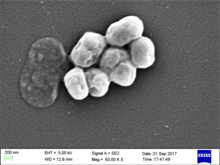
Acinetobacter baumannii
| Acinetobacter baumannii | |
|---|---|
| |
| Acinetobacter baumannii | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Pseudomonadota |
| Class: | Gammaproteobacteria |
| Order: | Pseudomonadales |
| Family: | Moraxellaceae |
| Genus: | Acinetobacter |
| Species: | A. baumannii
|
| Binomial name | |
| Acinetobacter baumannii Bouvet and Grimont 1986[1]
| |
Acinetobacter baumannii is a typically short, almost round, rod-shaped (
Bacteria of this genus lack
A. baumannii is part of the ACB complex (A. baumannii,
Colloquially, A. baumannii is referred to as "Iraqibacter" due to its seemingly sudden emergence in military treatment facilities during the Iraq War.[10] It has continued to be an issue for veterans and soldiers who served in Iraq and Afghanistan. Multidrug-resistant A. baumannii has spread to civilian hospitals in part due to the transport of infected soldiers through multiple medical facilities.[5] During the COVID-19 pandemic, coinfection with A. baumannii secondary to SARS-CoV-2 infections has been reported multiple times in medical publications.[11]
OmpA
Antibiotic resistance
Mechanisms of antibiotic resistance can be categorized into three groups. First, resistance can be achieved by reducing membrane permeability or increasing efflux of the antibiotic and thus preventing access to the target. Second, bacteria can protect the antibiotic target through genetic mutation or
AbaR resistance islands
Efflux pumps
Small RNA
Bacterial small RNAs are noncoding RNAs that regulate various cellular processes. Three sRNAs, AbsR11, AbsR25, and AbsR28, have been experimentally validated in the MTCC 1425 (ATCC15308) strain, which is a (multidrug-resistant) strain showing resistance to 12 antibiotics. AbsR25 sRNA could play a role in the efflux pump regulation and drug resistance.[20]
Beta-lactamase
A. baumannii has been shown to produce at least one
Biofilm formation
A. baumannii has been noted for its apparent ability to survive on artificial surfaces for an extended period of time, therefore allowing it to persist in the hospital environment. This is thought to be due to its ability to form
The formation of biofilm involves cell attachment, a fundamental process typically triggered by environmental metabolites. A. baumannii is able to use vanillic acid as its sole carbon source, like its close relative A. baylyi. This metabolic pathway is regulated by transcriptional repressor VanR. When vanillic acid enters the cell through VanP and VanK porins it binds to the VanR regulator, which is usually bound to PvanABKP and Pcsu promoters. This binding ables the repression of PvanABKP and Pcsu promoters, which leads to increased expression of VanP and VanK porins in the cell membrane and increased expression of Csu pili. The increased expression of Csu pili results a high biofilm formation phenotype of A. baumannii. [26]
Signs and symptoms of infection
A. baumannii is an opportunistic pathogen with a range of different diseases, each with their own symptoms. Some possible types of A. baumannii infections include:[citation needed]
- Pneumonia
- Bloodstream infections
- Meningitis
- Wound and surgical site infections, including necrotizing fasciitis
- Urinary tract infections
Symptoms of A. baumannii infections are often indistinguishable from other opportunistic infections caused by other opportunistic bacteria - including Klebsiella pneumoniae and Streptococcus pneumoniae.[citation needed]
Symptoms of A. baumannii infections in turn range from fevers and chills, rash, confusion and/or altered mental states, pain or burning sensations when urinating, strong urge to urinate frequently, sensitivity to bright light, nausea (with or without vomiting), muscle and chest pains, breathing problems, and cough (with or without yellow, green, or bloody mucus).[27] In some cases, A. baumannii may present no infection or symptoms, as with colonizing an open wound or tracheostomy site.[28]
Treatment
When infections are caused by antibiotic-susceptible Acinetobacter isolates, there may be several therapeutic options, including a broad-spectrum


Scientists at MIT, Harvard's Broad Institute and MIT's CSAIL found a compound named
Occurrence in veterans injured in Iraq and Afghanistan
American and other western soldiers in Iraq and Afghanistan were at risk of traumatic injury due to gunfire and improvised explosive devices. Previously, infection was thought to occur due to contamination with A. baumannii at the time of injury. Subsequent studies showd that although A. baumannii may be infrequently isolated from the natural environment, the infection was more likely nosocomially acquired, likely due to the ability of A. baumannii to persist on artificial surfaces for extended periods, and the several facilities to which injured soldiers were exposed during the casualty-evacuation process. Injured soldiers were first taken to level-I facilities, where they were stabilized. Depending on the severity of the injury, the soldiers might then be transferred to a level-II facility, which consists of a forward surgical team, for additional stabilization. Depending on the logistics of the locality, the injured soldiers might be transfer between these facilities several times before finally being taken to a major hospital within the combat zone (level III). Generally after 1–3 days, when the patients were stabilized, they were transferred by air to a regional facility (level IV) for additional treatment. For soldiers serving in Iraq or Afghanistan, this was typically Landstuhl Regional Medical Center in Germany. Finally, the injured soldiers were transferred to hospitals in their home country for rehabilitation and additional treatment.