Smoke





Smoke is a suspension[3] of airborne particulates and gases[4] emitted when a material undergoes combustion or pyrolysis, together with the quantity of air that is entrained or otherwise mixed into the mass. It is commonly an unwanted by-product of fires (including stoves, candles, internal combustion engines, oil lamps, and fireplaces), but may also be used for pest control (fumigation), communication (smoke signals), defensive and offensive capabilities in the military (smoke screen), cooking, or smoking (tobacco, cannabis, etc.). It is used in rituals where incense, sage, or resin is burned to produce a smell for spiritual or magical purposes. It can also be a flavoring agent and preservative.
Smoke inhalation is the primary cause of death in victims of indoor fires. The smoke kills by a combination of thermal damage, poisoning and pulmonary irritation caused by carbon monoxide, hydrogen cyanide and other combustion products.
Smoke is an
Chemical composition
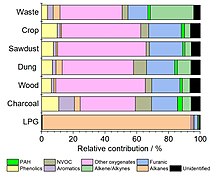
The composition of smoke depends on the nature of the burning fuel and the conditions of combustion. Fires with high availability of oxygen burn at a high temperature and with a small amount of smoke produced; the particles are mostly composed of

Pyrolysis of burning material, especially
Presence of sulfur can lead to formation of gases like hydrogen sulfide,
The visible
Smoke particulates, like other aerosols, are categorized into three modes based on particle size:
- nuclei mode, with geometric mean radius between 2.5 and 20 nm, likely forming by condensation of carbon moieties.
- accumulation mode, ranging between 75 and 250 nm and formed by coagulation of nuclei mode particles
- coarse mode, with particles in micrometer range
Most of the smoke material is primarily in coarse particles. Those undergo rapid
Aerosol of particles beyond visible size is an early indicator of materials in a preignition stage of a fire.[11]
Burning of hydrogen-rich fuel produces water vapor; this results in smoke containing droplets of water. In absence of other color sources (nitrogen oxides, particulates...), such smoke is white and cloud-like.
Smoke emissions may contain characteristic trace elements.
Traces of vanadium in high-temperature combustion products form droplets of molten
Some components of smoke are characteristic of the combustion source.
Many compounds can be associated with particulates; whether by being adsorbed on their surfaces, or by being dissolved in liquid droplets. Hydrogen chloride is well absorbed in the soot particles.[20]
Inert particulate matter can be disturbed and entrained into the smoke. Of particular concern are particles of asbestos.
Deposited
Polymers are a significant source of smoke. Aromatic
Visible and invisible particles of combustion


The
An
Smoke from a typical house fire contains hundreds of different chemicals and fumes. As a result, the damage caused by the smoke can often exceed that caused by the actual heat of the fire. In addition to the physical damage caused by the smoke of a
Dangers
Smoke from oxygen-deprived fires contains a significant concentration of compounds that are flammable. A cloud of smoke, in contact with atmospheric oxygen, therefore has the potential of being ignited – either by another open flame in the area, or by its own temperature. This leads to effects like backdraft and flashover. Smoke inhalation is also a danger of smoke that can cause serious injury and death.[citation needed]

Many compounds of smoke from fires are highly toxic and/or irritating. The most dangerous is carbon monoxide leading to carbon monoxide poisoning, sometimes with the additive effects of hydrogen cyanide and phosgene. Smoke inhalation can therefore quickly lead to incapacitation and loss of consciousness. Sulfur oxides, hydrogen chloride and hydrogen fluoride in contact with moisture form sulfuric, hydrochloric and hydrofluoric acid, which are corrosive to both lungs and materials. When asleep the nose does not sense smoke nor does the brain, but the body will wake up if the lungs become enveloped in smoke and the brain will be stimulated and the person will be awoken. This does not work if the person is incapacitated or under the influence of drugs and/or alcohol.[citation needed]


