Tacticity
| Polymer science |
|---|
 |

Tacticity (from
A tactic macromolecule in the

Describing tacticity





Diads
Two adjacent structural units in a polymer molecule constitute a diad. Diads overlap: each structural unit is considered part of two diads, one diad with each neighbor. If a diad consists of two identically oriented units, the diad is called a meso diad (abbreviated m) as in a
Triads
The stereochemistry of macromolecules can be defined even more precisely with the introduction of triads. An isotactic triad (mm) is made up of two adjacent meso diads, a syndiotactic triad (also spelled syndyotactic[2]) (rr) consists of two adjacent racemo diads, and a heterotactic triad (rm) is composed of a racemo diad adjacent to a meso diad. The mass fraction of isotactic (mm) triads is a common quantitative measure of tacticity.
When the stereochemistry of a macromolecule is considered to be a Bernoulli process, the triad composition can be calculated from the probability Pm of a diad being meso. For example, when this probability is 0.25 then the probability of finding:
- an isotactic triad is Pm2, or 0.0625
- an heterotactic triad is 2Pm(1–Pm), or 0.375
- a syndiotactic triad is (1–Pm)2, or 0.5625
with a total probability of 1. Similar relationships with diads exist for tetrads.[3]: 357
Tetrads, pentads, etc.
The definition of tetrads and pentads introduce further sophistication and precision to defining tacticity, especially when information on long-range ordering is desirable. Tacticity measurements obtained by
Other conventions for quantifying tacticity
The primary convention for expressing tacticity is in terms of the relative weight fraction of triad or higher-order components, as described above. An alternative expression for tacticity is the average length of meso and racemo sequences within the polymer molecule. The average meso sequence length may be approximated from the relative abundance of pentads as follows:[4]
Polymers

Isotactic polymers
Isotactic polymers are composed of isotactic macromolecules (IUPAC definition).
Syndiotactic polymers
In syndiotactic or syntactic macromolecules the substituents have alternate positions along the chain. The macromolecule consists 100% of racemo diads. Syndiotactic
Atactic polymers
In atactic macromolecules the substituents are placed randomly along the chain. The percentage of meso diads is between 1 and 99%. With the aid of spectroscopic techniques such as NMR it is possible to pinpoint the composition of a polymer in terms of the percentages for each triad.[8]
Polymers that are formed by
Atactic polymers are technologically very important. A good example is polystyrene (PS). If a special catalyst is used in its synthesis it is possible to obtain the syndiotactic version of this polymer, but most industrial polystyrene produced is atactic. The two materials have very different properties because the irregular structure of the atactic version makes it impossible for the polymer chains to stack in a regular fashion. The result is that, whereas syndiotactic PS is a semicrystalline material, the more common atactic version cannot crystallize and forms a glass instead. This example is quite general in that many polymers of economic importance are atactic glass formers.
Eutactic polymers
In eutactic macromolecules, substituents may occupy any specific (but potentially complex) sequence of positions along the chain. Isotactic and syndiotactic polymers are instances of the more general class of eutactic polymers, which also includes heterogeneous macromolecules in which the sequence consists of substituents of different kinds (for example, the side-chains in proteins and the bases in nucleic acids).
Head/tail configuration
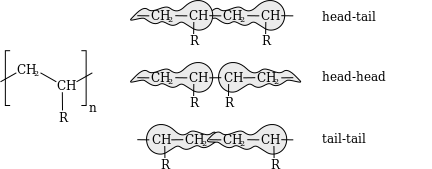
In vinyl polymers the complete configuration can be further described by defining polymer head/tail configuration. In a regular macromolecule all monomer units are normally linked in a head to tail configuration so that all β-substituents are separated by three carbon atoms. In head to head configuration this separation is only by 2 carbon atoms and the separation with tail to tail configuration is by 4 atoms. Head/tail configurations are not part of polymer tacticity but should be taken into account when considering polymer defects.
Techniques for measuring tacticity
Tacticity may be measured directly using
Other techniques sensitive to tacticity include
References
- ISBN 0-412-22170-5
- ^ Webster's Third New International Dictionary of the English Language, Unabridged; Oxford English Dictionary.
- S2CID 59059402.
- .
- ^ Stevens, P. S. Polymer Chemistry: An Introduction, 3rd ed.; Oxford Press: New York, 1999; pp 234-235
- ^ Brandrup, Immergut, Grulke (Editors), Polymer Handbook 4th edition, Wiley-Interscience, New York, 1999. VI/11
- S2CID 98943399. Archived from the original(PDF) on 2019-07-14. Retrieved 2019-07-13.
- .
- .
- .
- .
- .