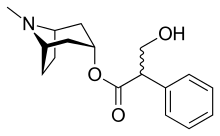
Atropine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Atropen, others |
| Other names | Daturin[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682487 |
| License data | |
| Pregnancy category |
|
antimuscarinic (anticholinergic) | |
| ATC code | |
| Legal status | |
| Legal status | |
hydrolysed to tropine and tropic acid | |
| Onset of action | c. 1 minute[5] |
| Elimination half-life | 2 hours |
| Duration of action | 30 to 60 min[5] |
| Excretion | 15–50% excreted unchanged in urine |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Atropine is a
Common
Atropine occurs naturally in a number of plants of the
Medical uses

Eyes
In refractive and accommodative amblyopia, when occlusion is not appropriate sometimes atropine is given to induce blur in the good eye.[16] Evidence suggests that atropine penalization is just as effective as occlusion in improving visual acuity.[17][18]
Antimuscarinic topical medication is effective in slowing myopia progression in children; accommodation difficulties and papillae and follicles are possible side-effects.[19] All doses of atropine appear similarly effective, while higher doses have greater side effects.[20] The lower dose of 0.01% is thus generally recommended due to fewer side effects and potential less rebound worsening when the atropine is stopped.[20][21]
Heart
Injections of atropine are used in the treatment of symptomatic or unstable bradycardia.
Atropine was previously included in international resuscitation guidelines for use in cardiac arrest associated with asystole and PEA, but was removed from these guidelines in 2010 due to a lack of evidence for its effectiveness.[22] For symptomatic bradycardia, the usual dosage is 0.5 to 1 mg IV push; this may be repeated every 3 to 5 minutes, up to a total dose of 3 mg (maximum 0.04 mg/kg).[23]
Atropine is also useful in treating
Atropine has also been used in an effort to prevent a low heart rate during intubation of children; however, evidence does not support this use.[24]
Secretions
Atropine's actions on the parasympathetic nervous system inhibit salivary and mucus glands. The drug may also inhibit sweating via the sympathetic nervous system. This can be useful in treating hyperhidrosis, and can prevent the death rattle of dying patients. Even though atropine has not been officially indicated for either of these purposes by the FDA, it has been used by physicians for these purposes.[25]
Poisonings
Atropine is not an actual
Some of the nerve agents attack and destroy acetylcholinesterase by phosphorylation, so the action of acetylcholine becomes excessive and prolonged. Pralidoxime (2-PAM) can be effective against organophosphate poisoning because it can re-cleave this phosphorylation. Atropine can be used to reduce the effect of the poisoning by blocking muscarinic acetylcholine receptors, which would otherwise be overstimulated, by excessive acetylcholine accumulation.
Atropine or diphenhydramine can be used to treat muscarine intoxication.[medical citation needed]
Atropine was added to cafeteria salt shakers in an attempt to poison the staff of
Irinotecan induced diarrhea
Atropine has been observed to prevent or treat irinotecan induced acute diarrhea.[28]
Side effects
Adverse reactions to atropine include ventricular
In overdoses, atropine is poisonous.[medical citation needed] Atropine is sometimes added to potentially addictive drugs, particularly antidiarrhea opioid drugs such as diphenoxylate or difenoxin, wherein the secretion-reducing effects of the atropine can also aid the antidiarrhea effects.[medical citation needed]
Although atropine treats bradycardia (slow heart rate) in emergency settings, it can cause paradoxical heart rate slowing when given at very low doses (i.e. <0.5 mg),[29] presumably as a result of central action in the CNS.[30] One proposed mechanism for atropine's paradoxical bradycardia effect at low doses involves blockade of inhibitory presynaptic muscarinic autoreceptors, thereby blocking a system that inhibits the parasympathetic response.[31]
Atropine is incapacitating at doses of 10 to 20 mg per person. Its LD50 is estimated to be 453 mg per person (by mouth) with a probit slope of 1.8.[32] The antidote to atropine is physostigmine or pilocarpine.[medical citation needed]
A common
Contraindications
It is generally
Chemistry
This article needs more primary sources. (January 2022) |  |
Atropine, a
The most common atropine compound used in medicine is atropine sulfate (monohydrate) (C
17H
23NO
3)2·H2SO4·H2O, the full chemical name is 1α H, 5α H-Tropan-3-α ol (±)-tropate(ester), sulfate monohydrate.
