Copper(II) carbonate

| |
| Names | |
|---|---|
| IUPAC name
Copper(II) carbonate
| |
| Other names
Cupric carbonate, neutral copper carbonate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.013.338 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CuCO3 | |
| Molar mass | 123.5549 |
| Appearance | green or blue Powder[1] |
| insoluble in water [clarification needed] | |
Solubility product (Ksp)
|
10−11.45 ± 0.10 at 25 °C.[2][3][4] |
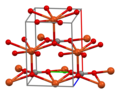
| Structure | |
| Pa-C2s (7) [1] | |
a = 6.092 Å, b = 4.493 Å, c = 7.030 Å α = 90°, β = 101,34°°, γ = 90°
| |
| 5 [1] | |
| Hazards | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Copper(II) sulfate |
Other cations
|
Nickel(II) carbonate Zinc carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Copper(II) carbonate or cupric carbonate is a
3.
This compound is rarely encountered because it is difficult to prepare[2] and readily reacts with water moisture from the air. The terms "copper carbonate", "copper(II) carbonate", and "cupric carbonate" almost always refer (even in chemistry texts) to a basic copper carbonate (or copper(II) carbonate hydroxide), such as Cu
2(OH)2CO
3 (which occurs naturally as the mineral malachite) or Cu
3(OH)2(CO
3)2 (azurite). For this reason, the qualifier neutral may be used instead of "basic" to refer specifically to CuCO
3.
Preparation
Reactions that may be expected to yield CuCO
3, such as mixing solutions of
2, due to the great affinity of the Cu2+
ion for the hydroxide anion HO−
.[5]
Thermal decomposition of the basic carbonate at atmospheric pressure yields copper(II) oxide CuO rather than the carbonate.
In 1960, C. W. F. T. Pistorius claimed synthesis by heating basic copper carbonate at 180 °C in an atmosphere of
2CO
3(OH)2, but a small yield of a rhombohedral substance was also obtained, claimed to be CuCO
3.[6] However, this synthesis was apparently not reproduced.[2]
Reliable synthesis of true copper(II) carbonate was reported for the first time in 1973 by Hartmut Ehrhardt and others. The compound was obtained as a gray powder, by heating basic copper carbonate in an atmosphere of carbon dioxide (produced by the decomposition of silver oxalate Ag
2C
2O
4) at 500 °C and 2 GPa (20,000 atm). The compound was determined to have a monoclinic structure.[7]
Chemical and physical properties
The stability of dry CuCO
3 depends critically on the partial pressure of carbon dioxide (pCO2). It is stable for months in dry air, but decomposes slowly into CuO and CO
2 if pCO2 is less than 0.11 atm.[3]
In the presence of water or moist air at 25 °C, CuCO
3 is stable only for pCO2 above 4.57 atmospheres and pH between about 4 and 8.[8] Below that partial pressure, it reacts with water to form a basic carbonate (azurite, Cu
3(CO
3)2(OH)2).[3]
- 3 CuCO
3 + H
2O → Cu
3(CO
3)
2(OH)
2 + CO
2
In highly basic solutions, the complex anion Cu(CO
3)2−
2 is formed instead.[3]
The solubility product of the true copper(II) carbonate was measured by Reiterer and others as pKso = 11.45 ± 0.10 at 25 °C.[2][3][4]
Structure
In the
-
Unit cell of CuCO3
-
Copper coordination environment
-
Carbonate coordination environment
References
- ^
- ^ a b c d Rolf Grauer (1999) "Solubility Products of M(II) Carbonates Archived 2018-11-01 at the Wayback Machine". Technical Report NTB-99-03, NAGRA - National Cooperative for the Disposal of Radioactive Waste; pages 8, 14, and 17. Translated by U. Berner.
- ^
- ^ a b F. Reiterer (1980): "Löslichkeitskonstanten und Freie Bildungsenthalpien neutraler Übergangsmetallcarbonate". Thesis, Montanuniversität Leoben.
- ISBN 9780750659246.