Lead compounds

Compounds of lead exist with



Chemistry
Various oxidized forms of lead are easily reduced to the metal. An example is heating PbO with mild organic reducing agents such as glucose. The mixture of the oxide and the sulfide heated together will also form the metal.[2]
- 2 PbO + PbS → 3 Pb + SO2
Metallic lead is attacked (oxidized) only superficially by air, forming a thin layer of lead oxide that protects it from further oxidation. The metal is not attacked by sulfuric or hydrochloric acids. It dissolves in nitric acid with the evolution of nitric oxide gas to form dissolved Pb(NO3)2.
- 3 Pb + 8 H+ + 8 NO−
3 → 3 Pb2+ + 6 NO−
3 + 2 NO + 4 H2O
When heated with
3(OH)
2(CO
3)
2) salts of lead. The sulfide can also be precipitated from acetate solutions. These salts are all poorly soluble in water. Among the halides, the iodide is less soluble than the bromide, which, in turn, is less soluble than the chloride.[3]
Lead(II) oxide is also soluble in alkali hydroxide solutions to form the corresponding plumbite salt.[2]
- PbO + 2 OH− + H2O → Pb(OH)2−
4
Chlorination of plumbite solutions causes the formation of lead's +4 oxidation state.
- Pb(OH)2−
4 + Cl2 → PbO2 + 2 Cl− + 2 H2O
- PbO2 + 2 OH− + 2 H2O → Pb(OH)2−
6
Lead also has an oxide with mixed +2 and +4 oxidation states,
3O
4), also known as minium.
Lead readily forms an equimolar alloy with
Oxides and sulfide
There are three oxides known:
The dioxide may be prepared by, for example, halogenization of lead(II) salts. The alpha allotrope is
Reaction of lead with sulfur or hydrogen sulfide yields lead sulfide. The solid has the NaCl-like structure (simple cubic), which it keeps up to the melting point, 1114 °C (2037 °F). If the heating occurs in presence of air, the compounds decomposes to give the monoxide and the sulfate.[7] The compounds are almost insoluble in water, weak acids, and (NH4)2S/(NH4)2S2 solution is the key for separation of lead from analytical groups I to III elements, tin, arsenic, and antimony. The compounds dissolve in nitric and hydrochloric acids, to give elemental sulfur and hydrogen sulfide, respectively.[7] Heating mixtures of the monoxide and the sulfide forms the metal.[2]
- 2 PbO + PbS → 3 Pb + SO2↑
Halides and other salts
Heating lead carbonate with hydrogen fluoride yields the hydrofluoride, which decomposes to the difluoride when it melts. This white crystalline powder is more soluble than the diiodide, but less than the dibromide and the dichloride. No coordinated lead fluorides exist (except the unstable PbF+ cation).[8] The tetrafluoride, a yellow crystalline powder, is unstable.
Other dihalides are received upon heating lead(II) salts with the halides of other metals; lead dihalides precipitate to give white
The metal is not attacked by
Chloride complexes

Lead(II) forms a series of complexes with chloride, the formation of which alters the corrosion chemistry of the lead. This will tend to limit the solubility of lead in saline media.
| Pb2+ + Cl− → PbCl+ | K1 = 12.59 |
| PbCl+ + Cl− → PbCl2 | K2 = 14.45 |
| PbCl2 + Cl− → PbCl− 3 |
K3 = 0.398 |
| PbCl− 3 + Cl− → PbCl2− 4 |
K4 = 0.0892 |
Organolead
The best-known compounds are the two simplest plumbane derivatives: tetramethyllead (TML) and tetraethyllead (TEL); however, the homologs of these, as well as hexaethyldilead (HEDL), are of lesser stability. The tetralkyl deratives contain lead(IV); the Pb–C bonds are covalent. They thus resemble typical organic compounds.[14]
Lead readily forms an equimolar alloy with
Phase diagrams of solubilities
Lead(II) sulfate is poorly soluble, as can be seen in the following diagram showing addition of SO2−
4 to a solution containing 0.1 M of Pb2+. The pH of the solution is 4.5, as above that, Pb2+ concentration can never reach 0.1 M due to the formation of Pb(OH)2. Observe that Pb2+ solubility drops 10,000 fold as SO2−
4 reaches 0.1 M.

|

|
| Plot showing aqueous concentration of dissolved Pb2+ as a function of SO2− 4[12] |
Diagram for lead in sulfate media[12] |
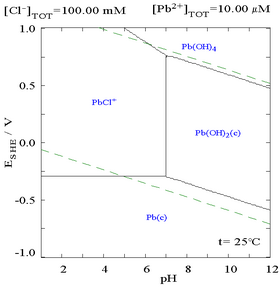
The addition of chloride can lower the solubility of lead, though in chloride-rich media (such as aqua regia) the lead can become soluble again as anionic chloro complexes.

|
|
| Diagram showing the solubility of lead in chloride media. The lead concentrations are plotted as a function of the total chloride present.[12] | Pourbaix diagram for lead in chloride (0.1 M) media[12] |
References
- ^ Polyanskiy 1986, pp. 14–15.
- ^ ISBN 978-0-486-65622-9.
- ^ ISBN 978-0-471-13557-9.
- ISBN 978-0-911910-26-1.
- ^ Polyanskiy 1986, p. 21.
- ^ a b c Polyanskiy 1986, p. 22.
- ^ a b Polyanskiy 1986, p. 28.
- ^ a b Polyanskiy 1986, p. 32.
- ^ Polyanskiy 1986, p. 33.
- ^ Polyanskiy 1986, p. 34.
- ISBN 978-0-471-18656-4.
- ^ a b c d e Puigdomenech, Ignasi (2004). Hydra/Medusa Chemical Equilibrium Database and Plotting Software. KTH Royal Institute of Technology. Archived from the original on 2007-09-29.
- ISBN 1566704626.
- ^ a b Polyanskiy 1986, p. 43.
- ISBN 0-911910-26-3.
- ^ a b Polyanskiy 1986, p. 44.
Bibliography
- Polyanskiy, N. G. (1986). Fillipova, N. A (ed.). Аналитическая химия элементов: Свинец [Analytical Chemistry of the Elements: Lead] (in Russian). Nauka.
