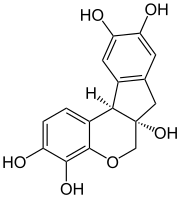
Haematoxylin

| |
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
(6aS,11bR)-7,11b-Dihydroindolo[2,1-c] [1]benzopyran-3,4,6a,9,10(6H)-pentol | |
| Other names
Hematoxylin; Natural Black 1; Hematoxyline; Hydroxybrazilin; Hydroxybrasilin; C.I. 75290
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.007.490 |
| MeSH | Hematoxylin |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H14O6 | |
| Molar mass | 302.282 g·mol−1 |
| Melting point | 100-120 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Haematoxylin
Although the stain is commonly called haematoxylin, the active colourant is the
Extraction and purification

Haematoxylin has been synthesized,[19][20] although never in commercially viable amounts.[14][21] Historically the logwood was exported and the haematoxylin extracted in Europe. More recently extraction takes place closer to where the logwood is harvested.[18] Extraction of haematoxylin from logwood on industrial scales has been accomplished in the 'French process' by boiling the wood chips or in the 'American process' with steam and pressure.[9][22] Once extracted, the dye can be sold as a liquid concentrate or dried and sold in a crystalline form.[9] Modern production methods use water, ether or alcohol as a solvent, at which point the extracts may be further refined to the level of purity needed.[18]
The commercial product may vary from batch to batch and between manufacturers[18] in both the level of impurities and in the ratio of haematoxylin to haematein.[23][2][24] For histologic use, this variability can affect the stains interaction with biological tissue samples, and is therefore of concern to histologists and pathologists.[23][2][18] Haematoxylin, like other biological stains, may be certified by the Biological Stain Commission, signifying that a particular batch of stain works in a standardized test, although this does not specify the dye's actual purity.[23]
Use as a histologic stain

Haematoxylin stain is commonly followed (or counterstained) with another histologic stain,
Principally used as a
Stain formulations

