Heme C
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChemSpider | |
| MeSH | heme+C |
PubChem CID
|
|
| |
| |
| Properties | |
| C34H36O4N4S2Fe | |
| Molar mass | 684.64904 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
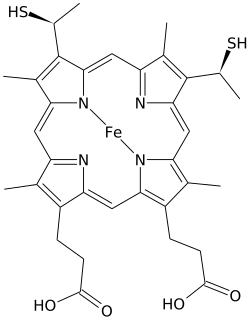
Heme C (or haem C) is an important kind of heme.
History
The correct structure of heme C was published in mid 20th century by the Swedish biochemist K.-G. Paul.
Properties
Heme C differs from
The number of heme C units bound to a
The thioether linkages seem to allow a great freedom of function for the holoproteins. In general, the c type cytochromes can be "fine tuned" over a wider range of oxidation-reduction potential than cytochromes b. This may be an important reason why cytochrome c is nearly ubiquitous throughout life. Heme C also plays an important role in apoptosis where just a few molecules of cytoplasmic cytochrome c, which must still contain heme C, leads to programmed cell death.[6] Cytochrome c can be measured in human serum and can be used as a marker for inflammation.[7]
In addition to these equatorial covalent bonds, the heme iron is also usually axially coordinated to the side chains of two amino acids, making the iron hexacoordinate. For example, mammalian and tuna cytochrome c contain a single heme C that is axially coordinated to side chains of both histidine and methionine.[8] Perhaps because of the two covalent bonds holding the heme to the protein, the iron of heme C is sometimes axially ligated to the amino group of lysine or even water.
See also
- Heme
- Protoporphyrin IX
- Heme A
- Heme B
- Hemeprotein
References
- .
- PMID 170266.
- PMID 188826.
- PMID 1311391.
- PMID 16248601.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 19030605.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 27489552.
- doi:10.1021/ar970084p.)
{{cite journal}}: CS1 maint: multiple names: authors list (link
