Polio vaccine
Poliomyelitis | |
| Vaccine type | IPV: inactivated OPV: attenuated |
|---|---|
| Clinical data | |
| Trade names | Ipol, Poliovax, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601177 |
| License data | |
| Pregnancy category |
|
parenteral OPV: oral | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| DrugBank | |
| ChemSpider |
|
| | |
Polio vaccines are
The inactivated polio vaccines are very safe.[2] Mild redness or pain may occur at the site of injection.[2] Oral polio vaccines cause about three cases of vaccine-associated paralytic poliomyelitis per million doses given.[2] This compares with 5,000 cases per million who are paralysed following a polio infection.[8] Both types of vaccine are generally safe to give during pregnancy and in those who have HIV/AIDS but are otherwise well.[2] However, the emergence of circulating vaccine-derived poliovirus (cVDPV), a form of the vaccine virus that has reverted to causing poliomyelitis, has led to the development of novel oral polio vaccine type 2 (nOPV2) which aims to make the vaccine safer and thus stop further outbreaks of cVDPV2.[9]
The first successful demonstration of a polio vaccine was by
Polio vaccine is on the World Health Organization's List of Essential Medicines.[13][14]
Medical uses

Interruption of person-to-person transmission of the virus by vaccination is important in global
Inactivated
When the IPV (injection) is used, 90% or more of individuals develop protective antibodies to all three serotypes of polio virus after two doses of inactivated polio vaccine (IPV), and at least 99% are immune to polio virus following three doses. The duration of immunity induced by IPV is not known with certainty, although a complete series is thought to provide protection for many years.[17] IPV replaced the oral vaccine in many developed countries in the 1990s mainly due to the (small) risk of vaccine-derived polio in the oral vaccine.[18][19]
Attenuated
Oral polio vaccines were easier to administer than IPV, as it eliminated the need for sterile syringes and therefore was more suitable for mass vaccination campaigns. OPV also provided longer-lasting
One dose of OPV produces immunity to all three poliovirus serotypes in roughly 50% of recipients.
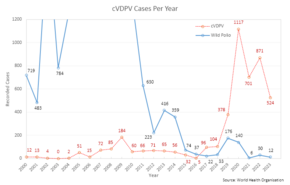
The trivalent (against wild types 1, 2, and 3) OPV has been used to nearly eradicate polio infection worldwide.[23] Led by the Global Polio Eradication Initiative, 155 countries switched to use the bivalent (against wild types 1 and 3) between 17 April and 1 May 2016.[24] The bivalent OPV is more effective against types 1 and 3, but does not cover type 2.[25] The switch to the bivalent vaccine and associated missing immunity against type 2 strains, among other factors, led to outbreaks of circulating vaccine-derived poliovirus type 2(cVDPV2), which increased from 2 cases in 2016 to 1037 cases in 2020.[26]
An improved oral vaccine (Novel oral polio vaccine type 2 - nOPV2) began development in 2011 and was granted emergency licencing in 2021, and subsequently full licensure in December 2023.[27] This has greater genetic stability than the traditional oral vaccine and is less likely to revert to a virulent form.[27][9]
Schedule
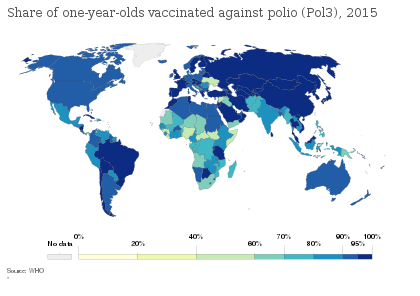
In countries with endemic polio or where the risk of imported cases is high, the WHO recommends OPV vaccine at birth followed by a primary series of three OPV doses and at least one IPV dose starting at 6 weeks of age, with a minimum of 4 weeks between OPV doses. In countries with >90% immunization coverage and low risk of importation, the WHO recommends one or two IPV doses starting at 2 months of age followed by at least two OPV doses, with the doses separated by 4–8 weeks depending on the risk of exposure. In countries with the highest levels of coverage and the lowest risks of importation and transmission, the WHO recommends a primary series of three IPV injections, with a booster dose after an interval of six months or more if the first dose was administered before 2 months of age.[2]
Side effects
The inactivated polio vaccines are very safe. Mild redness or pain may occur at the site of injection. Oral polio vaccine results in vaccine-associated paralytic poliomyelitis in about three per million doses. They are generally safe to be given to those who are pregnant and those who have HIV/AIDS but are otherwise well.[2]
Allergic reaction to the vaccine
Inactivated polio vaccine can cause an allergic reaction in a few people since the vaccine contains trace amounts of antibiotics, streptomycin, polymyxin B, and neomycin. It should not be given to anyone who has an allergic reaction to these medicines. Signs and symptoms of an allergic reaction, which usually appear within minutes or a few hours after receiving the injected vaccine, include breathing difficulties, weakness, hoarseness or wheezing, heart rate fluctuations, skin rash and dizziness.[29]
Vaccine-induced polio
A potential, adverse effect of the original OPV is its known ability to recombine to a form that causes neurological infection and paralysis.[30]
The vaccine-derived attenuated virus used in the OPV is normally excreted from vaccinated people for a limited period. Thus, in areas with poor sanitation and low vaccination coverage, it can spread to other unvaccinated individuals. Over time, and after passing through numerous hosts, it can potentially revert to a virulent form.[31] This genetic reversal of the pathogen to a virulent form takes a considerable time (at least 12 months) and does not affect the person who was originally vaccinated. Clinical disease, including paralysis, caused by vaccine-derived poliovirus (VDPV) is indistinguishable from that caused by wild polioviruses.[32] Outbreaks of vaccine-associated paralytic poliomyelitis (VAPP), caused by a circulating vaccine-derived poliovirus (cVDPV),[33] have been reported, and tend to occur in areas of low coverage by OPV, presumably because the OPV is itself protective against the related outbreak strain.[34][35] With wild polio cases at record lows, 2017 was the first year where more cases of cVDPV were recorded than the wild poliovirus, a trend that is expected to continue.[36]
To combat this, the WHO in 2016, decided to switch from the trivalent polio vaccine to the bivalent polio vaccine.[37] This vaccine no longer contains the type 2 polio virus because it was eradicated in 1999.[38]
In December 2023 an improved oral vaccine - nOPV2 - with greater genetic stability received full licensure. This is less likely to recombine to a form which can cause paralysis.[27][9]
Contamination concerns
In 1960, the
SV40 was found to be present in stocks of the injected form of the IPV in use between 1955 and 1963.
