Half sandwich compound
Half sandwich compounds, also known as piano stool complexes, are
-
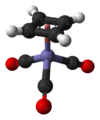
MMT is a commercially useful antiknock compound.
-
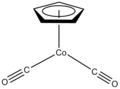
CpCo(CO)2 is a catalyst for the synthesis of pyridines.
-
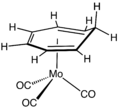
(C4H4)Fe(CO)3.
-
CpFe(CO)2I is an example of a piano stool complex with two different monodentate ligands.
-
The diruthenium of cymene is readily cleaved by ligands to give monoRu half-sandwich derivatives.
-
Cp2V2(CO)5 featuring a pair of semi-bridging CO ligands.[2]
(η5-C5H5) piano stool compounds
Half sandwich complexes containing cyclopentadienyl ligands are common. Well studied examples include (η5-C5H5)V(CO)4, (η5-C5H5)Cr(CO)3H, (η5-CH3C5H4)Mn(CO)3, (η5-C5H5)Cr(CO)3H, [(η5-C5H5)Fe(CO)3]+, (η5-C5H5)V(CO)4I, and (η5-C5H5)Ru(NCMe)+
3. (η5-C5H5)Co(CO)2 is a two-legged piano stool complex. Bulky cyclopentadienyl ligands such as 1,2,4-C5H2(tert-Bu)3− form unusual half-sandwich complexes.[3]
(η6-C6H6) piano stool compounds

In
Cr and Mn(I) (η6-C6H6) piano stool complexes
Piano stool complexes of the type (η6-C6H6)M(CO)3 are typically synthesized by heating the appropriate
Reactivity of (η6-C6H6)Cr(CO)3
The benzene ligand in (η6-C6H6)Cr(CO)3Mi is prone to deprotonation.
(η6-C6H6)Cr(CO)3 complexes exhibit "cine" and "tele" nucleophilic aromatic addition.
Reflecting its increased acidity, the benzene ligand can be lithiated with
(η6-C6H6)Cr(CO)3 is a useful
A variety of arenes ligands have been installed aside from benzene.
(η6-C6H6)M(CO)3 complexes serve as models for the interaction of metal carbonyls with graphene and carbon nanotubes.[16] The presence of M(CO)3 on extended π-network materials has been shown to improve electrical conductivity across the material.[17]
Reactivity of [(η6-C6H6)Mn(CO)3]+
Typical arene tricarbonyl piano stool complexes of Mn(I) and Re(I) are cationic and thus exhibit enhanced reactivity toward nucleophiles. Subsequent to nucleophilic addition, the modified arene can be recovered from the metal.[18][19]
(η6-C6H6)Ru complexes
(η6-C6H6)RuCl2 readily undergoes ligand exchange via cleavage of the chloride bridges, making this complex a versatile precursor to Ru(II) piano stool derivatives.[23]
References
- OCLC 1004583759.
- .
- S2CID 105376454.
- ^ Natta, G.; Ercoli, R.; F., Calderazzo (1958). "(η-C6H6)Cr(CO)3". Chimica e Industria. 40: 1003.
- .
- .
- .
- ^ ISBN 978-1-891389-53-5.
- ISBN 978-0-470-25762-3.
- ISBN 978-3-540-46128-9.
- ISBN 978-0471936237.
- .
- .
- PMID 21413775.
- .
- .
- .
- .
- .
- PMID 17960897.
- ISBN 9780470132524.
- )
- .




![Cp2V2(CO)5 featuring a pair of semi-bridging CO ligands.[2]](http://upload.wikimedia.org/wikipedia/commons/thumb/7/74/CPPCDV01.png/120px-CPPCDV01.png)





