Valence electron
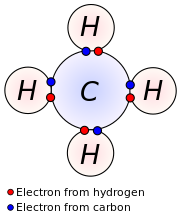
In
The presence of valence electrons can determine the element's chemical properties, such as its valence—whether it may bond with other elements and, if so, how readily and with how many. In this way, a given element's reactivity is highly dependent upon its electronic configuration. For a main-group element, a valence electron can exist only in the outermost electron shell; for a transition metal, a valence electron can also be in an inner shell.
An atom with a
Similar to a core electron, a valence electron has the ability to absorb or release energy in the form of a photon. An energy gain can trigger the electron to move (jump) to an outer shell; this is known as atomic excitation. Or the electron can even break free from its associated atom's shell; this is ionization to form a positive ion. When an electron loses energy (thereby causing a photon to be emitted), then it can move to an inner shell which is not fully occupied.
Overview
Electron configuration
The electrons that determine valence – how an atom reacts chemically – are those with the highest energy.
For a main-group element, the valence electrons are defined as those electrons residing in the electronic shell of highest principal quantum number n.[1] Thus, the number of valence electrons that it may have depends on the electron configuration in a simple way. For example, the electronic configuration of phosphorus (P) is 1s2 2s2 2p6 3s2 3p3 so that there are 5 valence electrons (3s2 3p3), corresponding to a maximum valence for P of 5 as in the molecule PF5; this configuration is normally abbreviated to [Ne] 3s2 3p3, where [Ne] signifies the core electrons whose configuration is identical to that of the noble gas neon.
However,
4). (But note that merely having that number of valence electrons does not imply that the corresponding oxidation state will exist. For example, fluorine is not known in oxidation state +7; and although the maximum known number of valence electrons is 16 in ytterbium and nobelium
The farther right in each transition metal series, the lower the energy of an electron in a d subshell and the less such an electron has valence properties. Thus, although a nickel atom has, in principle, ten valence electrons (4s2 3d8), its oxidation state never exceeds four. For zinc, the 3d subshell is complete in all known compounds, although it does contribute to the valence band in some compounds.[4] Similar patterns hold for the (n−2)f energy levels of inner transition metals.
The d electron count is an alternative tool for understanding the chemistry of a transition metal.
The number of valence electrons
The number of valence electrons of an element can be determined by the
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | H 1 |
He 2 | ||||||||||||||||||||||||||||||
| 2 | Li 1 |
Be 2 |
B 3 |
C 4 |
N 5 |
O 6 |
F 7 |
Ne 8 | ||||||||||||||||||||||||
| 3 | Na 1 |
Mg 2 |
Al 3 |
Si 4 |
P 5 |
S 6 |
Cl 7 |
Ar 8 | ||||||||||||||||||||||||
| 4 | K 1 |
Ca 2 |
Sc 3 |
Ti 4 |
V 5 |
Cr 6 |
Mn 7 |
Fe 8 |
Co 9 |
Ni 10 |
Cu 11 |
Zn 12 |
Ga 3 |
Ge 4 |
As 5 |
Se 6 |
Br 7 |
Kr 8 | ||||||||||||||
| 5 | Rb 1 |
Sr 2 |
Y 3 |
Zr 4 |
Nb 5 |
Mo 6 |
Tc 7 |
Ru 8 |
Rh 9 |
Pd 10 |
Ag 11 |
Cd 12 |
In 3 |
Sn 4 |
Sb 5 |
Te 6 |
I 7 |
Xe 8 | ||||||||||||||
| 6 | Cs 1 |
Ba 2 |
La 3 |
Ce 4 |
Pr 5 |
Nd 6 |
Pm 7 |
Sm 8 |
Eu 9 |
Gd 10 |
Tb 11 |
Dy 12 |
Ho 13 |
Er 14 |
Tm 15 |
Yb 16 |
Lu 3 |
Hf 4 |
Ta 5 |
W 6 |
Re 7 |
Os 8 |
Ir 9 |
Pt 10 |
Au 11 |
Hg 12 |
Tl 3 |
Pb 4 |
Bi 5 |
Po 6 |
At 7 |
Rn 8 |
| 7 | Fr 1 |
Ra 2 |
Ac 3 |
Th 4 |
Pa 5 |
U 6 |
Np 7 |
Pu 8 |
Am 9 |
Cm 10 |
Bk 11 |
Cf 12 |
Es 13 |
Fm 14 |
Md 15 |
No 16 |
Lr 3 |
Rf 4 |
Db 5 |
Sg 6 |
Bh 7 |
Hs 8 |
Mt 9 |
Ds 10 |
Rg 11 |
Cn 12 |
Nh 3 |
Fl 4 |
Mc 5 |
Lv 6 |
Ts 7 |
Og 8 |
Helium is an exception: despite having a 1s2 configuration with two valence electrons, and thus having some similarities with the alkaline earth metals with their ns2 valence configurations, its shell is completely full and hence it is chemically very inert and is usually placed in group 18 with the other noble gases.
