Polyimide

Polyimide (sometimes abbreviated PI) is a
History
The first polyimide was discovered in 1908 by Bogart and Renshaw.[2] They found that 4-amino phthalic anhydride does not melt when heated but does release water upon the formation of a high molecular weight polyimide. The first semialiphatic polyimide was prepared by Edward and Robinson by melt fusion of diamines and tetra acids or diamines and diacids/diester.[3]
However, the first polyimide of significant commercial importance - Kapton - was pioneered in the 1950s by workers at Dupont who developed a successful route for synthesis of high molecular weight polyimide involving a soluble polymer precursor. Up to today this route continues being the primary route for the production of most polyimides. Polyimides have been in mass production since 1955. The field of polyimides is covered by various extensive books[4][5][6] and review articles.[7][8]
Classification
According to the composition of their main chain, polyimides can be:
- Aliphatic,
- Semi-aromatic (also referred to as alipharomatic),
- Aromatic: these are the most used polyimides because of their thermostability.
According to the type of interactions between the main chains, polyimides can be:
- Thermoplastic: very often called pseudothermoplastic.
- Thermosetting: commercially available as uncured resins, polyimide solutions, stock shapes, thin sheets, laminates and machined parts.
Synthesis
Several methods are possible to prepare polyimides, among them:
- The reaction between a dianhydride and a diamine (the most used method).
- The reaction between a dianhydride and a diisocyanate.
The polymerization of a diamine and a dianhydride can be carried out by a two-step method in which a poly(amidocarboxylic acid) is prepared first, or directly by a one-step method. The two-step method is the most widely used procedure for polyimide synthesis. First a soluble poly(amidocarboxylic acid) (2) is prepared which is cyclized after further processing in a second step to the polyimide (3). A two-step process is necessary because the final polyimides are in most cases infusible and insoluble due to their aromatic structure.
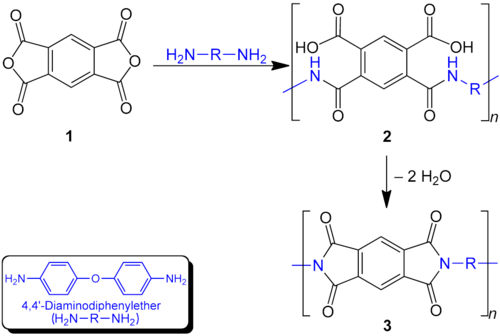
Dianhydrides used as precursors to these materials include pyromellitic dianhydride,
Analysis
The imidization reaction can be followed via IR spectroscopy. The IR spectrum is characterized during the reaction by the disappearance of absorption bands of the poly(amic acid) at 3400 to 2700 cm−1 (OH stretch), ~1720 and 1660 (amide C=O) and ~1535 cm−1 (C-N stretch). At the same time, the appearance of the characteristic imide bands can be observed, at ~1780 (C=O asymm), ~1720 (C=O symm), ~1360 (C-N stretch) and ~1160 and 745 cm−1 (imide ring deformation).[10] Detailed analyses of polyimide[11] and carbonized polyimide[12] and graphitized polyimide[13] have been reported.
Properties
Thermosetting polyimides are known for thermal stability, good chemical resistance, excellent mechanical properties, and characteristic orange/yellow color. Polyimides compounded with
Typical polyimide parts are not affected by commonly used solvents and oils – including hydrocarbons, esters, ethers, alcohols and freons. They also resist weak acids but are not recommended for use in environments that contain alkalis or inorganic acids. Some polyimides, such as CP1 and CORIN XLS, are solvent-soluble and exhibit high optical clarity. The solubility properties lend them towards spray and low temperature cure applications.
Applications


Insulation and passivation films
Polyimide materials are lightweight, flexible, resistant to heat and chemicals. Therefore, they are used in the electronics industry for flexible cables and as an insulating film on magnet wire. For example, in a laptop computer, the cable that connects the main logic board to the display (which must flex every time the laptop is opened or closed) is often a polyimide base with copper conductors. Examples of polyimide films include Apical, Kapton, UPILEX, VTEC PI, Norton TH and Kaptrex.

Polyimide is used to coat optical fibers for medical or high temperature applications.[15]
An additional use of polyimide resin is as an insulating and
Multi-layer insulation used on
Mechanical parts
Polyimide powder can be used to produce parts and shapes by sintering technologies (hot compression molding, direct forming, and isostatic pressing). Because of their high mechanical stability even at elevated temperatures they are used as bushings, bearings, sockets or constructive parts in demanding applications. To improve
Filters
In coal-fired power plants, waste incinerators, or cement plants, polyimide fibres are used to filter hot gases. In this application, a polyimide needle felt separates dust and particulate matter from the exhaust gas.
Polyimide is also the most common material used for the reverse osmotic film in purification of water, or the concentration of dilute materials from water, such as maple syrup production.[21][22]
Flexible circuits
Polyimide is used as the core of flexible circuit boards and flat-flex cables. Flexible circuit boards are thin and can be placed in odd-shaped electronics.[23]
Other
Polyimide is used for medical tubing, e.g. vascular
The semiconductor industry uses polyimide as a high-temperature adhesive; it is also used as a mechanical stress buffer.
Some polyimide can be used like a photoresist; both "positive" and "negative" types of photoresist-like polyimide exist in the market.
The
See also
- Polyamide – Macromolecule with repeating units linked by amide bonds
- Polyamide-imide – class of polymers
- Polymerization – Chemical reaction to form polymer chains
References
- ^
- ISSN 0002-7863.
- ^ US 2710853, Edwards, W. M.; Robinson, I. M., "Polyimides of pyromellitic acid"
- ISBN 978-0-471-23896-6, retrieved 2 December 2020
- )
- )
- .
- S2CID 93989040.
- .
- ^ K. Faghihi, J. Appl. Polym. Sci., 2006, 102, 5062–5071. Y. Kung and S. Hsiao, J. Mater. Chem., 2011, 1746–1754. L. Burakowski, M. Leali and M. Angelo, Mater. Res., 2010, 13, 245–252.
- S2CID 233539984.
- S2CID 233539984.
- S2CID 235793266.
- ^ P2SI 900HT Tech Sheet. proofresearchacd.com
- S2CID 123400822.
- S2CID 136747058.
- ^ Krakauer, David (December 2006) Digital Isolation Offers Compact, Low-Cost Solutions to Challenging Design Problems. analog.com
- ^ Chen, Baoxing. iCoupler Products with isoPower Technology: Signal and Power Transfer Across Isolation Barrier Using Microtransformers. analog.com
- ^ "Apple to adopt speedy LCP circuit board tech across major product lines in 2018".
- ^ "Thermal Control Overview" (PDF). Sheldahl Multi Layer Insulation. Retrieved 28 December 2015.
- ^ What is a reverse osmosis water softener? wisegeek.net
- ^ Shuey, Harry F. and Wan, Wankei (22 December 1983) U.S. patent 4,532,041 Asymmetric polyimide reverse osmosis membrane, method for preparation of same and use thereof for organic liquid separations.
- ^ MCL (13 June 2017). "What is the Difference between FR4 and Polyamide PCB". mcl. Retrieved 4 September 2023.
- ^ Courtland, Rachel (10 May 2010). "Maiden voyage for first true space sail". The New Scientist. Retrieved 11 June 2010.
Further reading
- Modern Plastic Mid-October Encyclopedia Issue, Polyimide, thermoset, p. 146.
- Varun Ratta: POLYIMIDES: Chemistry & structure-property relationships – literature review (Chapter 1).


