Coenzyme F420
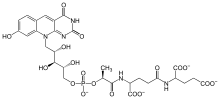
Coenzyme F420 is a family of
F420 was originally discovered in methanogenic archaea[1] and in Actinomycetota (especially in Mycobacterium).[2] It is now known to be used also by Cyanobacteria and by soil Proteobacteria, Chloroflexi and Firmicutes.[3] Eukaryotes including the fruit fly Drosophila melanogaster and the algae Ostreococcus tauri also use Coenzyme FO.[4]
F420 is structurally similar to
A number of F420 molecules, differing by the length of the oligoglutamyl tail, are possible; F420-2, for example, refers to the version with two glutamyl units attached. Lengths from 4 to 9 are typical.[3]
Biosynthesis
Coenzyme F420 is synthesized via a multi-step pathway:
- 7,8-didemethyl-8-hydroxy-5-deazariboflavin synthase (FbiC) produces Coenzyme FO (also written F0), itself a cofactor of DNA photolyase (antenna). This is the head portion of the molecule.[4]
- 2-phospho-L-lactate transferase (FbiA) produces Coenzyme F420-0, the portion containing the head, the diphosphate bridge, and ending with a carboxylic acid group.
- Coenzyme F420-0:L-glutamate ligase (one part of FbiB) puts a glutamate residue at the -COOH end, producing Coenzyme F420-1.
- Coenzyme F420-1:gamma-L-glutamate ligase (other part of FbiB) puts a gamma-glutamate residue at the -COOH end, producing Coenzyme F420-2, the final compound (in its oxidized form). Also responsible for adding additional units.
Oxidized F420 can be converted to reduced F420-H2 by multiple enzymes such as Glucose-6-phosphate dehydrogenase (coenzyme-F420) (Fgd1).[5]
Function
The coenzyme is a substrate for coenzyme F420 hydrogenase,[6] 5,10-methylenetetrahydromethanopterin reductase and methylenetetrahydromethanopterin dehydrogenase.[7][8]
A long list of other enzymes use F420 to oxidize (dehydrogenate) or F420-H2 to reduce substrates.[5]
Clinical relevance
See also
References
External links
- KEGG:
