.[51]
CMV encodes a protein,
A substantial portion of the immune system is involved in continuously controlling CMV, which drains the resources of the immune system.[53][54] Death rates from infectious disease accelerate with age,[55] and CMV infection correlates with reduced effectiveness of vaccination.[56] Persons with the highest levels of CMV antibodies have a much higher risk of death from all causes compared with persons having few or no antibodies.[57][58]
Diagnosis

Most infections with CMV go undiagnosed, as the virus usually produces few, if any, symptoms and tends to reactivate intermittently without symptoms. Persons who have been infected with CMV develop
The CMV pp65 antigenemia test is an immunofluorescence-based assay which utilizes an indirect immunofluorescence technique for identifying the pp65 protein of cytomegalovirus in peripheral blood leukocytes.[59] The CMV pp65 assay is widely used for monitoring CMV infection and its response to antiviral treatment in people who are under immunosuppressive therapy and have had renal transplantation surgery, as the antigenemia results are obtained about 5 days before the onset of symptomatic CMV disease. The advantage of this assay is the rapidity in providing results in a few hours and that the pp65 antigen determination represents a useful criterion for the physician to initiate antiviral therapy. The major disadvantage of the pp65 assay is that only a limited number of samples can be processed per test batch.
CMV should be suspected if a person has symptoms of infectious mononucleosis but has negative test results for mononucleosis and Epstein–Barr virus, or if they show signs of hepatitis, but have negative test results for hepatitis A, B, and C.
For best diagnostic results, laboratory tests for CMV antibody should be performed by using paired serum samples. One blood sample should be taken upon suspicion of CMV, and another one taken within 2 weeks. A virus culture can be performed any time the person is symptomatic. Laboratory testing for antibodies to CMV can be performed to determine if a woman has already had CMV infection. However, routine testing of all pregnant women is costly and the need for testing should therefore be evaluated on a case-by-case basis.
Serologic testing
The enzyme-linked immunosorbent assay (or ELISA) is the most commonly available serologic test for measuring antibody to CMV. The result can be used to determine if acute infection, prior infection, or passively acquired maternal antibody in an infant is present. Other tests include various fluorescence assays, indirect hemagglutination, (PCR) and latex agglutination.[60][61]
An ELISA technique for CMV-specific
Relevance to blood donors
Although the risks discussed above are generally low, CMV assays are part of the standard screening for non-directed blood donation (donations not specified for a particular person) in the U.S., the UK and many other countries. CMV-negative donations are then earmarked for transfusion to infants or people who are immunocompromised. Some blood donation centers maintain lists of donors whose blood is CMV negative due to special demands.[62]
Relevance to bone marrow donors
During allogeneic
Prevention
Vaccination
A
In 2013, Astellas Pharma started on individuals who received a hematopoietic stem cell transplant a phase 3 trial with its CMV deoxyribonucleic acid DNA cytomegalovirus vaccine ASP0113.[66]
In 2015, Astellas Pharma commenced on healthy volunteers a phase 1 trial with its cytomegalovirus vaccine ASP0113.[67]
Further cytomegalovirus vaccines candidates are the CMV-MVA Triplex vaccine and the CMVpp65-A*0201 peptide vaccine. Both vaccine candidates are sponsored by the City of Hope National Medical Center. As of 2016, the development is in clinical phase 2 trial stage.[68][69]
Hygiene
The Centers for Disease Control and Prevention (CDC) recommend regular hand washing,[70] especially after changing diapers.[71] Hand washing is also recommended after feeding a child, wiping a child's nose or mouth, or handling children's toys.[72]
Treatment
- Reduce the risk of CMV-related disease and death in some of the highest-risk transplant recipients
- Provide a measurable long-term survival benefit
- Produce minimal treatment-related side effects and adverse events.[73]
Ganciclovir (Cytovene) treatment is used for people with depressed immunity who have either sight-related or life-threatening illnesses. Valganciclovir (Valcyte) is an antiviral drug that is also effective and is given orally: it is a pro-drug that gets converted into ganciclovir in the body, but is much better absorbed orally than the latter. The therapeutic effectiveness is frequently compromised by the emergence of drug-resistant virus isolates. A variety of amino acid changes in the UL97 protein kinase and the viral DNA polymerase have been reported to cause drug resistance. Foscarnet or cidofovir are only given to people with CMV resistant to ganciclovir, because foscarnet has notable nephrotoxicity, resulting in increased or decreased Ca2+ or PO43−, and decreased Mg2+ levels.[74][75]
