Hydrogen selenide
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydrogen selenide
| |||
| Other names
Hydroselenic acid
selane selenium hydride | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.029.071 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 2202 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| H2Se | |||
| Molar mass | 80.98 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | decayed horseradish[1] | ||
| Density | 3.553 g/dm3 | ||
| Melting point | −65.73 °C (−86.31 °F; 207.42 K) | ||
| Boiling point | −41.25 °C (−42.25 °F; 231.90 K) | ||
| 0.70 g/100 mL | |||
| Solubility | soluble in CS2, phosgene | ||
| Vapor pressure | 9.5 atm (21°C)[1] | ||
| Acidity (pKa) | 3.89 | ||
Conjugate acid
|
Selenonium | ||
Conjugate base
|
Selenide | ||
| Structure | |||
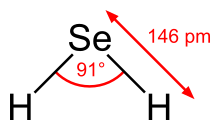
| Bent | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Extremely toxic and flammable | ||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H220, H280, H330, H410 | |||
| P210, P260, P271, P273, P284, P304+P340, P310, P320, P377, P381, P391, P403, P403+P233, P405, P410+P403, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | flammable gas | ||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
0.3 ppm (guinea pig, 8 hr) 5.9 ppm (rat, 1 hr)[2] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 0.05 ppm (0.2 mg/m3)[1] | ||
REL (Recommended)
|
TWA 0.05 ppm (0.2 mg/m3)[1] | ||
IDLH (Immediate danger) |
1 ppm[1] | ||
| Safety data sheet (SDS) | ICSC 0284 | ||
| Related compounds | |||
Other anions
|
H2Po
| ||
Other cations
|
Na2Se Ag2Se | ||
Related compounds
|
Arsine | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrogen selenide is an
Even at extremely low concentrations, this compound has a very irritating smell resembling that of decayed horseradish or "leaking gas", but smells of rotten eggs at higher concentrations.Structure and properties
H2Se adopts a bent structure with a H−Se−H bond angle of 91°[citation needed]. Consistent with this structure, three IR-active vibrational bands are observed: 2358, 2345, and 1034 cm−1.[6]
The properties of H2S and H2Se are similar, although the selenide is more acidic with pKa = 3.89 and the second pKa = 11,[6] or 15.05 ± 0.02 at 25 °C.[7]
Preparation
Industrially, it is produced by treating elemental selenium at T > 300 °C with hydrogen gas.
- Al2Se3 + 6 H2O ⇌ 2 Al(OH)3 + 3 H2Se
H2Se can also be prepared by means of different methods based on the in situ generation in aqueous solution using
Reactions
Elemental selenium can be recovered from H2Se through a reaction with aqueous sulfur dioxide (SO2).
- 2 H2Se + SO2 ⇌ 2 H2O + 2 Se + S
Its decomposition is used to prepare the highly pure element.
Applications
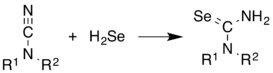
H2Se is commonly used in the synthesis of Se-containing compounds. It adds across alkenes. Illustrative is the synthesis of
H2Se gas is used to dope semiconductors with selenium.
Safety
Hydrogen selenide is hazardous, being the most toxic selenium compound[3] and far more toxic than its congener hydrogen sulfide. The threshold limit value is 0.05 ppm. The gas acts as an irritant at concentrations higher than 0.3 ppm, which is the main warning sign of exposure; below 1 ppm, this is "insufficient to prevent exposure", while at 1.5 ppm the irritation is "intolerable".[5] Exposure at high concentrations, even for less than a minute, causes the gas to attack the eyes and mucous membranes; this causes cold-like symptoms for at least a few days afterwards. In Germany, the limit in drinking water is 0.008 mg/L, and the US EPA recommends a maximum contamination of 0.01 mg/L.[8][12]
Despite being extremely toxic, no human fatalities have yet been reported. It is suspected that this is due to the gas' tendency to oxidise to form red selenium in mucous membranes; elemental selenium is less toxic than selenides are.[4]
See also
References
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0336". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Hydrogen selenide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b http://www.epa.gov/ttnatw01/hlthef/selenium.html, US Environmental Protection Agency, Air Toxins website
- ^ a b "CDC - Immediately Dangerous to Life or Health Concentrations (IDLH): Hydrogen selenide (as Se) - NIOSH Publications and Products". www.cdc.gov. 2018-11-02. Retrieved 2023-01-09.
- ^ a b https://www.cdc.gov/niosh/docs/81-123/pdfs/0336.pdf Occupational Health Guideline for Hydrogen Selenide, The National Institute for Occupational Safety and Health, 1978
- ^ )
- .
- ^ .
- ^ Féher, F. In "Handbook of Preparative Inorganic Chemistry"; Brauer, E., Ed.; Academic: New York, 1963; 1, p 418.
- ^ Sonoda, N.; Kondo K.; Nagano, K.; Kambe, N.; Morimoto, F. Angewandte Chemie International Edition English 1980, vol. 19, page 308
- S2CID 96314420.
- ^ https://www.osha.gov/dts/chemicalsampling/data/CH_246700.html Archived 2017-05-02 at the Wayback Machine, OSHA GENERAL INDUSTRY PEL: 0.05 ppm, 0.2 mg/m3 ,OSHA CONSTRUCTION INDUSTRY PEL: 0.05 ppm, 0.2 mg/m3 TWA