Cytokinesis

Cytokinesis (
Particular functions demand various deviations from the process of symmetrical cytokinesis; for example in
Plant cytokinesis differs from animal cytokinesis, partly because of the rigidity of plant cell walls. Instead of plant cells forming a cleavage furrow such as develops between animal daughter cells, a dividing structure known as the cell plate forms in the cytoplasm and grows into a new, doubled cell wall between plant daughter cells. It divides the cell into two daughter cells.
Cytokinesis largely resembles the
Etymology and pronunciation
The word "cytokinesis" (
Origin of this term is from Greek κύτος (kytos, a hollow), Latin derivative cyto (cellular), Greek κίνησις (kínesis, movement).
Animal cell
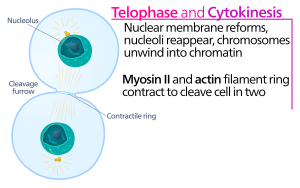
Animal cell cytokinesis begins shortly after the onset of sister chromatid separation in the anaphase of mitosis. The process can be divided to the following distinct steps: anaphase spindle reorganization, division plane specification, actin-myosin ring assembly and contraction, and abscission.[5] Faithful partitioning of the genome to emerging daughter cells is ensured through the tight temporal coordination of the above individual events by molecular signaling pathways.
Anaphase spindle reorganization
Animal cell cytokinesis starts with the stabilization of microtubules and reorganization of the mitotic spindle to form the central spindle. The
Division plane specification
The second step of animal cell cytokinesis involves division plane specification and cytokinetic furrow formation. Precise positioning of the division plane between the two masses of segregated chromosomes is essential to prevent chromosome loss. Meanwhile, the mechanism by which the spindle determines the division plane in animal cells is perhaps the most enduring mystery in cytokinesis and a matter of intense debate. There exist three hypotheses of furrow induction.[6] The first is the astral stimulation hypothesis, which postulates that astral microtubules from the spindle poles carry a furrow-inducing signal to the cell cortex, where signals from two poles are somehow focused into a ring at the spindle. A second possibility, called the central spindle hypothesis, is that the cleavage furrow is induced by a positive stimulus that originates in the central spindle equator. The central spindle may contribute to the specification of the division plane by promoting concentration and activation of the small GTPase RhoA at the equatorial cortex. A third hypothesis is the astral relaxation hypothesis. It postulates that active actin-myosin bundles are distributed throughout the cell cortex, and inhibition of their contraction near the spindle poles results in a gradient of contractile activity that is highest at the midpoint between poles. In other words, astral microtubules generate a negative signal that increases cortical relaxation close to the poles. Genetic and laser-micromanipulation studies in C. elegans embryos have shown that the spindle sends two redundant signals to the cell cortex, one originating from the central spindle, and a second signal deriving from the spindle aster, suggesting the involvement of multiple mechanisms combined in the positioning of the cleavage furrow. The predominance of one particular signal varies between cell types and organisms. And the multitude and partial redundancy of signals may be required to make the system robust and to increase spatial precision.[5]
Actin-myosin ring assembly and contraction
At the cytokinesis
Abscission
The cytokinetic furrow ingresses until a midbody structure (composed of electron-dense, proteinaceous material) is formed, where the actin-myosin ring has reached a diameter of about 1–2 μm. Most animal cell types remain connected by an intercellular cytokinetic bridge for up to several hours until they are split by an actin-independent process termed abscission, the last step of cytokinesis.[5][8]
The process of abscission physically cleaves the midbody into two. Abscission proceeds by removal of cytoskeletal structures from the cytokinetic bridge, constriction of the cell cortex, and plasma membrane fission. The intercellular bridge is filled with dense bundles of antiparallel microtubules that derive from the central spindle. These microtubules overlap at the midbody, which is generally thought to be a targeting platform for the abscission machinery.
The microtubule severing protein spastin is largely responsible for the disassembly of microtubule bundles inside the intercellular bridge. Complete cortical constriction also requires removal of the underlying cytoskeletal structures. Actin filament disassembly during late cytokinesis depends on the PKCε–14-3-3 complex, which inactivates RhoA after furrow ingression. Actin disassembly is further controlled by the GTPase Rab35 and its effector, the phosphatidylinositol-4,5-bisphosphate 5-phosphatase OCRL. Understanding the mechanism by which the plasma membrane ultimately splits requires further investigation.