A better understanding of how HCMV supports viral latency and reactivation should allow for the development of new therapies that target the latent reservoir.[78]
Drug resistance
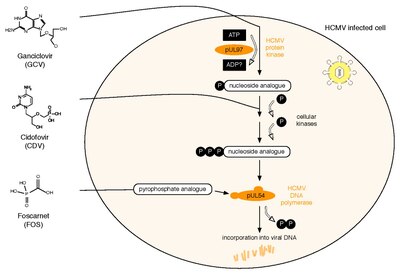
All three currently licensed anti-HCMV drugs target the viral DNA polymerase, pUL54. Ganciclovir (GCV) acts as nucleoside analogue. Its antiviral activity requires phosphorylation by the HCMV protein kinase, pUL97.[79] The second drug, Cidofovir (CDV), is a nucleotide analogue, which is already phosphorylated and thus active. Finally, Foscarnet (FOS) has a different mode of action. It directly inhibits polymerase function by blocking the pyrophosphate binding site of pUL54 (note: investigational drug letermovir acts through a mechanism that involves viral terminase).[80] Two HCMV proteins are implicated in antiviral resistance against these three drugs: pUL97 and pUL54. Specific mutations in pUL97 can cause reduced phosphorylation activity of this viral protein kinase. Thus, fewer monophosphorylated – and thus active – GCV can be synthesized,[81] leading to antiviral resistance against GCV. About 90% of all GCV resistances are caused by such mutations in UL97.[82] Mutations in pUL54 may have different effects leading to antiviral drug resistance: A. They can lead to decreased affinity to antiviral compounds. This resistance mechanism concerns GCV, CDV and FOS and may lead to multidrug resistance.[83] B. Some mutations in pUL54 can increase the polymerase's exonuclease activity. This causes enhanced recognition of incorporated GCV and CDV. As a result, these dNTP analogues are excised more efficiently. Major risk factors for HCMV drug resistance are the residual capacity of the host's immune system to control viral replication and the overall amount and duration of viral replication.[84] HCMV antiviral drug resistance can be detected by phenotypic or by genotypic drug resistance testing. Phenotypic resistance testing involves cultivation of the virus in cell culture and testing its susceptibility using different antiviral drug concentrations in order to determine EC50 values. In contrast, genotypic resistance testing means the detection of resistance associated mutations in UL97 and UL54 by sequencing. Genotypic resistance testing is becoming the method of choice because it is faster, but requires previous phenotypic characterisation of each newly found mutation. This can be performed via a web-based search tool that links a person's HCMV sequence to a database containing all published UL97 and UL54 mutations and corresponding antiviral drug susceptibility phenotypes.[85]
Epidemiology
In the United States, CMV infection rises with age from about 60% of people infected by 6 years of age[34] leveling off at about 85–90% of the population by ages 75–80.[86]
References
- ^ Davison, Andrew (27 January 2016). "Rename species in the family Herpesviridae to incorporate a subfamily designation" (PDF). International Committee on Taxonomy of Viruses (ICTV). Retrieved 13 March 2019.
- ^ taxonomy. "Taxonomy browser (Human betaherpesvirus 5)". www.ncbi.nlm.nih.gov. Retrieved 25 July 2020.
- ^ ISBN 978-0-8385-8529-0.
- ^ ISBN 978-0-521-82714-0.
- PMID 32847820.
- S2CID 41446671.
- PMID 68820.
- PMID 29628341.
- ^
T. Fülöp; A. Larbi & G. Pawelec (September 2013). "Human T cell aging and the impact of persistent viral infections". Frontiers in Immunology. 4: 271. PMID 24062739. article: 271.
- ^
S. Varani & M. P. Landini (2011). "Cytomegalovirus-induced immunopathology and its clinical consequences". Herpesviridae. 2 (6): 6. PMID 21473750.
- ^
Staras SA, Dollard SC, Radford KW, Flanders WD, Pass RF, Cannon MJ (November 2006). "Seroprevalence of cytomegalovirus infection in the United States, 1988–1994". Clin. Infect. Dis. 43 (9): 1143–51. PMID 17029132.
- PMID 28490582.
- PMID 11843023.
- ^
Caruso C, et al. (2009). "Mechanisms of immunosenescence". Immun Ageing. 6: 10. PMID 19624841.
- ^ "Human Herpesvirus 5 - an overview | ScienceDirect Topics".
- S2CID 80706263.
- PMID 21413307.
- ^ a b Larsen, Laura. Sexually Transmitted Diseases Sourcebook. Health Reference Series Detroit: Omnigraphics, Inc., 2009. Online.
- PMID 12588074.
- PMID 10906217.
- PMID 10432281.
- ^ PMID 31336680. 70.
- ^ PMID 6281461.
- PMID 23180859.
- PMID 9889260.
- PMID 9426445.
- PMID 14623981.
- ^ "Human Cytomegalovirus (HCMV) | British Society for Immunology".
- ^ "Congenital CMV Infection | CDC". 3 December 2021.
- PMID 21364849.
- PMID 30081449.
- PMID 25466679.
- ^ PMID 17922387.
- S2CID 5969716.
- ISBN 978-3-13-125852-6. Retrieved 26 June 2010.