In 1998, the National Cancer Institute undertook a large study, using cancer case information from the institute's SEER database. The published findings from the study revealed no increased incidence of cancer in persons who may have received vaccine containing SV40.[45] Another large study in Sweden examined cancer rates of 700,000 individuals who had received potentially contaminated polio vaccine as late as 1957; the study again revealed no increased cancer incidence between persons who received polio vaccines containing SV40 and those who did not.[46] The question of whether SV40 causes cancer in humans remains controversial, however, and the development of improved assays for detection of SV40 in human tissues will be needed to resolve the controversy.[43]

During the race to develop an oral polio vaccine, several large-scale human trials were undertaken. By 1958, the National Institutes of Health had determined that OPV produced using the Sabin strains were the safest.
Manufacture
Inactivated
The Salk vaccine, IPV, is based on three wild,
In the United States, vaccine is administered along with the
Attenuated

OPV is an
OPV is usually provided in vials containing 10–20 doses of vaccine. A single dose of oral polio vaccine (usually two drops) contains 1,000,000 infectious units of Sabin 1 (effective against PV1), 100,000 infectious units of the Sabin 2 strain, and 600,000 infectious units of Sabin 3. The vaccine contains small traces of
History
In a generic sense, vaccination works by priming the immune system with an 'immunogen'. Stimulating immune response, by use of an infectious agent, is known as immunization. The development of immunity to polio efficiently blocks person-to-person transmission of wild poliovirus, thereby protecting both individual vaccine recipients and the wider community.[15]
The development of two polio vaccines led to the first modern mass
1930s
In the 1930s, poliovirus was perceived as especially terrifying, as little was known of how the disease was transmitted or how it could be prevented. This virus was also notable for primarily impacting affluent children, making it a prime target for vaccine development, despite its relatively low mortality and morbidity.[65] Despite this, the community of researchers in the field thus far had largely observed an informal moratorium on any vaccine development as it was perceived to present too high a risk for too little likelihood of success.[66][67]
This shifted in the early 1930s when American groups took up the challenge:
Kolmer's live vaccine
Kolmer began his vaccine development project in 1932 and ultimately focused on producing an attenuated or live virus vaccine. Inspired by the success of vaccines for rabies and yellow fever, he hoped to use a similar process to denature the polio virus.[68] In order to go about attenuating his polio vaccine, he repeatedly passed the virus through monkeys.[70] Using methods of production that were later described as "hair-raisingly amateurish, the therapeutic equivalent of bath-tub gin,"[71] Kolmer ground the spinal cords of his infected monkeys and soaked them in a salt solution. He then filtered the solution through mesh, treated it with ricinolate, and refrigerated the product for 14 days[68] to ultimately create what would later be prominently critiqued as a "veritable witches brew".[72]
In keeping with the norms of the time, Kolmer completed a relatively small animal trial with 42 monkeys before proceeding to
Brodie's inactivated vaccine
At nearly the same time as Kolmer's project,
Soon after, following a similar protocol to Kolmer, Brodie proceeded with self experimentation upon himself and his co-workers at the NYC Health Department laboratory.[67] Brodie's progress was eagerly covered by popular press as the public hoped for a successful vaccine to become available.[77] Such reporting did not make mention of the 12 children in a New York City Asylum who were subjected to early safety trials.[67] As none of the subjects experienced ill effects, Park, described by contemporaries as "never one to let grass grow under his feet,"[78] declared the vaccine safe.[70] When a severe polio outbreak overwhelmed Kern County, California it became the first trial site for the new vaccine on very short notice. Between November 1934 - May 1935, over 1,500 doses of the vaccine were administered in Kern County. While initial results were very promising, insufficient staffing and poor protocol design left Brodie open to criticism when he published the California results in August 1935.[77][73] Through private physicians, Brodie also conducted a broader field study, including 9,000 children who received the vaccine and 4,500 age- and location-matched controls who did not receive a vaccine. Again, results were promising. Of those who received the vaccine, only a few went on to develop polio. Most had been exposed prior to vaccination and none had received the full series of vaccine doses being studied.[73] Additionally, a polio epidemic in Raleigh, North Carolina provided an opportunity for the U.S. Public Health Service to conduct a highly structured trial of the Brodie vaccine using funding from the Birthday Ball Commission.[70][73]