Timing cytokinesis
Cytokinesis must be temporally controlled to ensure that it occurs only after sister chromatids separate during the anaphase portion of normal proliferative cell divisions. To achieve this, many components of the cytokinesis machinery are highly regulated to ensure that they are able to perform a particular function at only a particular stage of the cell cycle.[9][10] Cytokinesis happens only after APC binds with CDC20.[citation needed] This allows for the separation of chromosomes and myosin to work simultaneously.
After cytokinesis, non-kinetochore
Plant cell
Due to the presence of a
The
These tubules then widen and fuse laterally with each other, eventually forming a planar, fenestrated sheet [8]. As the

The construction of the new
Forces
Animal cells
Cytokinetic furrow ingression is powered by Type II Myosin ATPase. Since Myosins are recruited to the medial region, the contractile forces acting on the cortex resemble a 'purse string' constriction pulling inwards. This leads to the inward constriction. The plasma membrane by virtue of its close association with the cortex via crosslinker proteins [21] To the constriction of the cleavage furrow, the total surface area should be increased by supplying the plasma membrane via exocytosis.[22]
Theoretical models show that symmetric constriction requires both lateral stabilization and constriction forces.[23] Reduction of external pressure and of surface tension (by membrane trafficking) reduce the required stabilization and constriction forces.
Proteins involved in cytokinesis
CEP55 is a mitotic phosphoprotein that plays a key role in cytokinesis, the final stage of cell division.[24][25]
Clinical significance
In some cases, a cell may divide its genetic material and grow in size, but fail to undergo cytokinesis. This results in larger cells with more than one nucleus. Usually this is an unwanted aberration and can be a sign of cancerous cells.[26]
See also
- Diploid– Number of sets of chromosomes in a cell
- Telophase – Final stage of a cell division for eukaryotic cells both in mitosis and meiosis
- Prophase – First phase of cell division in both mitosis and meiosis
- Anaphase – Stage of a cell division
- Metaphase – Stage of cell division
- Mitosis – Process in which chromosomes are replicated and separated into two new identical nuclei
- Cell theory – Biology of cells
- Cytoskeleton – Network of filamentous proteins that forms the internal framework of cells
References
- PMID 21268179.
- ^ "cytokinesis". Lexico UK English Dictionary. Oxford University Press. Archived from the original on 2020-03-22.
- ^ "cytokinesis". Merriam-Webster.com Dictionary. Retrieved 2016-01-21.
- ^ Battaglia, Emilio (2009). Caryoneme alternative to chromosome and a new caryological nomenclature. Caryologia 62 (4): 1–80. link.
- ^ S2CID 3355851.
- ^ a b c d Morgan, David (2007). The Cell Cycle. New Science Press. pp. 157–173.
- PMID 34321459.
- ^ "Cytokinetic bridge". proteinatlas.org. Archived from the original on 28 August 2019. Retrieved 28 August 2019.
- S2CID 4418281.
- PMID 17488623.
- PMID 11074381.
- PMID 7559757.
- PMID 11549762.
- PMID 15020749.
- S2CID 11881080.
- PMID 16399085.
- PMID 11880633.
- ISSN 1040-2519.
- S2CID 84936099.
- ISBN 978-0-470-04737-8.
- ISBN 978-0-8153-3218-3.
- PMID 32322581.
- PMID 31869912.
- PMID 19855176.
- PMID 34710087.
- ISBN 978-1-947172-04-3.
Further reading
- The Molecular Requirements for Cytokinesis by M. Glotzer (2005), Science 307, 1735
- "Animal Cytokinesis: from parts list to mechanism" by Eggert, U.S., Mitchison, T.J., Field, C.M. (2006), Annual Review of Cell Biology 75, 543-66
- Campbell Biology (2010), 580-582
- More description and nice images of cell division in plants, with a focus on fluorescence microscopy
- Nanninga N (June 2001). "Cytokinesis in Prokaryotes and Eukaryotes: Common Principles and Different Solutions". Microbiol. Mol. Biol. Rev. 65 (2): 319–33. PMID 11381104.
