NADPH oxidase
| NAD(P)H oxidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
| Ferric reductase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | NADPH oxidase | ||||||||
TCDB | 5.B.1 | ||||||||
| OPM superfamily | 464 | ||||||||
| OPM protein | 5o05 | ||||||||
| |||||||||
Reaction
- NADPH + 2O2 ↔ NADP+ + 2O−2 + H+
Types
In mammals, NADPH oxidase is found in two types: one in
Neutrophilic type
The isoform found in neutrophils is made up of six subunits. These subunits are:
- a Rho GTPase, usually Rac1 or Rac2 (Rac stands for Rho-related C3 botulinum toxin substrate)
- Five phagocyticoxidase subunits:
- gp91phox(NOX2)
- p22phox (CYBA)
- p40phox(NCF4)
- p47phox(NCF1)
- p67phox(NCF2)
Vascular type
There are several vascular isoforms of the complex which use paralogs the NOX2 subunit:
Thyroid type
There are two further paralogs of NOX2 subunit in the thyroid:
- DUOX1
- DUOX2
Structure
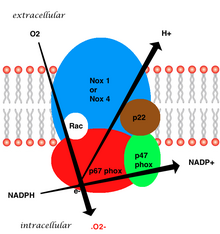
The whole structure of the membrane-bound vascular enzyme is composed of five parts: two
Biological function
NADPH oxidases (NOXes) are one of the major sources of cellular reactive oxygen species (ROS), and they still are the focus of extensive research interest due to their exclusive function in producing ROS under normal physiological conditions. The NADPH oxidase complex is dormant under normal circumstances but is activated to assemble in the membranes during respiratory burst. The activated NADPH oxidase generates superoxide which has roles in animal immune response and plant signalling.[9]
Superoxide can be produced in
In insects, NOXes had some functions clarified. Arthropods have three NOX types (NOX4-art, an arthropod-specific p22-phox-independent NOX4, and two calcium-dependent enzymes, DUOX).[16][17][18] In the gut, DUOX-dependent ROS production from bacteria-stimulated Drosophila melanogaster mucosa is an important pathogen-killing mechanism[19] and can increase defecation as a defense response.[20] In Aedes aegypti, DUOX is involved in the control of the gut indigenous microbiota.[21] Rhodnius prolixus has calcium activated DUOX, which is involved in eggshell hardening,[22] and NOX5, which is involved in the control of gut motility and blood digestion.[23][24]
Regulation
Careful regulation of NADPH oxidase activity is crucial to maintain a healthy level of ROS in the body. The enzyme is dormant in resting cells but becomes rapidly activated by several stimuli, including bacterial products and cytokines.
Ang-1 triggers NOX2, NOX4, and the mitochondria to release ROS and that ROS derived from these sources play distinct roles in the regulation of the Ang-1/Tie 2 signaling pathway and pro-angiogenic responses.[28]
Pathology
Superoxides are crucial in killing foreign bacteria in the human body. Consequently, under-activity can lead to an increased susceptibility to organisms such as catalase-positive microbes, and over-activity can lead to oxidative stress and cell damage.
Excessive production of ROS in vascular cells causes many forms of cardiovascular disease including
One study suggests a role for NADPH oxidase in
It has also been shown that NADPH oxidase plays a role in the mechanism that induces the formation of
Mutations
Mutations in the NADPH oxidase subunit genes cause several Chronic Granulomatous Diseases (CGD), characterized by extreme susceptibility to infection.[26] These include:
- X-linked chronic granulomatous disease (CGD)
- Autosomal recessive cytochrome b-negative CGD
- Autosomal recessive cytochrome b-positive CGD type I
- Autosomal recessive cytochrome b-positive CGD type II.
In these diseases, cells have a low capacity for phagocytosis, and persistent bacterial infections occur. Areas of infected cells are common, granulomas. A similar disorder called neutrophil immunodeficiency syndrome is linked to a mutation in the RAC2, also a part of the complex.
Inhibition
NADPH oxidase can be inhibited by apocynin, nitric oxide (NO), and diphenylene iodonium. Apocynin acts by preventing the assembly of the NADPH oxidase subunits. Apocynin decreases influenza-induced lung inflammation in mice in vivo and so may have clinical benefits in the treatment of influenza.[27]
Inhibition of NADPH oxidase by NO blocks the source of oxidative stress in the vasculature. NO donor drugs (
More advanced NADPH oxidase inhibitors include
References
- PMID 26814203.
- PMID 25263488.
- ^ PMID 15962105.
- PMID 9719063.
- PMID 8187280.
- PMID 9740615.
- PMC 10886416.
- PMID 8305740.
- ISSN 2090-0120.
- PMID 30755476.
- PMID 33669824.
- ^ PMID 21375590.
- PMID 23066164.
- PMID 15308657.
- PMID 27726789.
- PMID 28356077.
- PMID 17612411.
- ISBN 978-3-031-23751-5, retrieved 2023-10-19
- S2CID 12476863.
- PMID 26726767.
- PMID 21445237.
- PMID 24174530.
- PMID 29907654.
- PMID 33716782.
- PMID 16765921.
- ^ PMID 10720409.
- ^ PMID 21304882.
- PMID 28351775.
- PMID 11316520.
- PMID 19065049.
- PMID 19279347.
- S2CID 41932041.
- ^ Tom Fagan. Does Oxidative Stress Link NMDA and GABA Hypotheses of Schizophrenia? Archived 2007-12-30 at the Wayback Machine Schizophrenia Research Forum. December 09, 2007.
- PMID 24144948.
- PMID 22806357.
- ^ "Espacenet - Bibliographic data". worldwide.espacenet.com. Retrieved 2017-05-04.
- ^ "FDA granting Genkyotex Orphan Drug Designation of GKT137831 for IPF - Genkyotex S.A." pauahosting.co.nz. Retrieved 2017-05-04.[permanent dead link]
External links
- NADPH+Oxidase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- EC 1.6.3.1