Academic reception
While their work was ongoing, the larger community of bacteriologists began to raise concerns regarding the safety and efficacy of the new poliovirus vaccines.[66] At this time there was very little oversight of medical studies, and ethical treatment of study participants largely relied upon moral pressure from peer academic scientists.[68] Brodie's inactivated vaccines faced scrutiny from many who felt killed virus vaccines could not be efficacious. While researchers were able to replicate the tissue immunity he had produced in his animal trials, prevailing wisdom was that humoral immunity was essential for an efficacious vaccine.[73] Kolmer directly questioned the killed virus approach in scholarly journals.[75] Kolmer's studies however had raised even more concern with increasing reports of children becoming paralysed following vaccination with his live virus vaccine and notably, with paralysis beginning at the arm rather than the foot in many cases.[79] Both Kolmer and Brodie were called to present their research at the Annual Meeting of the American Public Health Association in Milwaukee WI in October 1935.[66] Additionally, Thomas M. Rivers was asked to discuss each of the presented papers as a prominent critic of the vaccine development effort.[66] This resulted in the APHA arranging a Symposium on Poliomyelitis to be delivered at the Annual Meeting of their Southern Branch the following month.[66] It was during the discussion at this meeting that James Leake of the U.S. Public Health Service stood to immediately present clinical evidence that the Kolmer vaccine had caused several deaths and then allegedly accused Kolmer of being a murderer.[66] As Rivers recalled in his oral history, "All hell broke loose, and it seemed as if everybody was trying to talk at the same time....Jimmy Leake used the strongest language that I have ever heard used at a scientific meeting."[66] In response to the attacks from all sides, Brodie was reported to have stood up and stated, "It looks as though, according to Dr. Rivers, my vaccine is no good, and, according to Dr. Leake, Dr Kolmer's is dangerous."[66] Kolmer simply responded by stating, "Gentlemen, this is one time I wish the floor would open up and swallow me."[66] Ultimately, Kolmer's live vaccine was undoubtedly shown to be dangerous and had already been withdrawn in September 1935 prior to the Milwaukee meeting.[79][74][73] While the consensus of the symposium was largely skeptical of the efficacy of Brodie's vaccine, its safety was not in question and the recommendation was for a much larger well-controlled trial.[79] However, when three children became ill with paralytic polio following a dose of the vaccine, the directors of the Warm Springs Foundation in Georgia (acting as the primary funders for the project) requested it be withdrawn in December 1935.[79] Following its withdrawal, the previously observed moratorium on human poliomyelitis vaccine development resumed and there would not be another attempt for nearly 20 years.[73][74]
While Brodie had arguably made the most progress in the pursuit of a poliovirus vaccine, he suffered the most significant career repercussions due to his status as a less widely known researcher.[77] Modern researchers recognize that Brodie may well have developed an effective polio vaccine, however the basic science and technology of the time was insufficient to understand and utilize this breakthrough.[73] Brodie's work using formalin-inactivated virus would later become the basis for the Salk vaccine, but he would not live to see this success.[73] Brodie was fired from his position within three months of the symposium's publication.[73] While he was able to find another laboratory position, he died of a heart attack only three years later at age 36.[73][77] By contrast, Park, who was believed in the community to be reaching senility at this point in his older age, was able to retire from his position with honors[66] prior to his death in 1939.[70] Kolmer, already an established and well respected researcher, returned to Temple University as a professor of medicine.[73] Kolmer had a very productive career, receiving multiple awards, and publishing countless papers, articles, and textbooks up until his retirement in 1957.[70][66][74][80]
1948
A breakthrough came in 1948 when a research group headed by
1950–1955

During the early 1950s, polio rates in the U.S. were above 25,000 annually; in 1952 and 1953, the U.S. experienced an outbreak of 58,000 and 35,000 polio cases, respectively, up from a typical number of some 20,000 a year, with deaths in those years numbering 3,200 and 1,400.
