Upper critical solution temperature
| Polymer science |
|---|
 |
The upper critical solution temperature (UCST) or upper consolute temperature is the
A solid state example is the palladium-hydrogen system which has a solid solution phase (H2 in Pd) in equilibrium with a hydride phase (PdHn) below the UCST at 300 °C. Above this temperature there is a single solid solution phase.[2]: 186
In the phase diagram of the mixture components, the UCST is the shared maximum of the concave down spinodal and binodal (or coexistence) curves. The UCST is in general dependent on pressure.
The phase separation at the UCST is in general driven by unfavorable energetics; in particular, interactions between components favor a partially demixed state.[4]
Polymer-solvent mixtures
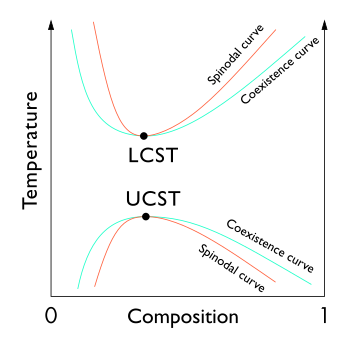
Some polymer solutions also have a lower critical solution temperature (LCST) or lower bound to a temperature range of partial miscibility. As shown in the diagram, for polymer solutions the LCST is higher than the UCST, so that there is a temperature interval of complete miscibility, with partial miscibility at both higher and lower temperatures.[5]
The UCST and LCST of polymer mixtures generally depend on polymer degree of polymerization and polydispersity.[6]
The seminal statistical mechanical model for the UCST of polymers is the Flory–Huggins solution theory.[7]
By adding soluble impurities the upper critical solution temperature increases and lower critical solution temperature decreases.[8]
See also
- Lower critical solution temperature – Critical temperature below which components of a mixture are miscible for all compositions
- Coil–globule transition – Collapse of a macromolecule from an expanded coil state to a collapsed globule state
References
- doi:10.1351/goldbook.UT07280. Retrieved 2012-10-18.)
{{cite journal}}: Cite journal requires|journal=(help - ^ a b Atkins, P.W.; de Paula, J.; Wong, Man. Atkins' Physical Chemistry 8e (2006).
- ^ OCLC 8112942.
- . Retrieved 2022-01-27.
- OCLC 756466890.
- ISSN 0024-9297.
- ISSN 2073-4360.
- ISSN 0021-9606.