Pharmacology
This article needs more primary sources. (January 2022) |  |
In general, atropine counters the "rest and digest" activity of glands regulated by the parasympathetic nervous system. This occurs because atropine is a competitive, reversible antagonist of the muscarinic acetylcholine receptors (acetylcholine being the main neurotransmitter used by the parasympathetic nervous system).
Atropine is a
In cardiac uses, it works as a nonselective muscarinic acetylcholinergic antagonist, increasing firing of the
In the eye, atropine induces
The vagus (parasympathetic) nerves that innervate the heart release acetylcholine (ACh) as their primary neurotransmitter. ACh binds to muscarinic receptors (M2) that are found principally on cells comprising the sinoatrial (SA) and atrioventricular (AV) nodes. Muscarinic receptors are coupled to the Gi subunit; therefore, vagal activation decreases cAMP. Gi-protein activation also leads to the activation of KACh channels that increase potassium efflux and hyperpolarizes the cells.
Increases in vagal activities to the SA node decreases the firing rate of the pacemaker cells by decreasing the slope of the pacemaker potential (phase 4 of the action potential); this decreases heart rate (negative chronotropy). The change in phase 4 slope results from alterations in potassium and calcium currents, as well as the slow-inward sodium current that is thought to be responsible for the pacemaker current (If). By hyperpolarizing the cells, vagal activation increases the cell's threshold for firing, which contributes to the reduction in the firing rate. Similar electrophysiological effects also occur at the AV node; however, in this tissue, these changes are manifested as a reduction in impulse conduction velocity through the AV node (negative dromotropy). In the resting state, there is a large degree of vagal tone on the heart, which is responsible for low resting heart rates.
There is also some vagal innervation of the atrial muscle, and to a much lesser extent, the ventricular muscle. Vagus activation, therefore, results in modest reductions in atrial contractility (inotropy) and even smaller decreases in ventricular contractility.
Muscarinic receptor antagonists bind to muscarinic receptors thereby preventing ACh from binding to and activating the receptor. By blocking the actions of ACh, muscarinic receptor antagonists very effectively block the effects of vagal nerve activity on the heart. By doing so, they increase heart rate and conduction velocity.
History

The name atropine was coined in the 19th century, when pure extracts from the belladonna plant
Atropine-rich extracts from the Egyptian
The pharmacological study of belladonna extracts was begun by the German chemist Friedlieb Ferdinand Runge (1795–1867). In 1831, the German pharmacist Heinrich F. G. Mein (1799-1864)[38] succeeded in preparing a pure crystalline form of the active substance, which was named atropine.[39] [40] The substance was first synthesized by German chemist Richard Willstätter in 1901.[41]
Natural sources
Atropine is found in many members of the family
Synthesis
Atropine can be synthesized by the reaction of tropine with tropic acid in the presence of hydrochloric acid.
Biosynthesis
The biosynthesis of atropine starting from
Name
The species name "belladonna" ('beautiful woman' in Italian) comes from the original use of deadly nightshade to dilate the pupils of the eyes for cosmetic effect. Both atropine and the genus name for deadly nightshade derive from Atropos, one of the three Fates who, according to Greek mythology, chose how a person was to die.[33]
See also
References
- ^ Rafinesque CS (1828). Medical Flora; Or, Manual of the Medical Botany of the United States of ... - Constantine Samuel Rafinesque - Internet Archive. Atkinson & Alexander. p. 148. Retrieved 2012-11-07.
- ^ "AusPAR: Atropine sulfate monohydrate". Therapeutic Goods Administration (TGA). 31 May 2022. Archived from the original on 31 May 2022. Retrieved 12 June 2022.
- ^ a b "Atropine sulfate". dailymed.nlm.nih.gov. U.S. National Library of Medicine. Archived from the original on 26 July 2020. Retrieved 30 October 2019.
- ^ "Atropine- atropine sulfate solution/ drops". DailyMed. 22 February 2022. Archived from the original on 16 March 2022. Retrieved 16 March 2022.
- ^ ISBN 9780781787635. Archivedfrom the original on 2015-11-24.
- ^ a b c d e f g "Atropine". The American Society of Health-System Pharmacists. Archived from the original on 2015-07-12. Retrieved Aug 13, 2015.
- ISBN 9781284056716. Archivedfrom the original on 2015-10-02.
- ^ "Amblyopia (Lazy Eye)". National Eye Institute. 2019-07-02. Archived from the original on 2020-01-31. Retrieved 2020-01-31.
