User:B1357M/sandbox
- This article is about molecular biology. For other uses, see Fadd (disambiguation).
Fas-Associated protein with Death Domain (FADD), also called MORT1, is encoded by the FADD gene on the 11q13.3 region of chromosome 11 in humans.[1]
FADD is an
Contents
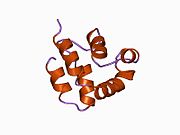
Structure
FADD is a 23KDa protein, made up of 280 amino acids. It contains two main domains: a C terminal
Functions
Extrinsic apoptosis
Upon stimulation by the
Binding of TRAIL to death rececptors four and five (DR4 and DR5) can lead to apoptosis by the same mechanism.[11]
Apoptosis can also be triggered by binding of a ligand to

Necroptosis
FADD also plays a role in regulating
Autophagic cell death
Autophagy is a process which allows cell survival under stressed conditions but can also lead to cell death.
Using its DD, FADD interacts with
In contrast, it has also been found to inhibit autophagic cell death and therefore promote cell survival. FADD binds to ATG5 in a complex which also contains ATG12, Caspase 8 and RIPK1. The formation of this complex is stimulated by autophagic signalling. Caspase 8 then cleaves RIPK1, leading to inhibition of this signalling, inhibiting cell death.[14]
Development
FADD knockout in mouse embryos is lethal, showing a role for FADD in
FADD also plays a role in the development of the
Cell cycle regulation
FADD is thought to have a role in regulating the cell cycle of T lymphocytes. This regulation is dependent on phosphorylation of FADD on Serine 194, which is carried out by Casein Kinase 1a (CKIα). This phosphorylated form of FADD is found mainly in the nucleus and the abundance of phosphorylated FADD increases significantly in the G2 phase of the cell cycle compared to the G1 phase where only very little can be detected. As it is found at the mitotic spindle during G2, it has been proposed to mediate the G2/M transition, however, the mechanism by which it does this it not yet known.[18]
Lymphocyte proliferation
FADD is essential for
Inflammation
Activation of Nuclear Factor Kappa B (NFκB) signalling leads to
Other
FADD is required for an efficient antiviral response. Upon viral infection, FADD is needed to increase the levels of
FADD is involved in the activation of the
Recent research has also shown that it may have a role in regulating glucose levels and the phosphorylated form of FADD is important for this function.[25]
Regulation
Subcellular localisation
FADD can be found in both the nucleus and cytoplasm of cells. Phosphorylation of Ser194 of FADD in humans (or Ser191 in mice) is thought to regulate its subcellular localisation. A nuclear localization sequence and nuclear export signal, both located in the DED of FADD, are also required for it to enter and exit the nucleus. Depending on its
c-FLIP
Cellular FLICE inhibitory protein (c-FLIP) is a regulatory protein which contains two DEDs. There are two isoforms of C-FLIP: C-FLIPs and FLIPl. It was originally thought to act as a negative regulator of apoptosis by binding to the DED of FADD and therefore preventing procaspase 8 from binding and inhibiting formation of the DISC.[27] However, it has been seen that both c-FLIP and procaspase 8 can be found at the same DISC.[28] Therefore it has been proposed that the presence of c-FLIP inhibits the close interaction of the procaspases to one another. Without this close proximity, the procaspases cannot be completely cleaved and remain in an inactive state.[27]

PKC
The activity of protein kinase C has a negative effect on Fas receptor mediated apoptosis. This is because it inhibits the recruitment of FADD to the receptor and so a DISC is not formed. It has been shown that by either increasing or decreasing the amount of PKC in T cells, more or less FADD is recruited to FasR respectively, when the FasR is stimulated.[29]
MKRN1
MKRN1 is an E3 ubiquitin ligase which negatively regulates FADD by targeting it for ubiquitin mediated degradation. In doing so, MKRN1 is able to control the level of apoptosis.[30]
Roles in inflammatory diseases
Increased levels of FADD were found in the leukocytes of patients with relapsing remitting multiple sclerosis, contributing to inflammation.[31] In
Roles in Cancer
As FADD has such an important role in apoptosis, loss of FADD can give cancer cells a proliferative advantage as apoptosis would no longer be induced when the Fas receptors are stimulated.[22]
However, there is significant upregulation of FADD in
Therapeutic Target
Taxol is a drug used in anticancer therapies due to its ability to interfere with microtubule assembly, which leads to cell cycle arrest. FADD phosphorylated at Ser194 makes cells more sensitive to cell cycle arrest induced by taxol.[18] Taxol can also cause apoptosis of cells and this requires procaspase 10, which is activated by recruitment to FADD.[35]
It has been shown that the activation of JNK leads to the phosphorylation of FADD. Phosphorylated FADD can induce G2/M cell cycle arrest, potentially by increasing the stability of p53. Therefore drugs which can activate this pathway may have a therapeutic potential.[36] However, high levels of phosphorylated FADD have been correlated with a poor prognosis in many cancers such as that of the head and neck. This is likely to be due to its activation of the NFkB pathway, which is antiapoptotic. Therefore, inhibition of FADD phosphorylation may be developed as a potential anti cancer stategy.[37]
Interactions
FADD has been seen to interact with
See also
- TRADD
- Intrinsic apoptosis
References
- PMID 8955195.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 8967952.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 9582077.
- ^ PMID 7536190.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 10347191.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 8681376.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 11583996.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 8521815.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21978761.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 23010170.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 10783243.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 12887920.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 15778222.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 18946037.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 9506948.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 23025414.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 17656375.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 16061179.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 9521326.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 16709845.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 10823821.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 20576468.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 17277150.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 22582393.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 23828893.)
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link - PMID 12815462.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 11713262.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 9880531.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 11581255.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 22864571.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 24603611.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 15004557.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 23763459.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 22669160.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 15452117.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 16450001.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21859840.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 12235128.
- PMID 10442631.
- PMID 12684039.
- PMID 11741985.
- )
Further reading
External links
- Fas-Associating+Protein+with+Death+Domain at the U.S. National Library of Medicine Medical Subject Headings (MeSH)