Gorgonopsia
| Gorgonopsians | |
|---|---|

| |
| Skeleton of Inostrancevia alexandri at the Museo delle Scienze, Trento, Italy | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Clade: | Synapsida |
| Clade: | Therapsida
|
| Clade: | Theriodontia |
| Clade: | †Gorgonopsia Lydekker, 1895 |
| Genera | |
Gorgonopsia (from the Greek
They markedly increased in size as time went on, growing from small skull lengths of 10–15 cm (4–6 in) in the Middle Permian to
The major therapsid groups had all evolved by 275 million years ago from a "
They became extinct during a phase of the
.Description

Earlier gorgonopsids in the Middle Permian were quite small, with skull lengths of 10–15 cm (4–6 in), whereas some later genera attained massive, bear-like sizes with the largest being Inostrancevia up to 3.5 m (11 ft) in length and 300 kg (660 lb) in body mass.[1] Nonetheless, small gorgonopsians remained abundant until extinction (though small species may actually represent juvenile specimens of other taxa).[2]
Like other Permian therapsids, gorgonopsians had developed several mammalian characteristics. These might have included a parasagittal gait (the limbs were vertically oriented and moved parallel to the spine) as opposed to the sprawling gait of
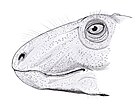
Skull
Anatomy varies incredibly little between gorgonopsians.[5] Many species are distinguished by vague proportional differences, and consequently smaller species may actually represent juveniles of larger taxa. Notably, the vomer at the tip of the snout varies among species in terms of the degree of its expansion, as well as the positions, degree of splay, and shape of the 3 ridges.[2] They typically feature a long and narrow skull.[4] Juvenile Rubidgea appear to have had snouts wider than long.[6] Unlike eutheriodonts, the occipital bone (at the back of the skull) is rectangular (wider than tall) and concave, as opposed to triangular.[7]: 279

The gorgonopsian brain, like other non-
Teeth

Like many mammals, gorgonopsians were heterodonts, with clearly defined incisors, canines, and postcanine teeth homologous with premolars and molars.[2] They had five incisors in the upper jaw (for most, the first three were the same size as each other, and the last two were shorter) and four on the bottom.[7]: 17–18
In the majority of gorgonopsians, the incisors were large, and the upper canines were elongated into sabres, much like those of later
Gorgonopsians were polyphyodonts, and teeth grew continuously throughout an individual's life.[12] Like some therapsids, while there was one functional canine, another canine was growing to replace it when it inevitably broke off. The left and right sides of the jaws did not have to be synchronous, so, for example, the first canine on the left side could be functional while the first canine on the right side was still growing.[12] Such a method might have been in play so as always to have a set of functional canines, as having a single or no canines would have severely impeded hunting, and growing such large teeth took a long time. On the other hand, because the functional canine is typically found in the foremost tooth socket (instead of equal occurrence in either socket), it is possible that canine replacement occurred a finite number of times, and the animal would eventually be left with a single, permanent set of functional canines in these sockets. In 1984, British palaeontologists Doris and Kenneth Kermack suggested that the canines grew to match the size of the skull, and continually broke off until the animal stopped growing, and that gorgonopsians featured an early version of finite tooth replacement exhibited in many mammals. The tooth replacement patterns of the other teeth are unclear.[9] The postcanine teeth were replaced more slowly than the other teeth, likely due to their lack of functional significance.[2]
Postcranium

The seven
The dorsals are spool-shaped and all appear about the same as each other. The spinous processes jut out steeply from the centra, and feature sharp keels on the front and back sides. Unlike eutheriodonts, gorgonopsians do not have distinguished lumbar vertebrae. Nonetheless, the dorsals equating to that series are similar to the lumbars of sabre-toothed cats with steeply oriented zygopophases, useful in stabilising the lower back especially when pinning down struggling prey.[7]: 293–295
There are three
Taxonomy
Fossil bearing sites

