Lead(II) nitrate

| |||
| |||
| Identifiers | |||
|---|---|---|---|
3D model (
JSmol ) |
|||
| ChemSpider | |||
ECHA InfoCard
|
100.030.210 | ||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1469 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| Pb(NO3)2 | |||
| Molar mass | 331.2 g/mol | ||
| Appearance | colorless or white | ||
| Density | 4.53 g/cm3 | ||
| Melting point | 470 °C (878 °F; 743 K)[2] decomposes | ||
| 376.5 g/L (0 °C) 597 g/L (25°C) 1270 g/L (100°C) | |||
| −74·10−6 cm3/mol[1] | |||
Refractive index (nD)
|
1.782[2] | ||
| Thermochemistry | |||
Std enthalpy of (ΔfH⦵298)formation |
−451.9 kJ·mol−1[1] | ||
| Hazards | |||
| GHS labelling:[4] | |||
   
| |||
| Danger | |||
| H302, H317, H318, H332, H360, H373, H410 | |||
| P201, P202, P210, P220, P221, P260, P261, P264, P270, P271, P272, P273, P280, P281, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P308+P313, P310, P312, P314, P321, P330, P333+P313, P363, P370+P378, P391, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LDLo (lowest published)
|
500 mg/kg (guinea pig, oral)[3] | ||
| Safety data sheet (SDS) | ICSC 1000 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Lead(II) nitrate is an
Known since the
Lead(II) nitrate is toxic and must be handled with care to prevent inhalation, ingestion and skin contact. Due to its hazardous nature, the limited applications of lead(II) nitrate are under constant scrutiny.
History
Lead nitrate was first identified in 1597 by the
Production
Lead nitrate is produced by reaction of lead(II) oxide with concentrated nitric acid:[10]
- PbO + 2 HNO3(concentrated) → Pb(NO3)2↓ + H2O
It may also be obtained evaporation of the solution obtained by reacting metallic lead with dilute nitric acid.[11]
- Pb + 4 HNO3 → Pb(NO3)2 + 2 NO2 + 2 H2O
Solutions and crystals of lead(II) nitrate are formed in the processing of lead–bismuth wastes from lead refineries.[12]
Structure


