Arsenous acid

| |
| |
| Names | |
|---|---|
| IUPAC name
Arsorous acid
| |
| Other names
Arsenious acid
Arsenic oxide | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| H3AsO3 | |
| Molar mass | 125.94 g/mol |
| Appearance | Only exists in aqueous solutions |
Conjugate base
|
Arsenite |
| -51.2·10−6 cm3/mol | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, corrosive |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
[1910.1018] TWA 0.010 mg/m3[1] |
REL (Recommended)
|
Ca C 0.002 mg/m3 [15-minute][1] |
IDLH (Immediate danger) |
Ca [5 mg/m3 (as As)][1] |
| Related compounds | |
Related compounds
|
Arsenic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
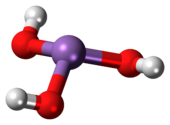
Arsenous acid (or arsenious acid) is the inorganic compound with the formula H3AsO3. It is known to occur in aqueous solutions, but it has not been isolated as a pure material, although this fact does not detract from the significance of As(OH)3.[2]
Properties

As(OH)3 is a pyramidal molecule consisting of three
One tautomer of arsenous acid is HAsO(OH)2, which is called arsonic acid. It has not been isolated or well-characterized.
Synthesis
The preparation of As(OH)3 involves a slow hydrolysis of arsenic trioxide in water. Addition of base converts arsenous acid to the arsenite ions [AsO(OH)2]−, [AsO2(OH)]2−, and [AsO3]3−.
Reactions
With its first pKa being 9.2, As(OH)3 is a weak acid.[4] Reactions attributed to aqueous arsenic trioxide are due to arsenous acid and its conjugate bases.
Like arsenic trioxide, arsenous acid is sometimes
- As(OH)3 + 3 HCl ⇌ AsCl3 + 3 H2O
- As(OH)3 + 3 HBr ⇌ AsBr3 + 3 H2O
- As(OH)3 + 3 HI ⇌ AsI3 + 3 H2O
Reaction of arsenous acid with
- As(OH)3 + CH3I + NaOH ⇌ CH3AsO(OH)2 + NaI + H2O
Alkylation occurs at arsenic, and the oxidation state of arsenic increases from +3 to +5.
Toxicology
Arsenic-containing compounds are highly toxic and
References
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0038". National Institute for Occupational Safety and Health (NIOSH).
- ^ Munoz-Hernandez, M.-A. (1994). "Arsenic: Inorganic Chemistry". In King, R. B. (ed.). Encyclopedia of Inorganic Chemistry. Chichester: John Wiley & Sons.
- PMID 18380458. Archived from the original(PDF) on 2012-04-25. Retrieved 2011-12-18.
- ^ ISBN 978-0-08-037941-8.
- .