In 1876, the first gorgonopsian remains were identified in the

Gorgonopsians were first identified in Russia in the 1890s at the Sokolki locality on the Northern Dvina in Siberia under the supervision of Russian palaeontologist Vladimir Prokhorovich Amalitskii. In a posthumous publication, it was described as Inostrancevia alexandri, and it is one of the best known and largest gorgonopsians. Since then, only a few more Russian taxa have been described: Pravoslavlevia, Sauroctonus, Viatkogorgon, Suchogorgon, and Nochnitsa.[13] One of the Russian genus named, Leogorgon, is now considered as a nomen dubium.[6]
Gorgonopsians are conspicuously absent beyond these 2 areas.[13] In 1979, Chinese palaeontologist Yang Zhongjian described a Chinese gorgonopsian "Wangwusaurus tayuensis" based on teeth from the Late Permian Jiyuan Formation,[16] but in 1981, palaeontologists Denise Sigogneau-Russell and Ai-Lin Sun found the assigned material to be a random assemblage of which only two have even a remote similarity to Gorgonopsia.[17] In 2022, paleontologists Jun Liu and Wan Yiang identified a possible gorgonopsian dating from the Upper Permian in China, although it is only known from dentary remains.[18] In 2003, Indian palaeontologists Sanghamitra Ray and Saswati Bandyopadhyay assigned some skull fragments from the Late Permian Kundaram Formation to a medium-sized gorgonopsian,[19] though the gorgonopsian characteristics have also been documented in some therocephalians.[13] In 2008, a large and probably rubidgeine upper jaw fragment and canine was identified at the Late Permian Moradi Formation in Niger (one of the few low-latitude Late Permian tetrapod-bearing formations), and is the first evidence of a low-latitude gorgonopsian.[20]
Classification
Upon discovery, Owen presumed that Gorgonops and several other taxa he described from the Karoo Supergroup were
The number of South African genera rapidly grew in the 20th-century, headed principally by Broom, whose extensive work on the Karoo therapsids—from the beginning of his career in the country in 1897 to his death in 1951—led to his description of 57 gorgonopsian

Among the first attempts to organise the clade was carried out by British zoologist
The first
| |||||||||||||||||||||
| Phylogeny of Gorgonopsia according to Kammerer and Rubidge (2022)[31] |
Evolution
Synapsida has traditionally been split into the basal "
| Synapsida[32] | |||||||||||||||||||||||||||||||||
|
The Permian progressively became dryer and dryer. In the Upper Carboniferous and Lower Permian, pelycosaurs seem to have clung to the everwet
Throughout the Middle Permian, the often gigantic dinocephalians were the dominant animals of their ecosystems. They disappear from the fossil record during the
Palaeobiology
Bite

Gorgonopsians were likely active predators. The rubidgeines have an especially robust skull among gorgonopsians, comparable to those of enormous macropredators which use their skulls as their primary weapon, such as
The elongated canines have generally been thought to have been instrumental in their hunting tactics. The gorgonopsian jaw hinge was double jointed and made up of somewhat mobile and rotatable bones, which would have allowed them to open their mouths incredibly wide–perhaps in excess of 90°–without having to unhinge the jaw.[34] It has alternatively been suggested (first in 2002 by biologists Blaire Van Valkenburgh and Tyson Secco, though in reference to cats) that sabres evolved primarily due to sexual selection as a form of mating display. This is exhibited in some modern deer species, but is difficult to test given the lack of living sabre-toothed synapsid predators. In sabre-toothed cats, long-sabred ("dirk-toothed") taxa are thought to have been pursuit hunters, whereas short-toothed ("scimitar-toothed") taxa are thought to have been ambush predators.[35] Among the dirk-toothed cats, these predators are suggested to have killed with a well-placed slash to the throat after grappling prey, but gorgonopsians may have been less precise with bite placement, armed with reptilian jaws and tooth arrangements. Instead, gorgonopsians possibly used a bite-and-retreat tactic: the predator would ambush its quarry and take a sizable and debilitating bite out of it, and then follow as the prey tried to escape before succumbing to its injury, whereupon the gorgonopsian would deliver a killing bite. Because the postcanines are reduced or entirely absent, meat would have been forcibly torn away from the carcass and swallowed whole.[34] This "puncture–pull" strategy is also hypothesised to have been used by theropod dinosaurs.[10]

