Plant holobiont
| Part of a series on |
| Microbiomes |
|---|
 |
Since the
Introduction

(B) The plant interacts with single organisms. It responds to the presence of a microbe and its metabolites and vice versa the microbe is affected by the plant environment and reacts to plant metabolism and physiology.
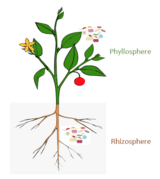
(C) The plant interacts with the microbiota in the soil and rhizosphere. Plant exudates attract microbes in the soil thereby directing a subset of them to the root zone. In turn, the activity of the microbiota in the root zone has strong impact on plant growth and health.
(D) The microorganisms within the root and rhizosphere microbiota dynamically interact with each other and the microbiota in the root.
Although most work on host-microbe interactions has been focused on animal systems such as corals, sponges, or humans, there is a substantial body of literature on plant holobionts.
The plant holobiont is relatively well-studied, with particular focus on agricultural species such as
Like all other organisms, plants do not lead solitary lives, as there are myriads of microbes and viruses living around and within them. Some microbes, whether
Research focussing on the widely accepted one pathogen–one disease hypothesis has led to many breakthroughs, such as the identification of diseases and novel disease-causing organisms, as well as the development of control strategies using effective compounds against individual pathogens, which have proven successful in controlling several diseases.[17] However, this came at the cost of neglect of plant pathology in a holistic approach—or systems-based plant pathology—in which communities and their interactions are considered rather than individual organisms. This reductionist scheme has limited our ability to overcome certain important challenges, such as the emergence of novel and severe diseases, with little that could be done to counter these diseases without considering the associated biotic factors.[17][16]
The plant holobiont

As functional macrobes living in a close association with diverse communities of microbes and viruses, plants should be considered a "holobiont", viewed as a complex system in continuous interaction with the resident microbes and the surrounding environment.[18] The microbes with their functional genes represent the plant microbiome, or the phytobiome, and their composition may differ among individual plants, as well as across various stages of growth or sites and tissues of the same plant. Despite the extensive taxonomic overlap between the microbiomes of different plant tissues, each compartment exhibits a unique composition of strains and species, as evidenced from the specificity of different operational taxonomic units (OTUs) in various tissues of plants within the same genus.[15][19] The beneficial roles of microbes associated with plants include, but are not limited to, supporting plant growth at different stages starting from seed germination, promoting plant resistance to biotic and abiotic stresses, and assisting plants in nutrient uptake.[20][21][4] The plant growth-promoting bacteria and the arbuscular mycorrhiza represent the beneficial microbes that are mostly involved in supporting plant growth and nutrition by facilitating nutrients mobilisation. The mycorrhizae were even reported to manipulate plant hormonal signalling to facilitate their colonisation of the root surface in a way similar to the mechanisms of some pathogenic microbes, while in this case, the hijacking is beneficial for the host plant.[4][16]
Alternatively, pathogenic microbes are also a part of the phytobiome, although this concept has been partly overlooked in most studies on this topic.[15] Despite their presence within the microbial communities, pathogenic microbes are differentiated from the rest of the phytobiome based on their ability to damage the plant tissues through transient blooming under specific conditions, which is consistent with the core concept of the disease triangle in epidemiology.[22][16]
Defining specific taxonomic groups as pathogenic or beneficial could be misleading, as some microbial genera might include beneficial members that support growth at certain stages of a plant species, but are pathogenic at another stage or to other plant species. For instance, while some members of the genus
In this context, plant pathogenic microbes may specifically manipulate the structure of the plant microbiome to generate conditions conducive to their own survival and colonisation. Kim et al. demonstrated that the plant pathogen Burkholderia glumae employs the specific type-6 secretion system (T6SS) for interaction with rice endophytic microbes, thereby reducing the populations of specific bacterial genera, such as Luteibacter and Dyella, which promote plant growth and contribute to protection against pathogenic bacteria.[29] Metagenomic analysis in their study also revealed significant changes in the community structure of endophytic microbiota in infected rice plants compared with non-infected plants or plants infected with a T6SS-defective B. glumae mutant. Specifically, these changes facilitated the colonisation and establishment of B. glumae at the early stages of infection.[29][16]
Another example in which the plant-associated beneficial bacteria turn harmful under specific conditions to support the development of the disease is the root-knot caused by the nematode Meloidogyne incognita. Nematode infection is associated with the presence of specific microbes harbouring abundant genes involved in pathogenesis, such as genes encoding plant polysaccharide-degrading enzymes.[30] Hence, assessments of the taxonomic composition should always be aligned to functional analyses of the existing microbial communities, and the plant holobiont should be separated into the symbiome or pathobiome under specific conditions based on function rather than taxonomy. Overall, the plant holobiont could be represented as a never-ending war between the allies of pathogenic microbes, as the pathobiome, and the key beneficial microbes, as the symbiome.[16]
Symbiotic relationships
Under natural conditions, plants are always associated with a well-orchestrated community of microbes — the phytomicrobiome. The nature and degree of microbial effect on the plant host can be positive, neutral, or negative, and depends largely on the environment. The phytomicrobiome is integral for plant growth and function; microbes play a key role in plant nutrient acquisition,
The phytomicrobiome is composed of a community of microorganisms that associate and interact with a host plant including