Putting special eye drops in the stronger eye. A once-a-day drop of the drug atropine can temporarily blur near vision, which forces the brain to use the other eye. For some children, this treatment works as well as an eye patch, and some parents find it easier to use (for example, because young children may try to pull off eye patches).
- ^ a b "Atropine Pregnancy and Breastfeeding Warnings". Archived from the original on 6 September 2015. Retrieved 14 August 2015.
- ISBN 9780750673136. Archivedfrom the original on 2015-10-02.
- ISBN 9781118819593. Archivedfrom the original on 2015-10-02.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ISBN 9781284056716. Archivedfrom the original on 2015-10-02.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ PMID 29132914.
- ^ Georgievski Z, Koklanis K, Leone J (2008). "Fixation behavior in the treatment of amblyopia using atropine". Clinical and Experimental Ophthalmology. 36 (Suppl 2): A764–A765.
- S2CID 243130859.
- PMID 31461545.
- PMID 31930781.
- ^ PMID 28494063.
- PMID 31409953.
- S2CID 1031566.
- ISBN 0-13-113607-0.
- PMID 26473000.
- ^ "Death Rattle and Oral Secretions, 2nd ed". eperc.mcw.edu. Archived from the original on 2014-04-14. Retrieved 2019-10-20.
- ^ The Battle Over Hearts and Minds A Cold War of Spies: Episode 4 |https://www.imdb.com/title/tt27484449/?ref_=ttep_ep4
- ^ "A Cold War of Spies | PBS America | UK".
- S2CID 11784173.
- ^ "Atropine Drug Information". uptodate.com. Archived from the original on 2014-02-20. Retrieved 2014-02-02.
- ISBN 978-0-443-06911-6.
- ISBN 978-0-07-162442-8.
- ISBN 978-0-9677264-3-4.
- ^ S2CID 28327277. Retrieved 2007-05-21. citing J. Arena, Poisoning: Toxicology-Symptoms-Treatments, 3rd edition. Springfield, Charles C. Thomas, 1974, p 345
- ^ Szajewski J (1995). "Acute anticholinergic syndrome". IPCS Intox Databank. Archived from the original on 2 July 2007. Retrieved 2007-05-22.
- S2CID 27306334.
- ISBN 978-0-443-07145-4.
- ^ Goodman and Gilman's Pharmacological Basis of Therapeutics, q.v. "Muscarinic receptor antagonists - History", p. 163 of the 2001 edition.
- ^ "Heinrich Friedrich Georg Mein". ostfriesischelandschaft.de (in German). Archived from the original on 2013-05-11. Retrieved 2019-10-20.
{{cite web}}: CS1 maint: unfit URL (link) - ^ Heinrich Friedrich Georg Mein (1833). "Ueber die Darstellung des Atropins in weissen Kristallen" [On the preparation of atropine as white crystals]. Annalen der Pharmacie (in German). Vol. 6 (1 ed.). pp. 67–72. Archived from the original on 2016-05-15. Retrieved 2016-01-05.
- ^ Atropine was also independently isolated in 1833 by Geiger and Hesse:
- Geiger, Hesse (1833). "Darstellung des Atropins" [Preparation of atropine]. Annalen der Pharmacie (in German). Vol. 5. pp. 43–81. Archived from the original on 2016-05-14. Retrieved 2016-01-05.
- Geiger, Hesse (1833). "Fortgesetzte Versuche über Atropin" [Continued experiments on atropine]. Annalen der Pharmacie (in German). Vol. 6. pp. 44–65. Archived from the original on 2016-06-10. Retrieved 2016-01-05.
- ^ See:
- Willstätter R (1901). "Synthese des Tropidins" [Synthesis of tropidine]. Berichte der Deutschen Chemischen Gesellschaft zu Berlin (in German). 34: 129–144. from the original on 2013-03-01.
- Willstätter R (1901). "Umwandlung von Tropidin in Tropin" [Conversion of tropidine into tropine]. Berichte der Deutschen Chemischen Gesellschaft zu Berlin (in German). 34 (2): 3163–3165. from the original on 2013-01-26.
- ^ ISBN 978-0-470-74167-2.
External links
 Media related to Atropine at Wikimedia Commons
Media related to Atropine at Wikimedia Commons- "Atropine". Drug Information Portal. U.S. National Library of Medicine.
- "Atropine sulfate". Drug Information Portal. U.S. National Library of Medicine.