Smoke can obscure visibility, impeding occupant exiting from fire areas. In fact, the poor visibility due to the smoke that was in the
Corrosion
Smoke can contain a wide variety of chemicals, many of them aggressive in nature. Examples are
Corrosivity of smoke produced by materials is characterized by the corrosion index (CI), defined as material loss rate (angstrom/minute) per amount of material gasified products (grams) per volume of air (m3). It is measured by exposing strips of metal to flow of combustion products in a test tunnel. Polymers containing halogen and hydrogen (polyvinyl chloride, polyolefins with halogenated additives, etc.) have the highest CI as the corrosive acids are formed directly with water produced by the combustion, polymers containing halogen only (e.g. polytetrafluoroethylene) have lower CI as the formation of acid is limited to reactions with airborne humidity, and halogen-free materials (polyolefins, wood) have the lowest CI.[20] However, some halogen-free materials can also release significant amount of corrosive products.[29]
Smoke damage to electronic equipment can be significantly more extensive than the fire itself. Cable fires are of special concern; low smoke zero halogen materials are preferable for cable insulation.[30]
When smoke comes into contact with the surface of any substance or structure, the chemicals contained in it are transferred to it. The corrosive properties of the chemicals cause the substance or structure to decompose at a rapid rate. Certain materials or structures absorb these chemicals, which is why clothing, unsealed surfaces, potable water, piping, wood, etc., are replaced in most cases of structural fires.[citation needed]
Health effects of wood smoke
Wood smoke is a major source of
In the
Wood smoke (for example from
Measurement
As early as the 15th century Leonardo da Vinci commented at length on the difficulty of assessing smoke, and distinguished between black smoke (carbonized particles) and white 'smoke' which is not a smoke at all but merely a suspension of harmless water particulates.[62]
Smoke from heating appliances is commonly measured in one of the following ways:
In-line capture. A smoke sample is simply sucked through a filter which is weighed before and after the test and the mass of smoke found. This is the simplest and probably the most accurate method, but can only be used where the smoke concentration is slight, as the filter can quickly become blocked.[63]
The ASTM smoke pump is a simple and widely used method of in-line capture where a measured volume of smoke is pulled through a filter paper and the dark spot so formed is compared with a standard.
Filter/dilution tunnel. A smoke sample is drawn through a tube where it is diluted with air, the resulting smoke/air mixture is then pulled through a filter and weighed. This is the internationally recognized method of measuring smoke from combustion.[64]
Electrostatic precipitation. The smoke is passed through an array of metal tubes which contain suspended wires. A (huge) electrical potential is applied across the tubes and wires so that the smoke particles become charged and are attracted to the sides of the tubes. This method can over-read by capturing harmless condensates, or under-read due to the insulating effect of the smoke. However, it is the necessary method for assessing volumes of smoke too great to be forced through a filter, i.e., from bituminous coal.
Ringelmann scale. A measure of smoke color. Invented by Professor Maximilian Ringelmann in Paris in 1888, it is essentially a card with squares of black, white and shades of gray which is held up and the comparative grayness of the smoke judged. Highly dependent on light conditions and the skill of the observer it allocates a grayness number from 0 (white) to 5 (black) which has only a passing relationship to the actual quantity of smoke. Nonetheless, the simplicity of the Ringelmann scale means that it has been adopted as a standard in many countries.
Optical scattering. A light beam is passed through the smoke. A light detector is situated at an angle to the light source, typically at 90°, so that it receives only light reflected from passing particles. A measurement is made of the light received which will be higher as the concentration of smoke particles becomes higher.
Optical obscuration. A light beam is passed through the smoke and a detector opposite measures the light. The more smoke particles are present between the two, the less light will be measured.
Combined optical methods. There are various proprietary optical smoke measurement devices such as the 'nephelometer' or the 'aethalometer' which use several different optical methods, including more than one wavelength of light, inside a single instrument and apply an algorithm to give a good estimate of smoke. It has been claimed that these devices can differentiate types of smoke and so their probable source can be inferred, though this is disputed.[65]
Inference from carbon monoxide. Smoke is incompletely burned fuel, carbon monoxide is incompletely burned carbon, therefore it has long been assumed that measurement of CO in flue gas (a cheap, simple and very accurate procedure) will provide a good indication of the levels of smoke. Indeed, several jurisdictions use CO measurement as the basis of smoke control. However it is far from clear how accurate the correspondence is.
Medicinal smoking
This section relies largely or entirely on a single source. (October 2023) |
Throughout recorded history, humans have used the smoke of
See also
- Air purifier
- Bonfire
- Great Smog of London
- Health impacts of sawdust
- Inversion (meteorology)
- Joss paper
- Open burning of waste
- Smog
- Ultrafine particle
References
- ^ .
- .
- ^ "15.11: Colloids". Chemistry LibreTexts. 27 June 2016. Archived from the original on 23 November 2022. Retrieved 23 November 2022.