Gorgonopsians, along with other early carnivores as well as crocodiles, predominantly relied on "Kinetic-Inertial system" (KI) of biting down onto prey, in which the pterygoid and temporalis muscles rapidly clamped the jaws shut, using momentum and the kinetic energy of the jaws and teeth to grapple the victim. Mammalian carnivores, including sabre-toothed cats, instead rely mainly on the "Static-Pressure system" (SP) where the temporalis and masseter muscles produce a strong bite force to kill prey.[11] The temporalis and masseter had only separated in mammals, and gorgonopsians instead had a muscle stretching from the underside of the skull roof, back to the squamosal bone (at the back of the skull), and across the cheekbones. The part anchored by the cheeks stabilised the jawbone and allowed it to move side-to-side while closing. This may have been very important in biting, as the cheekbones get stronger in tandem with the canines getting longer.[7]: 278
Smaller gorgonopsians, such as Cyonosaurus (which may actually represent a juvenile of a different species), had gracile skulls and sabres, and may have acted much like jackals and foxes. Bigger gorgonopsians, such as Gorgonops, had long robust snouts with strongly flared cheeks, which would have supported strong pterygoids and a powerful KI bite. The medium-size Arctognathus had a box-like skull and resultantly powerful snout, which would have allowed strong bending and torsion movements, and a combination of both KI and SP bite elements. Even bigger gorgonopsians, such as Arctops, had a shorter and more convex snout like the earlier sphenecodont Dimetrodon, and would have been able to rapidly clamp the jaws shut from a wide gape (which would have been necessary given the long canines). The even larger Rubidgeinae had extremely powerful, heavily built, buttressed skulls, with wide snouts, strongly flanged cheeks, and exceedingly long teeth; the sabres of Rubidgea atrox are longer than the teeth of Tyrannosaurus.[11]
Unlike mammalian carnivores, gorgonopsians (and therocephalians) had reduced or completely lacked postcanines, and the jaw likely could not exert shearing pressure necessary for crushing bones open to access the bone marrow.[36] It has largely been unclear if bone marrow had even evolved yet in Permian synapsids (fish and many amphibians lack this in present day), but in 2021 it was shown that the Early Permian amphibians Seymouria and Discosauriscus likely had haematopoietic (red-blood-cell-producing) bone marrow in their limbs.[37]
Locomotion
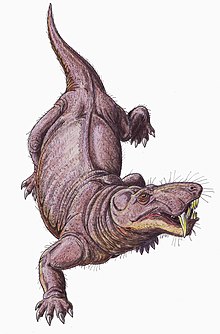
Gorgonopsians are considered to have been strictly
The gorgonopsian shoulder joint has a highly unusual configuration. The humeral head which connects to the shoulder is longer than the glenoid, so it could not fit into the cavity. Consequently, they may have been attached with a large mass of cartilage, with the humerus performing a rolling movement over the glenoid. This could theoretically make the angle between the humerus and the glenoid anywhere from 80 to 145° when facing the animal. If the angle was on the lower end, this would have been a rather firm joint, allowing the
The pelvis joint has the usual