Valence shell
The valence shell is the set of orbitals which are energetically accessible for accepting electrons to form chemical bonds.
For main-group elements, the valence shell consists of the ns and np orbitals in the outermost electron shell. For transition metals the orbitals of the incomplete (n−1)d subshell are included, and for lanthanides and actinides incomplete (n−2)f and (n−1)d subshells. The orbitals involved can be in an inner electron shell and do not all correspond to the same electron shell or principal quantum number n in a given element, but they are all at similar energies.[5]
| Element type | Hydrogen and helium | s- and p-blocks (main-group elements) |
d-block (Transition metals) |
f-block (Lanthanides and actinides) |
|---|---|---|---|---|
| Valence orbitals[6] |
|
|
|
|
| Electron counting rules | Duet/Duplet rule | Octet rule | 18-electron rule | 32-electron rule |
As a general rule, a main-group element (except hydrogen or helium) tends to react to form a s2p6 electron configuration. This tendency is called the octet rule, because each bonded atom has 8 valence electrons including shared electrons. Similarly, a transition metal tends to react to form a d10s2p6 electron configuration. This tendency is called the 18-electron rule, because each bonded atom has 18 valence electrons including shared electrons.
The heavy group 2 elements calcium, strontium, and barium can use the (n−1)d subshell as well, giving them some similarities to transition metals.[7][8][9]
Chemical reactions
The number of valence electrons in an atom governs its bonding behavior. Therefore, elements whose atoms have the same number of valence electrons are often grouped together in the periodic table of the elements, especially if they also have the same types of valence orbitals.[10]
The most
Within each group (each periodic table column) of metals, reactivity increases with each lower row of the table (from a light element to a heavier element), because a heavier element has more electron shells than a lighter element; a heavier element's valence electrons exist at higher principal quantum numbers (they are farther away from the nucleus of the atom, and are thus at higher potential energies, which means they are less tightly bound).[citation needed]
A
Within each group of nonmetals, reactivity decreases with each lower row of the table (from a light element to a heavy element) in the periodic table, because the valence electrons are at progressively higher energies and thus progressively less tightly bound. In fact, oxygen (the lightest element in group 16) is the most reactive nonmetal after fluorine, even though it is not a halogen, because the valence shells of the heavier halogens are at higher principal quantum numbers.
In these simple cases where the octet rule is obeyed, the valence of an atom equals the number of electrons gained, lost, or shared in order to form the stable octet. However, there are also many molecules that are exceptions, and for which the valence is less clearly defined.
Electrical conductivity
Valence electrons are also responsible for the bonding in the pure chemical elements, and whether their
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Group →
| ||||||||||||||||||||||||||||||||
| ↓ Period
| ||||||||||||||||||||||||||||||||
| 1 | H | He | ||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
A
A solid compound containing metals can also be an insulator if the valence electrons of the metal atoms are used to form
A
References
- OCLC 46872308.
- ^ THE ORDER OF FILLING 3d AND 4s ORBITALS. chemguide.co.uk
- ^ Miessler G.L. and Tarr, D.A., Inorganic Chemistry (2nd edn. Prentice-Hall 1999). p.48.
- .
- ^ ISBN 978-0-19-9604135.
- PMID 31276242.
- ISBN 978-0-08-037941-8.
- S2CID 235908113.
- PMID 32666598.
- ^ Jensen, William B. (2000). "The Periodic Law and Table" (PDF). Archived from the original (PDF) on 2020-11-10. Retrieved 10 December 2022.
External links
- Francis, Eden. Valence Electrons.