- S2CID 249441439.
- PMID 18371229.
- PMID 4316146.
- PMID 21427391.
- S2CID 42852009.
- S2CID 9783330.
- ^
Bottieau E, Clerinx J, Van den Enden E, et al. (2006). "Infectious mononucleosis-like syndromes in febrile travelers returning from the tropics". Journal of Travel Medicine. 13 (4): 191–7. PMID 16884400.
- ^ "Human Cytomegalovirus - an overview | ScienceDirect Topics".
- ^
Mattes FM, McLaughlin JE, Emery VC, Clark DA, Griffiths PD (August 2000). "Histopathological detection of owl's eye inclusions is still specific for cytomegalovirus in the era of human herpesviruses 6 and 7". J. Clin. Pathol. 53 (8): 612–4. PMID 11002765.
- ^
Bennekov T, Spector D, Langhoff E (March 2004). "Induction of immunity against human cytomegalovirus". Mt. Sinai J. Med. 71 (2): 86–93. PMID 15029400.
- ^
Cheng J, Ke Q, Jin Z, et al. (May 2009). Früh K (ed.). "Cytomegalovirus infection causes an increase of arterial blood pressure". PLOS Pathog. 5 (5): e1000427. PMID 19436702.
- ^ S2CID 51924586.
- PMID 27501258.
- PMID 18469829.
- PMID 34746804.
- S2CID 20718868.
- PMID 16456027.
- PMID 21813708.
- S2CID 45806175.
- S2CID 10483363. Archived from the original(PDF) on 19 April 2018.
- S2CID 14983626.
- PMID 21379581.
- PMID 21827433.
- ^ "CMV Infection Laboratory Testing | CDC". 3 December 2021.
- PMID 12364375.
- ^ "United Blood Services FAQs". Archived from the original on 19 May 2007. Retrieved 23 May 2007.
- PMID 24850801. Retrieved 3 September 2020.
- PMID 21236396.
- ^
Pass RF, Zhang C, Evans A, et al. (March 2009). "Vaccine prevention of maternal cytomegalovirus infection". N. Engl. J. Med. 360 (12): 1191–9. PMID 19297572.
- ^ "A Study to Evaluate a Therapeutic Vaccine, ASP0113, in Cytomegalovirus (CMV)-Seropositive Recipients Undergoing Allogeneic, Hematopoietic Cell Transplant (HCT) (HELIOS)". ClinicalTrials.gov. 12 June 2013. Retrieved 26 October 2015.
- ^ "An Evaluation of a Cytomegalovirus (CMV) Vaccine (ASP0113) in CMV-Seropositive and CMV-Seronegative Healthy Subjects and CMV-Seronegative Dialysis Patients". ClinicalTrials.gov. 8 July 2015. Retrieved 22 October 2015.
- ^ "Multi-antigen CMV-MVA Triplex Vaccine in Reducing CMV Complications in Patients Previously Infected With CMV and Undergoing Donor Hematopoietic Cell Transplant". ClinicalTrials.gov. 21 July 2015. Retrieved 23 January 2016.
- ^ "Vaccine Therapy in Reducing the Frequency of Cytomegalovirus Events in Patients With Hematologic Malignancies Undergoing Donor Stem Cell Transplant". ClinicalTrials.gov. 12 March 2015. Retrieved 23 January 2016.
- ^ "When & How to Wash Your Hands | Handwashing". CDC. 7 March 2016. Archived from the original on 13 September 2017. Retrieved 16 October 2017.
- ^ "CMV | Overview | Cytomegalovirus and Congenital CMV Infection". CDC. 17 June 2016. Archived from the original on 16 October 2017. Retrieved 16 October 2017.
- ^ "CMV Transmission". National CMV Foundation. Archived from the original on 16 October 2017. Retrieved 16 October 2017.
- ^ Cytogam Prescribing Info Archived 26 April 2012 at the Wayback Machine CSL Behring AG
- PMID 27495775.
- PMID 10194460.
- ^ "Previmys EPAR summary for the public" (PDF). European Medicines Agency. Archived (PDF) from the original on 27 June 2018. Retrieved 27 June 2018.
- ^ "Approval Package for Letermovir" (PDF). FDA. Archived (PDF) from the original on 27 June 2018. Retrieved 27 June 2018.
- PMID 34663377.
- S2CID 4309307.
- PMID 1706982.
- PMID 3022304.
- S2CID 23668301.
- PMID 15728878.
- S2CID 46533224.
- PMID 19853628.
- S2CID 623760. Archived from the original(PDF) on 19 April 2018.
External links
- Human betaherpesvirus 5 at Curlie
- Cytomegalovirus (CMV) US Centers for Disease Control and Prevention (CDC). 22 May 2009
- HCMV drug resistance mutations tool
- "Human herpesvirus 5". NCBI Taxonomy Browser. 10359.