- ^ "Smoke Production and Properties" (PDF). SFPE Handbook of Fire Protection Engineering. Archived from the original (PDF) on 21 August 2008.
- ^ The Virginia Journal of Science. Virginia Academy of Science. 1976. Archived from the original on 19 August 2020. Retrieved 5 July 2020.[page needed]
- ISBN 978-0-86587-848-8. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ISBN 978-1-284-05384-5. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ^ ISBN 978-0-03-093893-1. Archivedfrom the original on 4 April 2023. Retrieved 20 October 2020.
- ^ ISBN 978-0-08-045792-5. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ^ ISBN 978-1-85957-001-2. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ^ a b c National Research Council (U.S.). Task Force on Flammability, Smoke, Toxicity and Corrosive Gases of Electric Cable Materials (1978). Flammability, smoke, toxicity, and corrosive gases of electric cable materials: report of the Task Force on Flammability, Smoke, Toxicity, and Corrosive Gases of Electric Cable Materials, National Materials Advisory Board, Commission on Sociotechnical Systems, National Research Council. National Academies. pp. 107–. NAP:15488.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ISBN 978-0-444-82203-1. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ISBN 978-0-444-53113-1. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ISBN 978-1-4097-0169-9. Archivedfrom the original on 4 April 2023. Retrieved 20 October 2020.
- .
- ISBN 978-0-912212-11-1. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- JSTOR 4312673.
- .
- (PDF) from the original on 5 February 2022. Retrieved 25 January 2022.
- ^ ISBN 978-0-387-31235-4. Archivedfrom the original on 2 August 2020. Retrieved 25 September 2016.
- ^ "Organic Speciation International Workshop Synthesis_topic7". Wrapair.org. Archived from the original on 26 July 2017. Retrieved 19 February 2010.
- ISBN 978-0-08-054819-7. Archivedfrom the original on 14 July 2020. Retrieved 25 September 2016.
- PMID 21768054.
- ^ St Cyr, MD, Richard (16 January 2013). "Is PM2.5 From Air Pollution The Same As From Smoking?". My Health Beijing. Archived from the original on 16 February 2021. Retrieved 16 September 2015.
- PMID 7529707.
- ^ General, Surgeon. "The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General" (PDF). U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. Archived from the original (PDF) on 1 February 2017. Retrieved 27 February 2017.
- ^ "Secondhand Smoke". American Cancer Society. Archived from the original on 7 January 2017. Retrieved 11 January 2011.
- ^ "telegram.com – Warehouse Tragedy". Archived from the original on 29 August 2013. Retrieved 28 July 2007.
- ISBN 978-0-12-437160-6. Archivedfrom the original on 16 July 2020. Retrieved 25 September 2016.
- ISSN 2571-6255.
- ^ EPA, NSW. "reducing-wood-smoke-emissions". NSW Environment Protection Authority. Archived from the original on 19 April 2021. Retrieved 2 May 2021.
- ^ a b c "Department of Agriculture, Water and the Environment". Department of Agriculture, Water and the Environment. Archived from the original on 11 May 2019. Retrieved 11 November 2008.
- ^ "Wood smoke major source of pollution in winter" (PDF). ec.europa.eu. 2007. Archived (PDF) from the original on 8 December 2020. Retrieved 2 May 2021.
- ^ "Air pollution and health" (PDF). healthywa.wa.gov.au. Archived from the original on 21 April 2021. Retrieved 2 May 2021.
- ^ S2CID 245103794.
- ^ US EPA, OAR (28 May 2013). "Wood Smoke and Your Health". US EPA. Archived from the original on 14 May 2021. Retrieved 2 May 2021.
- S2CID 40717317.
- ^ Carrington, Damian (16 February 2021). "Wood burning at home now biggest cause of UK particle pollution". The Guardian. Archived from the original on 27 December 2022. Retrieved 13 February 2022.
- ^ a b "Wood burning heaters and your health - Fact sheets". www.health.nsw.gov.au. Archived from the original on 9 November 2022. Retrieved 2 May 2021.
- S2CID 41697986.
- ^ "Wood smoke and your health". Minnesota Pollution Control Agency. 16 November 2009. Archived from the original on 14 May 2021. Retrieved 2 May 2021.
- ^ "Wood stove pollution is a burning issue". www.iatp.org. Archived from the original on 9 December 2020. Retrieved 2 May 2021.
- S2CID 11668269.
- S2CID 108987257.
- ^ "Don't underestimate the health dangers of wood smoke". Archived from the original on 26 March 2021. Retrieved 2 May 2021.
- PMID 32211302.
- PMID 32493753.
- ^ "Air pollution particles in young brains linked to Alzheimer's damage". The Guardian. 6 October 2020. Archived from the original on 22 January 2022. Retrieved 22 January 2022.
- ^ "Air Pollution May Damage People's Brains". The Scientist Magazine®. Archived from the original on 12 May 2021. Retrieved 2 May 2021.
- ^ "Stanford study shows wood smoke can harm the brain". ABC7 San Francisco. 2 December 2017. Archived from the original on 1 August 2021. Retrieved 3 May 2021.
- PMID 27609410.
- PMID 31733973.
- PMID 27653281.
- PMID 26399256.
- S2CID 220942039.
- S2CID 201057595.
- ^ PMID 31850801.
- ^ S2CID 199147061.
- ^ S2CID 231677095.
- from the original on 31 March 2022. Retrieved 25 January 2022.
- PMID 27601646.
- ISBN 978-0-19-046863-7. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ISBN 978-0-323-06985-4. Archivedfrom the original on 3 April 2023. Retrieved 20 October 2020.
- ^ National Academies (1 January 1983). Polycyclic Aromatic Hydrocarbons: Evaluation of Sources and Effects (Report). National Academies. p. 4.
- (PDF) from the original on 7 March 2020. Retrieved 26 July 2019.
- PMID 17030480.
Sources
- . Encyclopædia Britannica. Vol. 25 (11th ed.). 1911.
External links
- 'Fire inversions' lock smoke in valleys
- Burning Issues wood smoke Site
- Shedding new light on wood smoke
- 7 things you need to know about smoke detectors
