Hexamethylenetetramine
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,3,5,7-Tetraazaadamantane | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 2018 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.002.642 | ||
| EC Number |
| ||
| E number | E239 (preservatives) | ||
| 26964 | |||
| KEGG | |||
| MeSH | Methenamine | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1328 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H12N4 | |||
| Molar mass | 140.186 g/mol | ||
| Appearance | White crystalline solid | ||
| Odor | Fishy, ammonia like | ||
| Density | 1.33 g/cm3 (at 20 °C) | ||
| Melting point | 280 °C (536 °F; 553 K) ( sublimes )
| ||
| 85.3 g/100 mL | |||
| Solubility | Soluble in chloroform, methanol, ethanol, acetone, benzene, xylene, ether | ||
| Solubility in chloroform | 13.4 g/100 g (20 °C) | ||
| Solubility in methanol | 7.25 g/100 g (20 °C) | ||
| Solubility in ethanol | 2.89 g/100 g (20 °C) | ||
| Solubility in acetone | 0.65 g/100 g (20 °C) | ||
| Solubility in benzene | 0.23 g/100 g (20 °C) | ||
| Acidity (pKa) | 4.89[1] | ||
| Pharmacology | |||
| J01XX05 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Highly combustible, harmful | ||
| GHS labelling: | |||
 
| |||
| Warning | |||
| H228, H317 | |||
| P210, P240, P241, P261, P272, P280, P302+P352, P321, P333+P313, P363, P370+P378, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 250 °C (482 °F; 523 K) | ||
| 410 °C (770 °F; 683 K) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hexamethylenetetramine, also known as methenamine, hexamine, or its trade name Urotropin, is a
Synthesis, structure, reactivity
Hexamethylenetetramine was discovered by
It is prepared industrially by combining formaldehyde and ammonia:[4]The reaction can be conducted in gas phase and in solution.
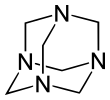
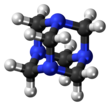
The molecule has a tetrahedral cage-like structure, similar to adamantane. Four vertices are occupied by nitrogen atoms, which are linked by methylene groups. Although the molecular shape defines a cage, no void space is available at the interior for binding other atoms or molecules,[citation needed] unlike crown ethers or larger cryptand structures.
The molecule behaves like an amine base, undergoing protonation and N-alkylation (e.g. quaternium-15[clarify]).
Applications
The dominant use of hexamethylenetetramine is in the production of solid (powder) or liquid
Medical uses
As the mandelic acid salt (methenamine mandelate) or the hippuric acid salt (methenamine hippurate),[5] it is used for the treatment of urinary tract infections. In an acidic environment, methenamine is believed to act as an antimicrobial by converting to formaldehyde.[5][6] A systematic review of its use for this purpose in adult women found there was insufficient evidence of benefit and further research was needed.[7] A UK study showed that methenamine is as effective as daily low-dose antibiotics at preventing UTIs among women who experience recurrent UTIs. As methenamine is an antiseptic, it may avoid the issue of antibiotic resistance.[8][9]
Methenamine acts as an over-the-counter
Histological stains
, including the following types:- organisms.
- Periodic acid-Schiff that stains for basement membrane, availing to view the "spiked" Glomerular basement membrane associated with membranous glomerulonephritis.
Solid fuel
Together with
Standardized 0.149 g tablets of methenamine (hexamine) are used by fire-protection laboratories as a clean and reproducible fire source to test the flammability of carpets and rugs.[12]
Food additive
Hexamethylenetetramine or hexamine is also used as a
Reagent in organic chemistry
Hexamethylenetetramine is a versatile
Explosives
Hexamethylenetetramine is the base component to produce RDX and, consequently, C-4[4] as well as octogen (a co-product with RDX), hexamine dinitrate, hexamine diperchlorate and HMTD.
Pyrotechnics
Hexamethylenetetramine is also used in
Historical uses
Hexamethylenetetramine was first introduced into the medical setting in 1895 as a urinary antiseptic.[22] It was officially approved by the FDA for medical use in the United States in 1967.[23] However, it was only used in cases of acidic urine, whereas boric acid was used to treat urinary tract infections with alkaline urine.[24] Scientist De Eds found that there was a direct correlation between the acidity of hexamethylenetetramine's environment and the rate of its decomposition.[25] Therefore, its effectiveness as a drug depended greatly on the acidity of the urine rather than the amount of the drug administered.[24] In an alkaline environment, hexamethylenetetramine was found to be almost completely inactive.[24]
Hexamethylenetetramine was also used as a method of treatment for soldiers exposed to phosgene in World War I. Subsequent studies have shown that large doses of hexamethylenetetramine provide some protection if taken before phosgene exposure but none if taken afterwards.[26]

Producers
Since 1990 the number of European producers has been declining. The French SNPE factory closed in 1990; in 1993, the production of hexamethylenetetramine in Leuna, Germany ceased; in 1996, the Italian facility of Agrolinz closed down; in 2001, the UK producer Borden closed; in 2006, production at Chemko, Slovak Republic, was closed. Remaining producers include INEOS in Germany, Caldic in the Netherlands, and Hexion in Italy. In the US, Eli Lilly and Company stopped producing methenamine tablets in 2002.[12] In Australia, Hexamine Tablets for fuel are made by Thales Australia Ltd. In México, Hexamine is produced by Abiya.[citation needed]
References
- .
- . In this article, Butlerov discovered formaldehyde, which he called "dioxymethylen" (methylene dioxide) [page 247] because his empirical formula for it was incorrect (C4H4O4). On pages 249–250, he describes treating formaldehyde with ammonia gas, creating hexamine.
- .
- ^ ISBN 9783527306732.
- ^ S2CID 207199202.
- PMID 31579504.
- PMID 34001538.
- S2CID 254965605.
- PMID 35264408.
- S2CID 24088433.
- S2CID 24088433.
- ^ a b "Re: Equialence of methenamine Tablets Standard for Flammability of Carpets and Rugs" (PDF). Alan H. Schoen. July 29, 2004. Archived from the original (PDF) on 2008-10-05. Many other countries who still produce this include Russia, Saudi Arabia, China and Australia.
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 - Labelling of ingredients". Retrieved 2011-10-27.
- S2CID 97756228.
- .
- ; Collected Volumes, vol. 3, p. 811.
- .
- ^ DE patent 3402546, Lohmann, Erwin, "Pyrotechnischer Satz zur Erzeugung von Lichtblitzen", issued 1985-08-01
- ^ US patent 6214139, Hiskey, Michael A.; Chavez, David E. & Naud, Darren L., "Low-smoke pyrotechnic compositions", issued 2001-04-10, assigned to The Regents of the University of California and Los Alamos National Laboratory
- ^ GB patent 2502460, Wraige, John Douglas Michael, "Pyrotechnic composition with spark producing material", issued 2013-11-27
- .
- ISBN 978-0-323-40181-4, retrieved 2023-11-15
- ^ a b c Elliot (1913). "On Urinary Antiseptics". British Medical Journal. 98: 685–686.
- ISSN 0007-1331.
- S2CID 2423812.