The earliest and arguably most essential example of a specific symbiotic function within the plant holobiome arose from the endosymbiosis of an
In this paper, we focus on how beneficial bacteria and fungi, a relatively small fraction of the phytomicrobiome, have had a disproportionately large influence on plant holobiont evolution. We also review the fundamental roles that the phytomicrobiome plays in plant holobiont development and survival. Finally, we propose that a greater integration of holobiont theory should be incorporated into the plant sciences.[31]
Origin of the plant holobiont
- Endosymbiosis of prokaryotes and the rise of plant holobionts
Life on earth is believed to stem from a single origin, the microbial ancestor that emerged as early as 3.5 billion years ago.[39][40] According to endosymbiosis theory, about 1.5 billion years ago, a proto-eukaryotic cell engulfed an alphaproteobacteria, forming an endosymbiotic relationship, and gradually developed into what is now recognized as the mitochondrion (see diagram).[41] Mitochondria use alternative electron acceptors to generate adenosine triphosphate (ATP) and are now the most important organelle for plant respiration since they enable metabolic reactions to convert energy into usable forms.[31]
Approximately half of a billion years later,[42] eukaryotic cells containing mitochondria engulfed cyanobacteria (photosynthetic prokaryotes), which like the alphaproteobacteria became fully incorporated into and dependent on plant cells, resulting in the chloroplast (diagram).[43][44][45] Chloroplasts convert energy from the sun into carbohydrates, using water as the electron donor. However, large-scale gene loss from plastids has occurred during the course of evolution,[46] and higher plant chloroplasts now contain only 120–130 genes [42] compared with the 1700 to 7500 genes contained in cyanobacterial genomes.[47] In spite of their reduced genome size, chloroplasts and cyanobacteria still carry out some of the same functions, ranging from gene expression to metabolism.[48][49] For example, it is clear that the protein targeting system of cyanobacteria is similar to that of the chloroplast.[50][31]
Organisms have been described as entities evolved from constituent elements that are highly cooperative and minimally conflicting; however, there is ongoing debate regarding the levels of cooperation and conflict within holobionts.[38][50] In plants, chloroplasts and mitochondria are highly cooperative with plant cells while relationships between the plant and the phytomicrobiome are more varied including the mutualistic and parasitic interactions. For example, relationships between plants and PGPB are organismal given that they are highly cooperative and low conflict in nature. On the other hand, some plant-microbe interactions are more opportunistic for one member and therefore are not organismal. The plant is therefore a eukaryotic organism, with prokaryotic constituents (entrained microbes), that interacts with its phytomicrobiome to form the plant holobiont.[38] As a result, the difference between the plant and the phytomicrobiome blurs and the concept of the holobiont becomes pre-eminent. It can then be argued that the influence of microbes on the evolution of plants is so profound that without microbes, the concept of the “plant” fails.[31]
Transition to land
The
See also
- Seagrass holobiont
References
- PMID 29587885..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 27418200..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 27065991.
- ^ PMID 25655016.
- S2CID 11555411.
- PMID 29244764.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 29695294.
- PMID 33427409.
- ^ PMID 3294806.
- PMID 31608075.
- PMID 25756954.
- PMID 29024322.
- S2CID 210924640.
- ^ PMID 24634890.
- ^ PMID 28545003.
- ^ PMID 33435275..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ PMID 18544040.
- ^ Gordon J, Knowlton N, Relman DA, Rohwer F and Youle M. (2013) "Superorganisms and holobionts". Microbe, 8(4): 152–153.
- S2CID 2845899.
- PMID 26547794.
- S2CID 10806491.
- S2CID 5911375.
- ISBN 978-90-481-4597-3.
- doi:10.1139/B09-092.
- ^ PMID 21677694.
- PMID 26113855.
- PMID 24421406.
- .
- ^ PMID 32643866.
- PMID 26603211.
- ^ PMID 33805166..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ PMID 32039567.
- ^ PMID 26284777.
- ^ S2CID 90306574.
- PMID 31849163.
- ^ S2CID 155103906.
- ^ Selosse, Marc-André and Joyard, Jacques (2021) Symbiosis and evolution: at the origin of the eukaryotic cell, Encyclopedia of the Environment, ISSN 2555-0950. Accessed: 6 June 2021.
- ^ S2CID 89188534.
- S2CID 10419280.
- S2CID 205250494.
- PMID 25848019.
- ^ PMID 24991417.
- PMID 18315522.
- PMID 20124341.
- PMID 28356502.
- PMID 12218172.
- S2CID 86165973.
- PMID 28887328.
- ISBN 978-3-642-64598-3.
- ^ S2CID 46881244.
- S2CID 84397640.
- S2CID 231955632.
- S2CID 198933932.
- PMID 30405652.
- PMID 32523595.