Jonas Salk


The first effective polio vaccine was developed in 1952 by
Salk's vaccine was then used in a test called the Francis Field Trial, led by
The results of the field trial were announced 12 April 1955 (the tenth anniversary of the death of President
A week before the announcement of the Francis Field Trial results in April 1955, Pierre Lépine at the Pasteur Institute in Paris had also announced an effective polio vaccine.[96][97]
Safety incidents
In April 1955, soon after mass polio vaccination began in the US, the Surgeon General began to receive reports of patients who contracted paralytic polio about a week after being vaccinated with Salk polio vaccine from the Cutter pharmaceutical company, with the paralysis starting in the limb the vaccine was injected into.[98] The Cutter vaccine had been used in vaccinating 409,000 children in the western and midwestern United States.[99] Later investigations showed that the Cutter vaccine had caused 260 cases of polio, killing 11.[98] In response, the Surgeon General pulled all polio vaccines made by Cutter Laboratories from the market, but not before 260 cases of paralytic illness had occurred. Eli Lilly, Parke-Davis, Pitman-Moore and Wyeth polio vaccines were also reported to have paralyzed numerous children. It was soon discovered that some lots of Salk polio vaccine made by Cutter, Wyeth, and the other labs had not been properly inactivated, allowing live poliovirus into more than 100,000 doses of vaccine. In May 1955, the National Institutes of Health and Public Health Services established a Technical Committee on Poliomyelitis Vaccine to test and review all polio vaccine lots and advise the Public Health Service as to which lots should be released for public use. These incidents reduced public confidence in polio vaccine, leading to a drop in vaccination rates.[100]
1961

At the same time that Salk was testing his vaccine, both Albert Sabin and Hilary Koprowski continued working on developing a vaccine using live virus. During a meeting in Stockholm to discuss polio vaccines in November 1955, Sabin presented results obtained on a group of 80 volunteers, while Koprowski read a paper detailing the findings of a trial enrolling 150 people.[47] Sabin and Koprowski both eventually succeeded in developing vaccines. Because of the commitment to the Salk vaccine in America, Sabin and Koprowski both did their testing outside the United States, Sabin in Mexico[57] and the Soviet Union,[101] Koprowski in the Congo and Poland.[49] In 1957, Sabin developed a trivalent vaccine containing attenuated strains of all three types of poliovirus.[101] In 1959, ten million children in the Soviet Union received the Sabin oral vaccine. For this work, Sabin was given the medal of the Order of Friendship of Peoples, described as the Soviet Union's highest civilian honor.[102] Sabin's oral vaccine using live virus came into commercial use in 1961.[2]
Once Sabin's oral vaccine became widely available, it supplanted Salk's injected vaccine, which had been tarnished in the public's opinion by the
1987
An enhanced-
1988

A global effort to eradicate polio, led by the World Health Organization (WHO), UNICEF,[106] and the Rotary Foundation, began in 1988, and has relied largely on the oral polio vaccine developed by Albert Sabin and Mikhail Chumakov (Sabin-Chumakov vaccine).[107]
After 1990
Polio was eliminated in the Americas by 1994.[108] The disease was officially eliminated in 36 Western Pacific countries, including China and Australia, in 2000.[109][110] Europe was declared polio-free in 2002.[111] Since January 2011, no cases of the disease have been reported in India, hence in February 2012, the country was taken off the WHO list of polio-endemic countries. In March 2014, India was declared a polio-free country.[112][113][114]
Although poliovirus transmission has been interrupted in much of the world, transmission of wild poliovirus does continue and creates an ongoing risk for the importation of wild poliovirus into previously polio-free regions. If importations of poliovirus occur, outbreaks of poliomyelitis may develop, especially in areas with low vaccination coverage and poor sanitation. As a result, high levels of vaccination coverage must be maintained.
Polio vaccination programs have been resisted by some people in Pakistan, Afghanistan, and Nigeria - the three countries as of 2017 with remaining polio cases. Almost all Muslim religious and political leaders have endorsed the vaccine,[117] but a fringe minority believes that the vaccines are secretly being used for sterilization of Muslims.[54] The fact that the CIA organized a fake vaccination program in 2011 to help find Osama bin Laden is an additional cause of distrust.[118] In 2015, the WHO announced a deal with the Taliban to encourage them to distribute the vaccine in areas they control.[119] However, the Pakistani Taliban was not supportive. On 11 September 2016, two unidentified gunmen associated with the Pakistani Taliban, Jamaat-ul-Ahrar, shot Zakaullah Khan, a doctor who was administering polio vaccines in Pakistan. The leader of the Jamaat-ul-Ahrar claimed responsibility for the shooting and stated that the group would continue this type of attack. Such resistance to and skepticism of vaccinations has consequently slowed down the polio eradication process within the two remaining endemic countries.[118]
Travel requirements
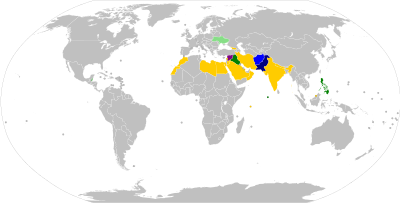
Travellers who wish to enter or leave certain countries must be vaccinated against polio, usually at most 12 months and at least 4 weeks before crossing the border, and be able to present a vaccination record/certificate at the border checks.[120]: 25–27 Most requirements apply only to travel to or from so-called 'polio-endemic', 'polio-affected', 'polio-exporting', 'polio-transmission', or 'high-risk' countries.[121] As of August 2020, Afghanistan and Pakistan are the only polio-endemic countries in the world (where wild polio has not yet been eradicated).[122] Several countries have additional precautionary polio vaccination travel requirements, for example to and from 'key at-risk countries', which as of December 2020 include China, Indonesia, Mozambique, Myanmar, and Papua New Guinea.[121][123]
| Polio vaccination requirements for international travel[121] | |
|---|---|
| Country | Details |
