Step-growth polymerization
| Polymer science |
|---|
 |

In
Historical aspects
Most natural polymers being employed at early stage of human society are of condensation type. The synthesis of first truly synthetic polymeric material, bakelite, was announced by Leo Baekeland in 1907, through a typical step-growth polymerization fashion of phenol and formaldehyde. The pioneer of synthetic polymer science, Wallace Carothers, developed a new means of making polyesters through step-growth polymerization in 1930s as a research group leader at DuPont. It was the first reaction designed and carried out with the specific purpose of creating high molecular weight polymer molecules, as well as the first polymerization reaction whose results had been predicted by scientific theory. Carothers developed a series of mathematic equations to describe the behavior of step-growth polymerization systems which are still known as the Carothers equations today. Collaborating with Paul Flory, a physical chemist, they developed theories that describe more mathematical aspects of step-growth polymerization including kinetics, stoichiometry, and molecular weight distribution etc. Carothers is also well known for his invention of Nylon.
Condensation polymerization
"Step growth polymerization" and condensation polymerization are two different concepts, not always identical. In fact polyurethane polymerizes with addition polymerization (because its polymerization produces no small molecules), but its reaction mechanism corresponds to a step-growth polymerization.
The distinction between "addition polymerization" and "condensation polymerization" was introduced by Wallace Carothers in 1929, and refers to the type of products, respectively:[2][3]
- a polymer only (addition)
- a polymer and a molecule with a low molecular weight (condensation)
The distinction between "step-growth polymerization" and "chain-growth polymerization" was introduced by Paul Flory in 1953, and refers to the reaction mechanisms, respectively:[4]
- by functional groups (step-growth polymerization)
- by free-radical or ion (chain-growth polymerization)
Differences from chain-growth polymerization
This technique is usually compared with chain-growth polymerization to show its characteristics.
| Step-growth polymerization | Chain-growth polymerization | |
|---|---|---|
| Chain growth profile | Growth throughout matrix | Growth by addition of monomer only at one end or both ends of chain |
| Usage of monomer in the reaction | Rapid loss of monomer early in the reaction | Some monomer remains even at long reaction times |
| Reaction steps | Similar steps repeated throughout reaction process | Different steps operate at different stages of mechanism (i.e. initiation, propagation, termination, and chain transfer) |
| Average molecular weight | Average molecular weight increases slowly at low conversion and high extents of reaction are required to obtain high chain length | Molar mass of backbone chain increases rapidly at early stage and remains approximately the same throughout the polymerization |
| Active chain remains after reaction? | Ends remain active (no termination) | Chains not active after termination |
| Initiators required? | No initiator necessary | Initiator required |
Classes of step-growth polymers

Classes of step-growth polymers are:[6][7]
- Polyester has high glass transition temperature Tg and high melting point Tm, good mechanical properties to about 175 °C, good resistance to solvent and chemicals. It can exist as fibers and films. The former is used in garments, felts, tire cords, etc. The latter appears in magnetic recording tape and high grade films.
- Polyamide (nylon) has good balance of properties: high strength, good elasticity and abrasion resistance, good toughness, favorable solvent resistance. The applications of polyamide include: rope, belting, fiber clothes, thread, substitute for metal in bearings, jackets on electrical wire.
- elastomerswith good abrasion resistance, hardness, good resistance to grease and good elasticity, as fibers with excellent rebound, as coatings with good resistance to solvent attack and abrasion and as foams with good strength, good rebound and high impact strength.
- Polyurea shows high Tg, fair resistance to greases, oils, and solvents. It can be used in truck bed liners, bridge coating, caulk and decorative designs.
- rubbers. Due to perfect thermal stability (thanks to silicon, Si) uses of this material include antifoam and release agents, gaskets, seals, cable and wire insulation, hot liquids and gas conduits, etc.
- F-22 Raptoris made of high optical quality polycarbonate.
- Polysulfides have outstanding oil and solvent resistance, good gas impermeability, good resistance to aging and ozone. However, it smells bad, and it shows low tensile strength as well as poor heat resistance. It can be used in gasoline hoses, gaskets and places that require solvent resistance and gas resistance.
- Polyethershows good thermoplastic behavior, water solubility, generally good mechanical properties, moderate strength and stiffness. It is applied in sizing for cotton and synthetic fibers, stabilizers for adhesives, binders, and film formers in pharmaceuticals.
- Phenol formaldehyde resin (bakelite) have good heat resistance, dimensional stability as well as good resistance to most solvents. It also shows good dielectric properties. This material is typically used in molding applications, electrical, radio, televisions and automotive parts where their good dielectric properties are of use. Some other uses include: impregnating paper, varnishes, decorative laminates for wall coverings.
- Poly1,3-dipolar cycloaddition, also called the azide-alkyne Huisgen cycloaddition. These polymers can take on the form of a strong resin,[8] or a gel.[9] With oligopeptide monomers containing a terminal alkyne and terminal azide the resulting clicked peptide polymer will be biodegradable due to action of endopeptidases on the oligopeptide unit.[10]
Branched polymers
A monomer with functionality of 3 or more will introduce
Kinetics
The kinetics and rates of step-growth polymerization can be described using a polyesterification mechanism. The simple esterification is an acid-catalyzed process in which protonation of the acid is followed by interaction with the alcohol to produce an ester and water. However, there are a few assumptions needed with this kinetic model. The first assumption is water (or any other condensation product) is efficiently removed. Secondly, the functional group reactivities are independent of chain length. Finally, it is assumed that each step only involves one alcohol and one acid.
This is a general rate law degree of polymerization for polyesterification where n= reaction order.
Self-catalyzed polyesterification
If no acid catalyst is added, the reaction will still proceed because the acid can act as its own catalyst. The rate of condensation at any time t can then be derived from the rate of disappearance of -COOH groups and
The second-order term arises from its use as a catalyst, and k is the rate constant. For a system with equivalent quantities of acid and glycol, the functional group concentration can be written simply as
After integration and substitution from Carothers equation, the final form is the following
For a self-catalyzed system, the number average degree of polymerization (Xn) grows proportionally with .[11]
External catalyzed polyesterification
The uncatalyzed reaction is rather slow, and a high Xn is not readily attained. In the presence of a catalyst, there is an acceleration of the rate, and the kinetic expression is altered to[1]
which is kinetically first order in each functional group. Hence,
and integration gives finally
For an externally catalyzed system, the number average degree of polymerization grows proportionally with .
Molecular weight distribution in linear polymerization
The product of a polymerization is a mixture of polymer molecules of different molecular weights. For theoretical and practical reasons it is of interest to discuss the distribution of molecular weights in a polymerization. The
Probability
Step-growth polymerization is a random process so we can use statistics to calculate the probability of finding a chain with x-structural units ("x-mer") as a function of time or conversion.
Probability that an 'A' functional group has reacted
Probability of finding an 'A' unreacted
Combining the above two equations leads to.
Where Px is the probability of finding a chain that is x-units long and has an unreacted 'A'. As x increases the probability decreases.
Number fraction distribution