In regard to how the feet were placed on the ground, gorgonopsians are the only early therapsids which present ectataxony (the last digit bears the most weight), homopody (footprints and handprints look the same), and semi-plantigrady (to some degree, the feet were placed flat on the ground).[39] These adaptations may have made gorgonopsians swifter and more agile than their prey.[34] Gorgonopsians had rather nimble digits, indicative of grasping capability for both the hands and feet, possibly for grappling struggling prey to prevent excessive load bearing on, and consequential fracturing or breaking of, the canines while they were sunk into the victim.[14]
Senses
Unlike eutheriodonts, but like some ectothermic creatures today, all gorgonopsians possessed a
Gorgonopsians have a rather short nasal cavity, like pelycosaurs, but it features abundant longitudinal ridges behind the

Early theriodonts (including gorgonopsians) may have possessed an eardrum, unlike earlier pelycosaurs, indicated by the reduction of the connection between the quadrate bone (at the jaw hinge) and the pterygoid bone (at the palate), allowing the quadrate to independently vibrate to a degree. This may have allowed the detection of air-borne sounds with a low amplitude of less than 1 mm (0.039 in), but the eardrum would have been supported by cartilage or ligaments instead of bone. If correct, then the postdentary bones (which in early mammals form the middle ear bones) would have needed to become detached from the dentary (jawbone); the gorgonopsian fossil record seems to indicate the postdentary-dentary connection was reduced. Though, given the specialisations required for biting, the condition of an isolating quadrate in gorgonopsians could alternatively be explained as streptostyly (rotatable quadrate) in order to widen the gape rather than facilitate hearing.[3]
Thermoregulation
A major anatomical shift occurred between earlier pelycosaurs and therapsids, which is postulated to have been related to an increasing
Modern large reptiles naturally give off body heat at a slower rate than smaller ones, and are considered "inertial homeotherms", but they maintain a low body temperature of 25–30 °C (77–86 °F). If therapsids required a higher body temperature of 35–40 °C (95–104 °F), they would either have needed to have been
If gorgonopsians were inertial homeotherms, it is not impossible that they had hair. The snout is typically riddled with
Palaeopathology
The anterolateral aspect of the left
The labial (lip/cheek) side of the tooth root of a functional canine of RB382 presents as many as 8 lesions, clustering along the midline of the tooth, which resemble miniature teeth with a
The adult snout SAM-PK-11490 from an indeterminate Middle Permian gorgonopsian species has an imbedded tooth from an unidentifiable animal. The bone developed a callus around the tooth, indicating it healed and the individual survived the attack. It either came from a predator — namely a biarmosuchian, a therocephalian, or another gorgonopsian — or intraspecific face biting as is commonly exhibited in social predators — such as big cats or monitor lizards, and it has been suggested for several extinct lineages such as theropods, aquatic reptiles, and saber-toothed cats. Social biting is intended to assert dominance or facilitate breeding, and, if correct, suggests at least some Middle Permian gorgonopsians were social carnivores. The tooth was initially overlooked so it is unclear how common this pathology actually is.[49]
Palaeoecology