Haematoxylin stain formulations can be broadly classified based on how the haematoxylin is oxidized (or ripened) and by choice of the mordant used.[1] Haematoxylin stain formulations may either be natural oxidized by exposure to air and sunlight, or more commonly, especially in commercially prepared solutions,[7] chemically oxidized using sodium iodate.[1][26][11] Commonly only enough oxidizer is added to convert one half of the haematoxylin to haematein, allowing the remainder to naturally oxidize during use, this extends the staining solution's useful life as more haematein is produced, while some haematein is further oxidized to oxyhaematein.[13][27][11] Of the metallic salts used as mordants, aluminium is the most common,[11] other mordants include salts of iron, tungsten, molybdenum and lead.[1]
Depending on the formulation or staining technique, haematoxylin stains may be used in what is called a progressive manner, in which the length of time the tissue remains in contact with the staining solution is used to control the amount of colouration, or in a regressive manner, in which the tissue is over-stained, and excess stain is removed in a secondary step of the procedure.[11][25][1] Removal of unwanted staining, or differentiation, typically involves a solution of diluted ethanol and hydrochloric acid.[11][1][20]
Table of significant formulations
| Formula name | Reference | Mordant | Oxidation method | Typical use |
|---|---|---|---|---|
| Ehrlich's Haematoxylin[26] | Ehrlich, 1886 | Potassium alum | Natural | Nuclear stain in H&E
|
| Delafield's Haematoxylin[26] | Prudden, 1855 | Ammonium alum | Natural | Nuclear stain in H&E |
| Mayer's Haematoxylin[26] | Mayer, 1903 | Potassium or Ammonium alum | Sodium iodate | Nuclear stain in H&E |
| Harris's Haematoxylin[26] | Harris, 1900[28] | Potassium alum | Mercuric oxide |
Nuclear stain in H&E, also used in the classical versions of the Papanicolaou stain[29] |
| Cole's Haematoxylin[1] | Cole, 1943[30] | Potassium alum | Iodine | Nuclear stain in H&E |
| Carazzi's Haematoxylin[1] | Carazzi, 1911 | Potassium alum | Potassium iodate | Nuclear stain in H&E, urgent biopsy sections |
| Weigert's Haematoxylin[26] | Weigert, 1904 | Ferric chloride |
Natural | Nuclear stain in H&E, resistant to acids |
| Verhoeff's Haematoxylin[1] | Verhoeff, 1908 | Ferric chloride | Iodine | |
phosphotungstic acid Haematoxylin[1] |
Mallory, 1897 | Phosphotungstic acid | Natural or chemical | Fibrin, muscle striations |
| Gill's Haematoxylin (I, II, and III) | Culling et al. 1985 [11][27] | Aluminium sulfate | Sodium iodate | Nuclear stain in H&E |
Early use as a histologic stain
In 1758,
Shortages and possible alternatives
During
Use as a textile dye
Haematoxylin was first used as a dye by the
Use as a writing and drawing ink
Haematoxylin has been used as the primary component of writing and drawing
See also
- Staining (biology)
- Histology
- Eosin
- H&E stain
- Haematein
- Verhoeff's stain
- Papanicolaou stain
- Natural dye
- Haematoxylum campechianum
Further reading
- Jocelyn H. Bruce-Gregorios, M.D.: Histopathologic Techniques, JMC Press Inc., Quezon City, Philippines, 1974.
- Meloan, S. M. & Puchtler, H. 1987. "Harris hematoxylin," what Harris really wrote and the mechanism of hemalum stains. Journal of Histotechnology 10: 257–261.
- Puchtler, H., Meloan, S.N. & Waldrop, F.S. 1986. Application of current chemical concepts to metal-haematein and -brazilein stains. Histochemistry 85: 353–364.
- Stainsfile
References
- ^ a b c d e f g h i j k l m n o p q r s Stevens, Alan (1982). "The Haematoxylins". In Bancroft, John; Stevens, Alan (eds.). The Theory and Practice of Histological Techniques (2nd ed.). Longman Group Limited. p. 109.
- ^ a b c d Lillie, Ralph Dougall (1977). H. J. Conn's Biological stains (9th ed.). Baltimore: Williams & Wilkins. pp. 692p.
- .
- ^ ISSN 0360-9081.
- ^ ISSN 0377-0486.
- ^ ISBN 9780300191875.
- ^ S2CID 20338201.
- ^ PMID 30001639.
- ^ ISSN 2499-8281.
- ^ S2CID 26847314.
- ^ S2CID 205713596.
- ^ PMID 16761865.
- ^ ISSN 1472-3581.
- ^ S2CID 28563610.
- ^ S2CID 26801839.
- doi:10.1002/chir.12.
- S2CID 23504301.
- ^ S2CID 7698557.
- .
- ^ S2CID 7384777.
- ^ S2CID 5297820.
- ^ PMID 4139897.
- ^ S2CID 29628388.
- PMID 4135791.
- ^ S2CID 13481905.
- ^ a b c d e f g Gatenby, J. B.; Beams, H. W. (1950). The Microtomist's Vade-Mecum (11th ed.). Philadelphia: The Blackstone Company.
- ^ S2CID 207513639.
- ^ Harris, H. F. (1900). "On the rapid conversion of haematoxylin into haematein in staining reactions". Journal of Applied Microscopic Laboratory Methods. 3 (3): 777.
- ISSN 1574-9053.
- ISSN 0038-9153.
- ^ ISBN 978-0940095007.
- ^ PMID 10605407.
- ^ Quekett, John Thomas (1848). A Practical treatise on the use of the microscope. Library of illustrated standard scientific works. Vol. VI. Paris: Hippolyte Bailliere.
- ^ a b c Mann, Gustav (1902). Physiological Histology, Methods and Theory. Clarendon Press. p. 488.
- ^ PMID 9389971.
- ^ Georg von Georgievics (1902). The Chemical Technology of Textile Fibres: Their Origin, Structure, Preparation, Washing, Bleaching, Dyeing, Printing and Dressing. Scott, Greenwood & Co. p. 180.
- ^ ISSN 0377-0486.

