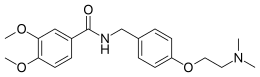
Itopride
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ganaton |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
FMO3), primarily N-oxidation[2] | |
| Elimination half-life | 5.7±0.3 hours |
| Excretion | Renal (3.7–4.1% as unchanged itopride, 75.4–89.4% as itopride-N-oxide)[1] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Itopride (
Medical uses
Typically, itopride is indicated in the treatment of GI symptoms caused by reduced GI motility:
- gastroparesis (delayed gastric emptying)[9][10]
- anorexia
- heartburn
- regurgitation
- bloating
- nausea and vomiting
- other possible gastric, prolactin, or dopamine related conditions
Itopride was shown to significantly improve symptoms in patients with functional dyspepsia and motility disorders in placebo-controlled trials.
These studies concluded that the reduction in the severity of symptoms of functional dyspepsia after 8 weeks of treatment with itopride indicated that itopride was significantly superior to placebo and that itopride yielded a greater rate of response than placebo in significantly reducing pain and fullness.[11]
Contraindications and precautions
Itopride is a relatively new drug and it is not currently approved for normal prescribed use nor OTC use in either the US nor the UK. However, this does not necessarily indicate that itopride is not effective or safe.
Patients taking itopride should report any side-effects to their treating physician.
Itopride is contraindicated in hypersensitivity to itopride or
Adverse drug reactions
The most common side-effects of itopride include mild to moderate abdominal pain and diarrhoea.[7] Some other side effects that may occur include: rash, giddiness, exhaustion, back or chest pain, increased salivation, constipation, headache, sleeping disorders, dizziness, galactorrhea, and gynecomastia.
Leukopenia, a reduction in the normal level of white blood cells, can be a potentially life-threatening reaction to itopride.
Central nervous system adverse effects do not tend to occur due to poor penetration across the blood brain barrier, although a slight raising of prolactin levels may occur.[7] Raising of prolactin levels is more common with high dose regimes of itopride.[12]
Cardiac studies
Itopride belongs to the same
Later, in a study conducted with healthy adult volunteers, itopride was shown as unlikely to cause cardiac arrhythmias or ECG changes in part to the lack of significant interaction and metabolism via the
The conclusion of this study revealed that itopride is devoid of any abnormal effect on QT interval. Therefore, it may be possible that itopride could be considered as a better and certainly safer prokinetic agent than either cisapride or mosapride, and itopride should also be considered a welcome treatment addition for symptomatic nonulcer dyspepsia and other gastric motility disorders.[13]
Pharmacology
Itopride acts as a selective dual
There is evidence that itopride may have prokinetic effects throughout the gastrointestinal tract from the stomach to the end of the colon.
Similarly to other D2 receptor antagonists, itopride has been found to dose-dependently increase prolactin levels.[6]
Pharmacokinetics
After oral administration itopride undergoes rapid and extensive absorption with levels of itopride peaking in the blood plasma after only 35 minutes. Itopride is primarily eliminated via the kidneys having an
Mechanism of action

Itopride increases acetylcholine concentrations by inhibiting dopamine D2 receptors and acetylcholinesterase. Higher acetylcholine increases GI peristalsis, increases the lower esophageal sphincter pressure, stimulates gastric motility, accelerates gastric emptying, and improves gastro-duodenal coordination.[7]
Itopride given as a single dose study found that it also raises levels of motilin, somatostatin and lowers levels of cholecystokinin, as well as adrenocorticotropic hormone. These effects may also contribute to itopride's pharmacology.[18]
Interactions
Anticholinergic agents reduce the action of itopride.[medical citation needed]
Society and culture
Names
Itopride is available under the brand names Ganaton (
References
- ^ "Ganaton (itopride hydrochloride) Tablets 50 mg. Prescribing Information" (PDF). Abbott Japan Co., Ltd. Archived from the original (PDF) on 11 December 2015. Retrieved 9 December 2015.
- ISBN 978-3-527-32954-0.
- PMID 8835639.
- PMID 16495395.
- ^ ISBN 978-1-60761-552-1.
- ^ ISBN 978-1-4557-0450-7.
- ^ PMID 23326147.
- PMID 18019739.
- PMID 15305632.
- ^ Kojecky V, Bernatek J, Bakala J, Weissova D (2005). "[The influence of itopride on the rate and course of the evacuation of stomach of the diabetic patients and their relationship to diabetes control]". Ces.Slov.Gastroent.Hepatol., 2005. 59 (1): 17–20.
- PMID 16495395.
- PMID 16015691.
- ^ Gupta S, Kapoor V, Gupta BM, Verma U (2005). "Effect Of Itopride hydrochloride on QT interval in adult healthy volunteers" (PDF). JK-Practitioner. 12 (4): 207–10.
- PMID 18581598.
- S2CID 25760696.
- PMID 11127807.
- PMID 23960836.
- S2CID 22219251.
- ^ "Z".
External links
- "Itopride". Drug Information Portal. U.S. National Library of Medicine.
- Abbott Labs "Ganaton"
- Clinical trial number NCT00272103 for "Itopride in Functional Dyspepsia:a Dose Finding Study" at ClinicalTrials.gov