The number fraction distribution is the fraction of x-mers in any system and equals the probability of finding it in solution.
Where N is the total number of polymer molecules present in the reaction.[14]
Weight fraction distribution

The weight fraction distribution is the fraction of x-mers in a system and the probability of finding them in terms of mass fraction.[1]
Notes:
- Mo is the molar mass of the repeat unit,
- No is the initial number of monomer molecules,
- and N is the number of unreacted functional groups
Substituting from the Carothers equation
We can now obtain:
PDI
The
However, for step-growth polymerization the Carothers equation can be used to substitute and rearrange this formula into the following.
Therefore, in step-growth when p=1, then the PDI=2.
Molecular weight control in linear polymerization
Need for stoichiometric control
There are two important aspects with regard to the control of molecular weight in polymerization. In the synthesis of polymers, one is usually interested in obtaining a product of very specific molecular weight, since the properties of the polymer will usually be highly dependent on molecular weight. Molecular weights higher or lower than the desired weight are equally undesirable. Since the degree of polymerization is a function of reaction time, the desired molecular weight can be obtained by quenching the reaction at the appropriate time. However, the polymer obtained in this manner is unstable in that it leads to changes in molecular weight because the ends of the polymer molecule contain functional groups that can react further with each other.
This situation is avoided by adjusting the concentrations of the two monomers so that they are slightly nonstoichiometric. One of the reactants is present in slight excess. The polymerization then proceeds to a point at which one reactant is completely used up and all the chain ends possess the same functional group of the group that is in excess. Further polymerization is not possible, and the polymer is stable to subsequent molecular weight changes.
Another method of achieving the desired molecular weight is by addition of a small amount of monofunctional monomer, a monomer with only one functional group. The monofunctional monomer, often referred to as a chain stopper, controls and limits the polymerization of bifunctional monomers because the growing polymer yields chain ends devoid of functional groups and therefore incapable of further reaction.[13]
Quantitative aspects
To properly control the polymer molecular weight, the stoichiometric imbalance of the bifunctional monomer or the monofunctional monomer must be precisely adjusted. If the nonstoichiometric imbalance is too large, the polymer molecular weight will be too low. It is important to understand the quantitative effect of the stoichiometric imbalance of reactants on the molecular weight. Also, this is necessary in order to know the quantitative effect of any reactive impurities that may be present in the reaction mixture either initially or that are formed by undesirable side reactions. Impurities with A or B functional groups may drastically lower the polymer molecular weight unless their presence is quantitatively taken into account.[13]
More usefully, a precisely controlled stoichiometric imbalance of the reactants in the mixture can provide the desired result. For example, an excess of diamine over an acid chloride would eventually produce a polyamide with two amine end groups incapable of further growth when the acid chloride was totally consumed. This can be expressed in an extension of the Carothers equation as,
where r is the ratio of the number of molecules of the reactants.
- were NBB is the molecule in excess.
The equation above can also be used for a monofunctional additive which is the following,
where NB is the number of monofunction molecules added. The coefficient of 2 in front of NB is require since one B molecule has the same quantitative effect as one excess B-B molecule.[15]
Multi-chain polymerization
A monomer with functionality 3 has 3 functional groups which participate in the polymerization. This will introduce
A more general functionality factor fav is defined for multi-chain polymerization, as the average number of functional groups present per monomer unit. For a system containing N0 molecules initially and equivalent numbers of two function groups A and B, the total number of functional groups is N0fav.
And the modified Carothers equation is[16]
- , where p equals to
Advances in step-growth polymers
The driving force in designing new polymers is the prospect of replacing other materials of construction, especially metals, by using lightweight and heat-resistant polymers. The advantages of lightweight polymers include: high strength, solvent and chemical resistance, contributing to a variety of potential uses, such as electrical and engine parts on automotive and aircraft components, coatings on cookware, coating and circuit boards for electronic and microelectronic devices, etc. Polymer chains based on aromatic rings are desirable due to high bond strengths and rigid polymer chains. High molecular weight and crosslinking are desirable for the same reason. Strong dipole-dipole, hydrogen bond interactions and crystallinity also improve heat resistance. To obtain desired mechanical strength, sufficiently high molecular weights are necessary, however, decreased solubility is a problem. One approach to solve this problem is to introduce of some flexibilizing linkages, such as isopropylidene, C=O, and SO
2 into the rigid polymer chain by using an appropriate monomer or comonomer. Another approach involves the synthesis of reactive telechelic oligomers containing functional end groups capable of reacting with each other, polymerization of the oligomer gives higher molecular weight, referred to as chain extension.[17]
Aromatic polyether