| Travellers from polio-endemic countries (Pakistan) need Carte Jaune proof of polio vaccination (received between 4 weeks and 12 months before departure) upon arrival. Residents and ALL travellers staying in Afghanistan longer than 4 weeks need proof of polio vaccination (received between 4 weeks and 12 months before departure) when departing from Afghanistan.[121][124]
| |
| Travellers from Afghanistan and Pakistan need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival. Belize residents travelling countries with confirmed polio cases also need proof of vaccination.[125]
| |
| Travellers from polio-exporting countries need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[126]
| |
| Travellers from Afghanistan, Angola, Benin, Cameroon, the Central African Republic, China, Congo-Kinshasa, Ethiopia, Ghana, Indonesia, Kenya, Mozambique, Myanmar, Niger, Nigeria, Pakistan, Papua New Guinea, Philippines, and Somalia need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[127]
| |
| Travellers from at-risk countries need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival. Travellers without proof are offered OPV vaccination upon arrival.[128]
| |
| Travellers from Afghanistan, Congo-Kinshasa, Ethiopia, Kenya, Nigeria, Pakistan, Somalia, and Syria need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[129]
| |
| Travellers from Afghanistan, Pakistan, and Nigeria need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival. Travellers without proof will be vaccinated upon arrival.[130]
| |
| Travellers aged 15+ from Afghanistan and Pakistan need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival; children under age 15 must have received three doses of polio vaccine before travel. Travellers without proof will be vaccinated upon arrival. Travellers departing Iraq to Afghanistan and Pakistan must also provide proof of vaccination upon departure.[131]
| |
| Travellers from Afghanistan and Pakistan need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[132]
| |
| Travellers from and to polio-affected countries need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[133]
| |
| Travellers from Afghanistan and Pakistan need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[134]
| |
| Travellers from and to polio-exporting countries, as well as Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[135]
| |
| Travellers from polio-affected countries need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[136]
| |
| Travellers from Afghanistan, Kenya, Nigeria, Pakistan, and Papua New Guinea need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[137]
| |
| Travellers from polio-exporting countries need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[138]
| |
| Travellers from ALL countries planning to stay in Pakistan for more than 4 weeks need | |
| Travellers from or to high-risk countries need ongoing local VDPV2 outbreak, the government recommends all others travellers to consider getting a polio vaccine or booster dose, depending on their situation.[140]
| |
| Travellers from polio-exporting countries (identified by Qatar as: Afghanistan, Nigeria, Pakistan and Philippines) need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[141]
| |
| Travellers from polio-endemic countries as identified by WHO (Afghanistan and Pakistan) need | |
| Travellers from active-transmission (including wild or vaccine-derived poliovirus) and at-risk countries, as well as all travellers from Afghanistan, Congo-Kinshasa, Mozambique, Myanmar, Niger, Nigeria, Pakistan, Papua New Guinea, Somalia, Syria, and Yemen, need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival. Regardless of immunisation status, all travellers from Afghanistan, Myanmar, Nigeria, Pakistan, Papua New Guinea, Somalia, Syria, and Yemen will be given an Oral Polio Vaccine dose upon arrival.[143]
| |
| Travellers from countries with polio outbreaks need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival.[144]
| |
| Travellers from Cameroon, Equatorial Guinea, and Pakistan need Carte Jaune proof of OPV or IPV vaccination (received between 4 weeks and 12 months before departure) upon arrival. ALL Syria residents departing Syria to any country also need proof of vaccination.[145]
| |
| Long-term visitors departing to states with wild or circulating vaccine-derived poliovirus transmission should present Carte Jaune proof of vaccination with at least one dose of bivalent OPV or IPV (received between 4 weeks and 12 months before departure). Persons obliged to undertake urgent international travel must be immunised with a single dose of polio vaccine before their departure.[121] There is also risk of poliovirus transmission inside Ukraine itself, and travellers to Ukraine are recommended to be up to date with their polio vaccination before entry.[146]
| |
Society and culture
Cost
As of 2015[update], the
Misconceptions
A misconception has been present in Pakistan that polio vaccine contained
References
- ^ Use During Pregnancy and Breastfeeding
- ^ PMID 27039410.
- hdl:10665/357168.
- S2CID 25327986.
- PMID 6740085.
- ^ "Global Wild Poliovirus 2014–2019" (PDF). Archived (PDF) from the original on 3 February 2019. Retrieved 3 February 2019.
- ^ "Does polio still exist? Is it curable?". World Health Organization (WHO). Archived from the original on 29 May 2018. Retrieved 21 May 2018.
- ^ "Poliomyelitis". World Health Organization (WHO). Archived from the original on 18 April 2017. Retrieved 25 April 2017.
- ^ a b c "GPEI-nOPV2". Archived from the original on 27 July 2021. Retrieved 1 August 2021.
- ^ a b c Fox M (20 April 2013). "Hilary Koprowski, Who Developed First Live-Virus Polio Vaccine, Dies at 96". The New York Times. Archived from the original on 25 August 2017. Retrieved 8 September 2017.
- ISBN 978-2742007752. Archivedfrom the original on 8 September 2017.
- PMID 16422178.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ PMID 10568615.
- PMID 1846972.
- ^ a b Robertson S. Module 6: Poliomyelitis (PDF). The Immunological Basis for Immunization Series. World Health Organization (WHO). WHO/EPI/GEN/93.16. Archived from the original (PDF) on 19 October 2019. Retrieved 18 October 2019.
- ^ Public Health Agency of Canada (18 July 2007). "Poliomyelitis vaccine: Canadian Immunization Guide". www.canada.ca. Archived from the original on 27 July 2021. Retrieved 1 August 2021.