[41] This repeated exposure to many different medical environments seems to be the reason A. baumannii infections have become increasingly common. Multidrug-resistant A. baumannii is a major factor in complicating the treatment and rehabilitation of injured soldiers, and has led to additional deaths.[7][42][43]
Incidence in hospitals
Being referred to as an opportunistic infection, A. baumannii infections are highly prevalent in hospital settings. A. baumannii poses very little risk to healthy individuals;[44] however, factors that increase the risks for infection include:
- Having a weakened immune system
- Chronic lung disease
- Diabetes
- Lengthened hospital stays
- Illness that requires use of a hospital ventilator
- Having an open wound treated in a hospital
- Treatments requiring invasive devices like urinary catheters
A. baumannii can be spread through direct contact with surfaces, objects, and the skin of contaminated persons.[27]
The importation of A. baumannii and subsequent presence in hospitals has been well documented.[45] A. baumannii is usually introduced into a hospital by a colonized patient. Due to its ability to survive on artificial surfaces and resist desiccation, it can remain and possibly infect new patients for some time. A baumannii growth is suspected to be favored in hospital settings due to the constant use of antibiotics by patients in the hospital.[46] Acinetobacter can be spread by person-to-person contact or contact with contaminated surfaces.[47] Acinetobacter can enter through open wounds, catheters and breathing tubes.[48] In a study of European intensive care units in 2009, A. baumannii was found to be responsible for 19.1% of ventilator-associated pneumonia cases.[49]
| Country | Reference |
|---|---|
| Australia | [50][51] |
| Brazil | [52][53][54][55] |
| China | [56][57][58][59] |
| Germany | [60][61][62] |
| India | [63][64][65] |
| South Korea | [66][67][68][69] |
| United Kingdom | [70][71] |
| United States | [72][73][74][75] |
References
- LPSN.
- PMID 25516853.
- S2CID 30201194.
- ^ PMID 23483923.
- ^ S2CID 18294862.
- ISBN 978-0-387-95040-2.
- ^ PMID 22459899.
- PMID 1383266.
- PMID 18419525.
- ^ Drummond, Katie (2010-05-24). "Pentagon to Troop-Killing Superbugs: Resistance Is Futile". Wired.com. Condé Nast. Retrieved 8 April 2013.
- ^ PMID 33808905.
- PMID 19068136.
- S2CID 33040805.
- PMID 22546906.
- PMID 30521623.
- PMID 22290980.
- PMID 20375036.
- PMID 11709311.
- PMID 15388479.
- PMID 24705412.
- PMID 24277043.
- PMID 23439638.
- PMID 21975219.
- PMID 14663080.
- PMID 22733439.
- PMID 38308563.
- ^ a b "What Is Acinetobacter Baumannii?". Everyday Health. 18 June 2015. Retrieved 18 April 2017.
- ^ "Acinetobacter". Centers for Disease Control and Prevention. 6 November 2019. Retrieved 2023-03-09.
- PMID 22629456.
- PMID 15705318.
- PMID 25801348.
- PMID 21906345.
- ^ "Acinetobacter in Healthcare Settings". CDC. Retrieved 8 April 2013.
- ^ "He was dying. Antibiotics weren't working. Then doctors tried a forgotten treatment". Mother Jones. Retrieved 2018-05-17.
- PMID 30651225.
- S2CID 231584777.
- ^ Ray, Tiernan (13 March 2020). "MIT's deep learning found an antibiotic for a germ nothing else could kill". ZDNet.
- PMID 32084340.
- PMID 38172634.
- PMID 38172635.
- ^ "Army Medical Logistics" (PDF). FM 4-02.1. United States. Archived from the original (PDF) on 2013-03-23. Retrieved April 8, 2013.
- PMID 22325667.
- PMID 18316967.
- ^ "Acinetobacter in Healthcare Settings". Centers for Disease Control and Prevention. US Department of Health and Human Services. Retrieved 18 April 2017.
- PMID 16728314.
- S2CID 3446152.
- ^ "Acinetobacter in Healthcare Settings - HAI - CDC". www.cdc.gov. Retrieved 2 April 2018.
- ^ "Multidrug-Resistant Acinetobacter baumannii (MDRAB)." WHO Western Pacific Region, WPRO | WHO Western Pacific Region, 1 Nov. 2010, www.wpro.who.int/mediacentre/factsheets/fs_20101102/en/.
- S2CID 205537662.
- PMID 16916866.
- PMID 23527001.
- PMID 21131320.
- PMID 22168176.
- S2CID 21076295.
- PMID 22736746.
- PMID 21281989.
- PMID 19865060.
- PMID 19770790.
- S2CID 37311301.
- PMID 20835812.
- S2CID 23112180.
- PMID 16213625.
- PMID 23427525.
- PMID 22285540.
- PMID 16230195.
- PMID 21980849.
- S2CID 1078450.
- PMID 19954727.
- S2CID 42715424.
- PMID 21378633.
- S2CID 38699601.
- PMID 11805753.
- S2CID 20744216.
- PMID 21817084.
- S2CID 41059355.