The oxidative coupling polymerization of many 2,6-disubstituted
Polyethersulfone

Polyethersulfone (PES) is also referred to as polyetherketone, polysulfone. It is synthesized by nucleophilic aromatic substitution between aromatic dihalides and bisphenolate salts. Polyethersulfones are partially crystalline, highly resistant to a wide range of aqueous and organic environment. They are rated for continuous service at temperatures of 240-280 °C. The polyketones are finding applications in areas like automotive, aerospace, electrical-electronic cable insulation.
Aromatic polysulfides
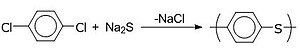
Poly(p-phenylene sulfide) (PPS) is synthesized by the reaction of
Aromatic polyimide

Aromatic polyimides are synthesized by the reaction of dianhydrides with diamines, for example, pyromellitic anhydride with
Telechelic oligomer approach
See also
- Conducting polymer
- Fire-safe polymers
- Liquid crystal polymer
- Random graph theory of gelation
- Thermosetting plastic
References
- ^ a b c Cowie JM, Arrighi V (2008). Polymers: Chemistry and Physics of Modern Materials (3rd ed.). CRC Press.
- .
- ISBN 0-8014-0134-8.
- ISBN 1-56990-372-7.
- PMID 30783151.
- ISBN 978-0-8247-8719-6.
- ^ Mark HF, Bikales NM, Overberger CG, Menges G (1988). Encyclopedia of Polymer Science and Engineering. New York: Wiley-Interscience.
- .
- .
- PMID 18817441.
- ISBN 978-0-471-72543-5.
- ISBN 9780801401343.
- ^ ISBN 978-0-471-61020-5.
- .
- ISBN 978-0195057591.
- .
- ^ Rogers ME, Long TE, Turners SR. Synthetic methods in step-growth polymers. Wiley-Interscience.
- ISBN 978-0-19-850389-7.

![{\displaystyle {\frac {1}{1-p^{n-1}}}=1+(n-1)kt[{\ce {COOH}}]^{n-1}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f40d7c3649f67e96e10edcd26e6fab23275516ae)
![{\displaystyle rate={\frac {-d[{\ce {COOH}}]}{dt}}=k[{\ce {COOH}}]^{2}[{\ce {OH}}]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e36e9f9e72374e37c029a720efe9cb5e244de56e)
![{\displaystyle [{\ce {COOH}}]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/de1374de9d6397122318e53d73c974969f07bdfc)
![{\displaystyle rate={\frac {-d[{\ce {COOH}}]}{dt}}=k[{\ce {COOH}}]^{3}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/c02467b42290958b886e503d42cab7b3882e6b9d)
![{\displaystyle {\frac {1}{(1-p)^{2}}}=2kt[{\ce {COOH}}]^{2}+1=X_{n}^{2}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/11bd2e3f368961bfa8f4002c38928e4755ed2cba)

![{\displaystyle {\frac {-d[{\ce {COOH}}]}{dt}}=k[{\ce {COOH}}][{\ce {OH}}]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/2fa3c69f4aa3a4c9646018d505e9e189a22e56ab)
![{\displaystyle {\frac {-d[{\ce {COOH}}]}{dt}}=k[{\ce {COOH}}]^{2}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/40623a5a6ecd32d96f118b41e932c376ed8c47f2)
![{\displaystyle {\frac {1}{1-p}}=1+[{\ce {COOH}}]kt=X_{n}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8addf76f8424ddca5c944ab775c0d7186bf58703)

