- ^ "Polio Vaccination: What Everyone Should Know | CDC". www.cdc.gov. 25 March 2021. Archived from the original on 16 January 2021. Retrieved 1 August 2021.
- PMID 15857985.
- ^ a b c d e f Atkinson W, Hamborsky J, McIntyre L, Wolfe S, eds. (2008). Epidemiology and Prevention of Vaccine-Preventable Diseases (The Pink Book) (PDF) (10th ed. (2nd printing) ed.). Washington, D.C.: Public Health Foundation. Archived from the original (PDF) on 24 September 2008. Retrieved 29 November 2008.
- ^ PMID 9024465.
- PMID 28081056.
- ^ Al Idrus A (13 April 2016). "Worldwide switch to bivalent oral polio vaccine to kick off on Sunday". fiercepharma.com. Archived from the original on 15 April 2016. Retrieved 1 May 2016.
- PMID 28838179.
- ^ "GPEI Strategy for the Response to cVDPV2 2020–2021" (PDF). Polio Global Eradication Initiative. Archived (PDF) from the original on 1 August 2021. Retrieved 1 August 2021.
- ^ a b c "GPEI-OPV Oral polio vaccine". Global Polio Eradication Initiative - World Health Organization. 12 April 2024. Retrieved 12 April 2024.
- ^ "Share of one-year-olds vaccinated against polio (Pol3)". Our World in Data. Archived from the original on 22 February 2020. Retrieved 4 March 2020.
- ^ "Common and Rare Side Effects for poliovirus vaccine injection". www.webmd.com. Archived from the original on 24 August 2021. Retrieved 24 August 2021.
- PMID 15564462.
- ^ "What is vaccine-derived polio?". World Health Organization (WHO). April 2017. Archived from the original on 11 April 2020. Retrieved 18 October 2019.
- ^ Cono J, Alexander LN (2002). "Chapter 10: Poliomyelitis" (PDF). Vaccine-Preventable Disease Surveillance Manual (3rd ed.). Archived from the original (PDF) on 22 October 2011.
- ^ "What is vaccine-derived polio?". World Health Organization (WHO). October 2014. Archived from the original on 7 September 2015. Retrieved 6 September 2015.
- from the original on 14 April 2020. Retrieved 30 June 2019.
- PMID 12857906.
- ^ Kunasekaran M. "Eradication of polio – Is Syria being left behind?". UNSW School of Public Health and Community Medicine. Archived from the original on 6 October 2018. Retrieved 6 October 2018.
- ^ "Replacing trivalent OPV with bivalent OPV". World Health Organization (WHO). Archived from the original on 11 March 2015. Retrieved 30 May 2019.
- ^ "Poliomyelitis". World Health Organization (WHO). 22 July 2019. Archived from the original on 17 October 2019. Retrieved 18 October 2019.
- ^ a b c d "Simian Virus 40 (SV40), Polio Vaccine, and Cancer". Vaccine Safety. Centers for Disease Control. 22 April 2004. Archived from the original on 22 May 2013. Retrieved 22 May 2013.
- S2CID 31275908.
- S2CID 795975.
- PMID 12798456.
- ^ from the original on 20 April 2020. Retrieved 30 June 2019.
- ^ Bookchin D (7 July 2004). "Vaccine scandal revives cancer fear". New Scientist. Archived from the original on 20 July 2004. Retrieved 29 November 2008.
- PMID 9450713.
- PMID 9776244.
- ^ a b c d e f "Competition to develop an oral vaccine". Conquering Polio. Sanofi Pasteur SA. 2 February 2007. Archived from the original on 7 October 2007.
- ^ PMID 11264036.
- ^ PMID 14410975.
- ^ Collins H (6 November 2000). "The Gulp Heard Round the World". The Philadelphia Inquirer. p. D-1. Archived from the original on 5 April 2004. Retrieved 29 November 2008.
- ^ "Nigeria Muslims oppose polio vaccination". BBC News Online. 27 June 2002. Archived from the original on 29 November 2008. Retrieved 29 November 2008.
- ^ Dugger CW, McNeil DG (20 March 2006). "Rumor, Fear and Fatigue Hinder Final Push to End Polio". The New York Times. Archived from the original on 10 December 2008. Retrieved 29 November 2008.
- ^ "Anti-polio vaccine Malians jailed". BBC News Online. 12 May 2005. Archived from the original on 10 January 2006. Retrieved 29 November 2008.
- ^ PMID 17388657.
- ^ from the original on 9 July 2020. Retrieved 30 June 2019.
- ^ ISBN 978-0-11-322528-6. Archived from the original(PDF) on 15 June 2007.
- ^ PMID 14440553.
- S2CID 45655185.
- PMID 6760938.
- PMID 12477816.
- PMID 9882296.
- PMID 15489385.
- ISBN 978-0-19-924474-4.[page needed]
- ISBN 978-92-75-11607-4.[page needed]
- ^ PMC 1472109.
- ^ ISBN 978-0262020268.
- ^ OCLC 877210630.
- ^ a b c d Hovern D (2018). The Trials and Triumphs of the American Polio Vaccine (Thesis). Cooper Medical School of Rowan University. Archived from the original on 21 July 2021. Retrieved 19 July 2021.
- ^ "Vaccine Testing and Vulnerable Human Subjects | History of Vaccines". www.historyofvaccines.org. Archived from the original on 2 August 2021. Retrieved 18 July 2021.