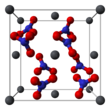
The crystal structure of solid lead(II) nitrate has been determined by neutron diffraction.[13][14] The compound crystallizes in the cubic system with the lead atoms in a face-centred cubic system. Its space group is Pa3Z=4 (Bravais lattice notation), with each side of the cube with length 784 picometres.
The black dots represent the lead atoms, the white dots the nitrate groups 27 picometres above the plane of the lead atoms, and the blue dots the nitrate groups the same distance below this plane. In this configuration, every lead atom is bonded to twelve oxygen atoms (bond length: 281 pm). All N–O bond lengths are identical, at 127 picometres.[15]
Research interest in the crystal structure of lead(II) nitrate was partly based on the possibility of free internal rotation of the nitrate groups within the crystal lattice at elevated temperatures, but this did not materialise.[14]
Chemical properties and reactions
Graphs are unavailable due to technical issues. There is more info on Phabricator and on MediaWiki.org. |
Lead nitrate decomposes on heating, a property that has been used in pyrotechnics .[9] It is soluble in water and dilute nitric acid.
Basic nitrates are formed in when alkali is added to a solution. Pb2(OH)2(NO3)2 is the predominant species formed at low pH. At higher pH Pb6(OH)5(NO3) is formed.[17] The cation [Pb6O(OH)6]4+ is unusual in having an oxide ion inside a cluster of 3 face-sharing PbO4 tetrahedra.[18] There is no evidence for the formation of the hydroxide, Pb(OH)2, in aqueous solution below pH 12.
Solutions of lead nitrate can be used to form co-ordination complexes. Lead(II) is a
The complex formed by lead nitrate with a
Applications
Lead nitrate has been used as a heat stabiliser in nylon and polyesters, as a coating for photothermographic paper, and in rodenticides.[10]
Heating lead nitrate is convenient means of making nitrogen dioxide
In the gold cyanidation process, addition of lead(II) nitrate solution improves the leaching process. Only limited amounts (10 to 100 milligrams lead nitrate per kilogram gold) are required.[22][23]
In organic chemistry, it may be used in the preparation of isothiocyanates from dithiocarbamates.[24] Its use as a bromide scavenger during SN1 substitution has been reported.[25]
Safety
Lead(II) nitrate is toxic, and ingestion may lead to acute lead poisoning, as is applicable for all soluble lead compounds.
References
- ^ )
- ^ OCLC 50252041.
- ^ "Lead compounds (as Pb)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "Lead nitrate". pubchem.ncbi.nlm.nih.gov. Retrieved 19 December 2021.
- ^ Libavius, Andreas (1595). Alchemia Andreæ Libavii. Francofurti: Iohannes Saurius.
- ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 16 (11th ed.). Cambridge University Press. pp. 314–320.
- ISBN 0-665-51791-2.
- ^ Partington, James Riddick (1950). A Text-book of Inorganic Chemistry. MacMillan. p. 838.
- ^ a b Barkley, J. B. (October 1978). "Lead nitrate as an oxidizer in blackpowder". Pyrotechnica. 4. Post Falls, Idaho: Pyrotechnica Publications: 16–18.
- ^ ISBN 0-7506-3365-4.
- ISBN 0-471-02040-0.
- ^ "Product catalog; other products". Tilly, Belgium: Sidech. Archived from the original on 2007-07-01. Retrieved 2008-01-05.
- .
- ^ .
- . Retrieved 15 July 2019.
- .
- .
- ISBN 978-0-08-037941-8. p. 395
- PMID 11666874.
- .
- doi:10.1039/b207568g.
- ^ Habashi, Fathi (1998). "Recent advances in gold metallurgy". Revisa de la Facultad de Ingeniera, Universidad Central de Venezuela. 13 (2): 43–54.
- ^ "Auxiliary agents in gold cyanidation". Gold Prospecting and Gold Mining. Retrieved 2008-01-05.
- ^ Dains, F. B.; Brewster, R. Q.; Olander, C. P. "Phenyl isothiocyanate". Organic Syntheses; Collected Volumes, vol. 1, p. 447.
- ^ Rapoport, H.; Jamison, T. (1998). "(S)-N-(9-Phenylfluoren-9-yl)alanine and (S)-Dimethyl-N-(9-phenylfluoren-9-yl)aspartate". Organic Syntheses; Collected Volumes, vol. 9, p. 344.
- ^ "Lead nitrate, Chemical Safety Card 1000". International Labour Organization, International Occupational Safety and Health Information Centre. March 1999. Retrieved 2008-01-19.
- ^ "Inorganic and Organic Lead Compounds" (PDF). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Suppl. 7. International Agency for Research on Cancer: 239. 1987. Archived from the original (PDF) on 2008-03-06. Retrieved 2008-01-19.
- ISBN 92-832-1287-8. Archived from the original(PDF) on 2007-10-21. Retrieved 2008-01-01.
- S2CID 40092031.
External links
- Woodbury, William D. (1982). "Lead". Mineral Yearbook Metals and Minerals. Bureau of Mines: 515–42. Retrieved 2008-01-18.
- "Lead". NIOSH Pocket Guide to Chemical Hazards. National Institute for Occupational Safety and Health. September 2005. NIOSH 2005-149. Retrieved 2008-01-19.
- "Lead and Lead Compounds Fact Sheet". National Pollutant Inventory. Australian Government, Department of the Environment and Water Resources. July 2007. Archived from the original on January 11, 2008. Retrieved 2008-01-19.
- "Lead". A Healthy Home Environment, Health Hazards. US Alliance for healthy homes. Archived from the original on 2008-02-20. Retrieved 2008-01-19.
- Material Safety Data Sheets



![{\displaystyle {\ce {2 Pb(NO_3)_2->[\Delta]2PbO + 4NO_2 +O_2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/06f64696eaa883432008d7e6af4c5af9b60a6b85)