18-electron rule
The 18-electron rule is a
Applicability
The rule usefully predicts the formulas for
Ligands in a complex determine the applicability of the 18-electron rule. In general, complexes that obey the rule are composed at least partly of
Consequences for reactivity
Compounds that obey the 18-electron rule are typically "exchange inert". Examples include
Complexes with fewer than 18 valence electrons tend to show enhanced reactivity. Thus, the 18-electron rule is often a recipe for non-reactivity in either a
Duodectet rule
Computational findings suggest valence p-orbitals on the metal participate in metal-ligand bonding, albeit weakly.
The current consensus in the general chemistry community is that unlike the singular octet rule for main group elements, transition metals do not strictly obey either the 12-electron or 18-electron rule, but that the rules describe the lower bound and upper bound of valence electron count respectively.[5][6] Thus, while transition metal d-orbital and s-orbital bonding readily occur, the involvement of the higher energy and more spatially diffuse p-orbitals in bonding depends on the central atom and coordination environment.[7][8]
Exceptions
π-donor or σ-donor ligands with small interactions with the metal orbitals lead to a weak ligand field which increases the energies of t2g orbitals. These molecular orbitals become non-bonding or weakly anti-bonding orbitals (small Δoct). Therefore, addition or removal of electron has little effect on complex stability. In this case, there is no restriction on the number of d-electrons and complexes with 12–22 electrons are possible. Small Δoct makes filling eg* possible (>18 e−) and π-donor ligands can make t2g antibonding (<18 e−). These types of ligand are located in the low-to-medium part of the spectrochemical series. For example: [TiF6]2− (Ti(IV), d0, 12 e−), [Co(NH3)6]3+ (Co(III), d6, 18 e−), [Cu(OH2)6]2+ (Cu(II), d9, 21 e−).
In terms of metal ions, Δoct increases down a group as well as with increasing
16-electron complexes
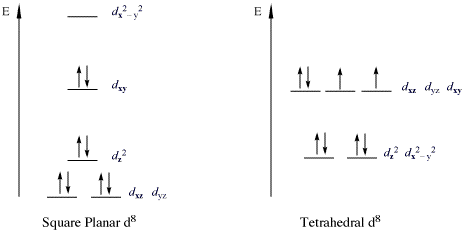
An important class of complexes that violate the 18e rule are the 16-electron complexes with metal d8 configurations. All
Many
Other violations can be classified according to the kinds of ligands on the metal center.
Bulky ligands
Bulky ligands can preclude the approach of the full complement of ligands that would allow the metal to achieve the 18 electron configuration. Examples:
- Ti(neopentyl)4 (8 e−)
- Cp*2Ti(C2H4) (16 e−)
- V(CO)6 (17 e−)
- Cp*Cr(CO)3 (17 e−)
- Pt(PtBu3)2 (14 e−)
- Co(norbornyl)4 (13 e−)
- [FeCp2]+ (17 e−)
Sometimes such complexes engage in agostic interactions with the hydrocarbon framework of the bulky ligand. For example:
- W(CO)3[P(C6H11)3]2 has 16 e− but has a short bonding contact between one C–H bond and the W center.
- Cp(PMe3)V(CHCMe3) (14 e−, diamagnetic) has a short V–H bond with the 'alkylidene-H', so the description of the compound is somewhere between Cp(PMe3)V(CHCMe3) and Cp(PMe3)V(H)(CCMe3).
High-spin complexes
High-spin metal complexes have singly occupied orbitals and may not have any empty orbitals into which ligands could donate electron density. In general, there are few or no π-acidic ligands in the complex. These singly occupied orbitals can combine with the singly occupied orbitals of radical ligands (e.g., oxygen), or addition of a strong field ligand can cause electron-pairing, thus creating a vacant orbital that it can donate into. Examples:
- CrCl3(THF)3 (15 e−)
- [Mn(H2O)6]2+ (17 e−)
- [Cu(H2O)6]2+ (21 e−, see comments below)
Complexes containing strongly π-donating ligands often violate the 18-electron rule. These ligands include
(RN2−). Examples:- [CrO4]2− (16 e−)
- Mo(=NR)2Cl2 (12 e−)
In the latter case, there is substantial donation of the nitrogen lone pairs to the Mo (so the compound could also be described as a 16 e− compound). This can be seen from the short Mo–N bond length, and from the angle Mo–N–C(R), which is nearly 180°. Counter-examples:
- trans-WO2(Me2PCH2CH2PMe2)2 (18 e−)
- Cp*ReO3 (18 e−)
In these cases, the M=O bonds are "pure" double bonds (i.e., no donation of the lone pairs of the oxygen to the metal), as reflected in the relatively long bond distances.
π-donating ligands
Ligands where the coordinating atom bear nonbonding lone pairs often stabilize unsaturated complexes. Metal amides and alkoxides often violate the 18e rule
Combinations of effects
The above factors can sometimes combine. Examples include
- Cp*VOCl2 (14 e−)
- TiCl4(8 e−)
Higher electron counts
Some complexes have more than 18 electrons. Examples:
- Cobaltocene (19 e−)
- Nickelocene (20 e−)
- The hexaaquacopper(II) ion [Cu(H2O)6]2+ (21 e−)
- TM(CO)8− (TM = Sc, Y) (20 e−)
Often, cases where complexes have more than 18 valence electrons are attributed to electrostatic forces – the metal attracts ligands to itself to try to counterbalance its positive charge, and the number of electrons it ends up with is unimportant. In the case of the metallocenes, the chelating nature of the cyclopentadienyl ligand stabilizes its bonding to the metal. Somewhat satisfying are the two following observations: cobaltocene is a strong electron donor, readily forming the 18-electron cobaltocenium cation; and nickelocene tends to react with substrates to give 18-electron complexes, e.g. CpNiCl(PR3) and free CpH.
In the case of nickelocene, the extra two electrons are in orbitals which are weakly metal-carbon antibonding; this is why it often participates in reactions where the M–C bonds are broken and the electron count of the metal changes to 18.[9]
The 20-electron systems TM(CO)8− (TM = Sc, Y) have a cubic (Oh) equilibrium geometry and a singlet (1A1g) electronic ground state. There is one occupied valence MO with a2u symmetry, which is formed only by ligand orbitals without a contribution from the metal AOs. But the adducts TM(CO)8− (TM=Sc, Y) fulfill the 18-electron rule when one considers only those valence electrons, which occupy metal–ligand bonding orbitals.[10]
See also
- Electron counting – Formalism used for classifying compounds
- Ligand field theory – Molecular orbital theory applied to transition metal complexes
- d electron count – Description of the electron configuration
- Tolman's rule – Rule describing chemical reactions
References
- ^ PMID 17843674.
- doi:10.1021/ed082p28.
- ISBN 978-3-527-33315-8.
- PMID 17063478.
- PMID 11749249.
- S2CID 195761899.
- .
- .
- ISBN 978-0-935702-48-4.
- PMID 29578636.
Further reading
- .