- ^ OCLC 38243151.
- OCLC 630735949.
- ^ Paul, J.R. (1971). A History of Poliomyelitis. New Haven: Yale University Press.[ISBN missing][page needed]
- ^ PMID 2692236.
- ^ ISBN 978-0-300-12605-1.
- ^ from the original on 19 July 2021. Retrieved 19 July 2021.
- .
- ^ a b c d e Johnston K (22 February 2021). "The tragic story of a Canadian vaccine trailblazer". Macleans.ca. Archived from the original on 19 July 2021. Retrieved 18 July 2021.
- ISBN 978-0-19-515294-4. Archivedfrom the original on 25 November 2023. Retrieved 23 March 2022.
- ^ PMID 18014373.
- ^ "John A. Kolmer, M.D." Archived from the original on 1 December 2017. Retrieved 28 November 2017.
- PMID 17794160.
- ^ "The Nobel Prize in Physiology or Medicine 1954". The Nobel Foundation. Archived from the original on 19 December 2008. Retrieved 29 November 2008.
- PMID 13034471.
- ^ Ochmann S, Roser M (9 November 2017). "Polio – Polio Cases (OWID based on US Public Health Service (1910–1951) and US Center for Disease Control (1960–2010))". Our World in Data. Archived from the original on 28 March 2018. Retrieved 26 March 2018.
- PMID 19316151.
- ^ Koprowski H (15 October 2010). "Interview with Hilary Koprowski, sourced at History of Vaccines website". College of Physicians of Philadelphia. Archived from the original on 15 May 2016. Retrieved 15 October 2010.
- ^ "science.ca : Leone N. Farrell". www.science.ca. Archived from the original on 28 June 2020. Retrieved 1 August 2021.
- ^ Elliott CK (March 2011). "Leone Norwood Farrell, PhD". polioplace. Post-polio Health International. Archived from the original on 9 August 2019. Retrieved 10 August 2019.
- ISBN 978-0-7637-2932-5. Archivedfrom the original on 21 July 2014. Retrieved 22 February 2011.
- ^ Conis E (2016). "Political Ills". Distillations. 2 (2): 34–37. Archived from the original on 20 March 2018. Retrieved 27 March 2018.
- ^ Oshinsky D (Winter 2010). "Miracle Workers". American Heritage. Archived from the original on 3 September 2014. Retrieved 1 September 2014.
- ^ "Polio Victory Remembered as March of Dimes Marks 50th Anniversary of Salk Vaccine Field Trials". News Desk. 26 April 2004. Archived from the original on 27 September 2015. Retrieved 14 November 2014.
- ISBN 978-0-688-09494-2.
- ISBN 978-0-7618-0144-3.
- PMID 6371280.
- ^ The Pasteur Institute stated that an antipoliomyelitis vaccine, developed by Professor Pierre Lepine would soon be produced in large quantities. (Times, London, 4 April 1955).
- ^ "Pierre Lépine (1901–1989) – Notice biographique". Archives de l'Institut Pasteur. Archived from the original on 28 November 2016. Retrieved 28 November 2016.
- ^ OCLC 919967059.
- ^ "Technical Report on Poliomyelitis Vaccine". Public Health Reports. 70 (8): 742. August 1955.
- PMID 20718820.
- ^ a b "Two Vaccines: Sabin and Salk". Smithsonian National Museum of American History. 27 September 2021. Archived from the original on 20 January 2017. Retrieved 24 April 2017.
- ^ "Sabin receives highest Soviet civilian honor". U.P.I. archives. 20 November 1986. Archived from the original on 25 April 2017. Retrieved 25 April 2017.
- ^ Diphtheria and Tetanus Toxoids and Acellular Pertussis Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus Vaccine Combined
- ^ a b "Pediarix". U.S. Food and Drug Administration (FDA). 6 November 2019. Archived from the original on 22 September 2019. Retrieved 8 July 2020.
- ^ "FDA Statistical Review and Evaluation" (PDF). U.S. Food and Drug Administration (FDA). Archived from the original on 6 August 2020. Retrieved 8 July 2020.
- ^ Mansoor H (23 October 2015). "Can India's social mobilisation strategy work in Pakistan?". The Express Tribune. Archived from the original on 24 October 2015. Retrieved 24 October 2015.
- ^ Mastny L (25 January 1999). "Eradicating Polio: A Model for International Cooperation". Worldwatch Institute. Archived from the original on 3 December 2006. Retrieved 29 November 2008.
- ^ from the original on 21 May 2017.
- .
- PMID 12206379.
- ^ "Europe achieves historic milestone as Region is declared polio-free" (Press release). World Health Organization (WHO). 21 June 2002. Archived from the original on 16 September 2008. Retrieved 23 August 2008.
- ^ Ray K (26 February 2012). "India wins battle against dreaded polio". Deccan Herald.
- ^ "India polio-free for a year: 'First time in history we're able to put up such a map'". The Telegraph. 26 February 2012. Archived from the original on 27 February 2012. Retrieved 26 February 2012.
- ^ "India three years polio-free". World Health Organization (WHO). Archived from the original on 1 March 2017. Retrieved 19 February 2017.
