Isocyanic acid
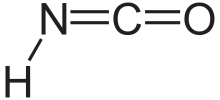
| |

| |
| Names | |
|---|---|
| IUPAC name
Isocyanic acid
| |
| Other names
Carbimide[1]
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.109.068 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| HNCO | |
| Molar mass | 43.025 g·mol−1 |
| Appearance | Colorless liquid or gas (boiling point near room temperature) |
| Density | 1.14 g/cm3 (20 °C) |
| Melting point | −86 °C (−123 °F; 187 K)[3] |
| Boiling point | 23.5 °C (74.3 °F; 296.6 K) |
| Dissolves | |
| Solubility | Soluble in benzene, toluene, diethyl ether |
Conjugate acid
|
Oxomethaniminium[2] |
Conjugate base
|
Cyanate |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Poisonous |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isocyanic acid is a
of cyanic acid (aka. cyanol) (H−O−C≡N).The derived
Isocyanic acid was discovered in 1830 by Justus von Liebig and Friedrich Wöhler.[5]
Isocyanic acid is the simplest stable
Structure
Isocyanic acid (HNCO)
Although the electronic structure according to
However, classic vibrational analysis would indicate that the 2268.8 cm−1 is the asymmetric N=C=O stretch, as per Colthup et al.,[10] as well as the NIST Chemistry WebBook,[11] which also reports the corresponding symmetric N=C=O stretch (weak in infrared, but strong in Raman) to be 1327 cm−1. Based on these classic assignments, there is no need to invoke a full charged state for the N and O atoms, to explain the vibrational spectral data.
Cyanic acid (HOCN)
The tautomer, known as cyanic acid, HOCN, in which the oxygen atom is protonated exists in equilibrium with isocyanic acid to the extent of about 3%.[citation needed][dubious ] The vibrational spectrum is indicative of the presence of a triple bond between the nitrogen and carbon atoms.[12]
Properties
In aqueous solution it is a
- HNCO ⇌ H+ + NCO−
Isocyanic acid hydrolyses to carbon dioxide and ammonia:
- HNCO + H2O → CO2 + NH3
Dilute solutions of isocyanic acid are stable in inert
At high concentrations, isocyanic acid oligomerizes to give the trimer cyanuric acid and cyamelide, a polymer. These species usually are easily separated from liquid- or gas-phase reaction products.
Isocyanic acid reacts with amines to give ureas (carbamides):
- HNCO + RNH2 → RNHC(O)NH2
This reaction is called carbamylation.
HNCO adds across electron-rich double bonds, such as vinylethers, to give the corresponding isocyanates.
Isocyanic acid, HNCO, is a Lewis acid whose free energy, enthalpy and entropy changes for its 1:1 association with a number of bases in carbon tetrachloride solution at 25 °C have been reported.[15] The acceptor properties of HNCO are compared with other Lewis acid in the ECW model.
Low-temperature photolysis of solids containing HNCO creates the tautomer cyanic acid H−O−C≡N, also called hydrogen cyanate.[16] Pure cyanic acid has not been isolated, and isocyanic acid is the predominant form in all solvents.[14] Sometimes information presented for cyanic acid in reference books is actually for isocyanic acid.[citation needed]
Preparation
Isocyanic acid can be made by protonation of the
HNCO also can be made by the high-temperature thermal decomposition of the trimer cyanuric acid:
In the reverse of the famous synthesis of urea by Friedrich Wöhler,
isocyanic acid is produced and rapidly trimerizes to cyanuric acid.
Occurrence
Isocyanic acid has been detected in many kinds of interstellar environments.[7]
Isocyanic acid is also present in various forms of
See also
References
- ^ Cyanamide also has this name, and for which it is more systematically correct
- ^ "Oxomethaniminium | CH2NO | ChemSpider". www.chemspider.com. Retrieved 27 January 2019.
- ISBN 0-07-049439-8
- ^
- .
- .
- ^
- ^ Nakamoto, part A, p 190
- .
- ISBN 978-0-12-182554-6.
- ^ "Isocyanic acid". National Institute of Standards and Technology (U.S. Department of Commerce). Retrieved 2023-04-20.
- .
- ^ IUPAC SC-Database Archived 2017-06-19 at the Wayback Machine A comprehensive database of published data on equilibrium constants of metal complexes and ligands
- ^ Article Online Posting Date: April 15, 2001.
- ^ Nelson, J. (1970) Hydrogen-bonded complexes of isocyanic acid: Infrared spectra and thermodynamic measurements. Spectrochimica Acta Part A: Molecular Spectroscopy 26,109-120.
- .
- S2CID 37461221.
- ^ Preidt, Robert. "Chemical in Smoke May Pose Health Risk". MyOptumHealth. AccuWeather. Retrieved 14 September 2011.
External links
- Walter, Wolfgang (1997). Organic Chemistry: A Comprehensive Degree Text and Source Book. Chichester: Albion Publishing. p. 364. ISBN 978-1-898563-37-2. Retrieved 2008-06-21.
- Cyanic acid from NISTChemistry WebBook (accessed 2006-09-09)