Following the extinction of the dinocephalians and (in South Africa) the basal therocephalians
Extinction
Gorgonopsians went extinct at the end of the Upper Permian during the
See also
Notes
- pontine flexure; rob, right olfactory bulb; vc, vidian canal; vc=spa, vidian canal where the sphenopalatine artery passes; vcd, vena capitis dorsalis
- ^ This skeleton, discovered in the Usili Formation in Tanzania, was previously assigned to the species Sauroctonus parringtoni,[7] but more recent studies have proven that this attribution is wrong.[13]
- niche partitioning to smaller representatives after the Capitanian extinction.[31]
References
- S2CID 246318785.
- ^ PMID 30485338.
- ^ .
- ^ a b Swinton, W. E. (1954). Fossil amphibians and reptiles. British Museum (Natural History). p. 33.
- ISBN 9783110275902.
- ^ PMID 26823998.
- ^ a b c d e f g h i j k l m n o p q Gebauer, E. V. I. (2007). Phylogeny and Evolution of the Gorgonopsia with a Special Reference to the Skull and Skeleton of GPIT/RE/7113 (PDF) (PhD). Eberhard-Karls University of Tübingen. Archived from the original on 2012-07-22.
- ^ PMID 28413721.
- ^ ISBN 978-1-4684-7817-4.
- ^ PMID 33321067.
- ^ .
- ^ .
- ^ PMID 29900078.
- ^ PMID 25517726.
- ^ a b Owen, R. (1986). Descriptive and illustrated catalogue of the fossil Reptilia of South Africa in the collection of the British museum. British Museum (Natural History). pp. 27–29.
- ^ Young, C. C. (1979). "A Late Permian fauna from Jiyuan, Henan". Vertebrata PalAsiatica. 17 (2): 99–113.
- .
- . Retrieved 24 September 2022.
- . Retrieved 24 September 2022.
- ^ S2CID 131082914.
- ^ tectus in Charlton T. Lewis (1891) An Elementary Latin Dictionary, Harper & Brothers
- ^ Lydekker, R. (1890). Catalogue of the fossil Reptilia and Amphibia in the British Museum (Natural history) Part IV. British Museum (Natural History). p. 111.
- .
- ^ Osborn, H. F. (1903). "The Reptilian Subclasses Diapsida and Synapsida and the Early History of the Diaptosauria. Volume I. Part VIII" (PDF): 451–466.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Broom, R. (1905). "On the use of the term Anomodontia". Records of the Albany Museum. 1 (4): 266–269.
- .
- ^ Wyllie, A. (2003). "A review of Robert Broom's therapsid holotypes: Have they survived the test of time?" (PDF). Palaeontologia Africana. 39 (39): 2.
- ISBN 978-94-007-6841-3.
- ^
Sigogneau-Russell, Denise (1989). Wellnhofer, Peter (ed.). Theriodontia I: Phthinosuchia, Biarmosuchia, Eotitanosuchia, Gorgonopsia. Encyclopedia of Paleoherpetology. Vol. 17 B/I. Stuttgart: Gustav Fischer Verlag. ISBN 978-3437304873.
- S2CID 128762256.
- ^ S2CID 249977414.
- ^ S2CID 3184629.
- PMID 26156768.
- ^ ISBN 978-0-253-01042-1.
- PMID 23951334.
- ^ Cruikshank, A. R. I. (1973). "The Mode of Life of Gorgonopsians" (PDF). Palaeontologica Africana. 15 (1–2): 65–67.
- PMID 33648627.
- ^
Kriloff, A.; Germain, D.; Canoville, A.; Vincent, P.; Sache, M.; Laurin, M. (2008). "Evolution of bone microanatomy of the tetrapod tibia and its use in palaeobiological inference" (PDF). Journal of Evolutionary Biology. 21 (3): 807–826. PMID 18312321.
- S2CID 133781923.
- S2CID 59143925.
- PMID 25186003.
- ^ hdl:1957/35986.
- S2CID 22452028.
- .
- ^ Turner, J. S.; Tracey, C. R. (1986). "Body Size, Homeothermy and the Control of Body Heat in Mammal-like Reptiles". The Ecology and Biology of Mammal-like reptiles (PDF). Smithsonian Institution Press.
- JSTOR 2405973.
- PMID 31928188.
- PMID 27930769.
- .
- PMID 29900076.
- .
- ^ .
- ^ Yakimenko, E.; Inozemtsev, S. A.; Naugolnykh, S. V. (2004). "Upper Permian paleosols (Salarevskian Formation) in the central part of the Russian Platform: Paleoecology and paleoenvironment". Revista Mexicana de Ciencias Geológicas. 21 (1): 110–119. (convenience link)
- .
- PMID 30177561.
- S2CID 1231897.
External links
 Media related to Gorgonopsia at Wikimedia Commons
Media related to Gorgonopsia at Wikimedia Commons Data related to Gorgonopsia at Wikispecies
Data related to Gorgonopsia at Wikispecies