- ^ Barron L (4 November 2013). "Armenian Health Ministry: Syrian Armenian children need polio vaccine". CISTran Finance. Archived from the original on 19 December 2013. Retrieved 18 December 2013.
- ISBN 978-0-12-417127-5. Archivedfrom the original on 23 April 2017.
- ^ Charles Kurzman. The Missing Martyrs. Oxford University Press. p. 130.
Resistance to polio vaccination is a fringe position. Almost every Muslim scholar and political leader has endorsed the vaccine, and all but a few Muslim-majority countries have wiped out the disease entirely.
- ^ a b "Pakistan polio official killed in Peshawar: police". The Daily Star: Lebanon. 11 September 2016. Archived from the original on 11 September 2016. Retrieved 11 September 2016.
- ^ Najafizada E (15 December 2015). "Taliban Join Global Effort to Kill Off Polio in 2016". Bloomberg.com. Archived from the original on 28 February 2017.
- ^ "International Travel and Health. Chapter 6 – Vaccine-preventable diseases and vaccines (2019 update)" (PDF). World Health Organization. United Nations. 2020. Archived (PDF) from the original on 11 April 2020. Retrieved 2 December 2020.
- ^ a b c d e f g h "Countries with risk of yellow fever transmission and countries requiring yellow fever vaccination (July 2019)". World Health Organization. United Nations. 4 July 2019. Archived from the original on 27 January 2021. Retrieved 2 December 2020.
- ^ Scherbel-Ball N (25 August 2020). "Africa declared free of polio". BBC News. Archived from the original on 26 August 2020. Retrieved 25 August 2020.
- ^ "Key At-Risk Countries". Global Polio Eradication Initiative. World Health Organization. Archived from the original on 29 April 2021. Retrieved 2 December 2020.
- ^ "Afghanistan Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 1 October 2020. Archived from the original on 21 October 2020. Retrieved 2 December 2020.
- ^ "Belize Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Brunei Darussalam Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 1 December 2020. Retrieved 2 December 2020.
- ^ "Egypt Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 8 March 2021. Retrieved 2 December 2020.
- ^ "Georgia Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 17 January 2021. Retrieved 2 December 2020.
- ^ "India Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Iran Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Iraq Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 20 January 2021. Retrieved 2 December 2020.
- ^ "Jordan Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Lebanon Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Libya Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Maldives Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Morocco Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 21 October 2020. Retrieved 2 December 2020.
- ^ "Nepal Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 21 September 2020. Retrieved 2 December 2020.
- ^ "Oman Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 15 August 2020. Retrieved 2 December 2020.
- ^ "Pakistan Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 21 October 2020. Retrieved 2 December 2020.
- ^ "Philippines Recommended Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 25 October 2020. Retrieved 2 December 2020.
- ^ "Qatar Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 2 December 2020. Retrieved 2 December 2020.
- ^ "Saint Kitts & Nevis Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 4 December 2020. Retrieved 2 December 2020.
- ^ "Saudi Arabia Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 27 February 2021. Retrieved 2 December 2020.
- ^ "Seychelles Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 4 August 2020. Retrieved 2 December 2020.
- ^ "Syria Required Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 30 October 2020. Retrieved 2 December 2020.
- ^ "Ukraine Recommended Vaccinations: Polio". iamat.org. International Association for Medical Assistance to Travellers (IAMAT). 23 October 2020. Archived from the original on 24 January 2021. Retrieved 2 December 2020.
- ^ "Availability and price of inactivated polio vaccine". The Global Polio Eradication Initiative. Archived from the original on 11 April 2015.
- ^ "Impotence fears hit polio drive". BBC News Online. 25 January 2007. Archived from the original on 9 October 2015. Retrieved 15 December 2015.
- ^ Junaidi I (14 January 2015). "Lab tests show polio vaccine is not 'Haram'". dawn.com. Archived from the original on 22 December 2015. Retrieved 15 December 2015.
Further reading
- Ramsay M, ed. (2013). "Polio: the green book, chapter 26". Immunisation against infectious disease. London: Public Health England. Archived from the original on 12 November 2019. Retrieved 23 December 2019.
- Hall E, Wodi AP, Hamborsky J, Morelli V, Schillie S, eds. (2021). "Chapter 18: Poliomyelitis". Epidemiology and Prevention of Vaccine-Preventable Diseases (14th ed.). Washington D.C.: U.S. Centers for Disease Control and Prevention (CDC). Archived from the original on 30 December 2016. Retrieved 23 December 2019.
- Routh JA, Oberste MS, Patel M (2018). "Chapter 12: Poliomyelitis". In Roush SW, Baldy LM, Hall MH (eds.). Manual for the surveillance of vaccine-preventable diseases. Atlanta, Georgia: U.S. Centers for Disease Control and Prevention (CDC). Archived from the original on 1 August 2020. Retrieved 23 December 2019.
External links
- "Polio Vaccine Information Statement". Centers for Disease Control and Prevention (CDC). August 2021.
- History of Vaccines Website – History of Polio History of Vaccines, a project of the College of Physicians of Philadelphia
- PBS.org – 'People and Discoveries: Salk Produces Polio Vaccine 1952', Public Broadcasting Service(PBS)
- "IPOL – Poliovirus Vaccine Inactivated (Monkey Kidney Cell)". U.S. Food and Drug Administration (FDA). 11 December 2019. STN: 103930.
- Poliovirus Vaccines at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
