Hyaluronan synthase
| Hyaluronan synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Hyaluronan synthases (HAS) are membrane-bound
Isoforms
There are three mammalian hyaluronan synthases described to date -
During the development of the embryo, each isoform is uniquely expressed, both spatially and temporally.
- HAS2 is probably the most important synthase at this time as mice lacking the ability to express HAS2 (
- HAS1 or HAS3 knock-out mice show no effect on foetal development.[7]
The isoforms of HAS also display varying physiological effects and therapeutic potentials. HAS2 is overexpressed in breast cancer cell lines and is associated with lymph node metastasis, while HAS1 and HAS3 lack any correlations with cancer development or metastasis.[8] HAS-2 has also been proposed as a nanotherapeutic agent to combat osteoarthritis in joints displaying synovial inflammation as a result of increased hyaluronan depolymerization.[9] Contrastingly, stimulation of HAS3 has been linked to increased inflammation and atheroprogression by means of increased interleukin release and macrophage activation.[10]
Structure
HAS1 has a single
HAS2 is regulated by
HAS3 is regulated through truncation of the
Mechanism
Initiation of hyaluronan biosynthesis by Class I hyaluronan synthases involves in situ generation of an
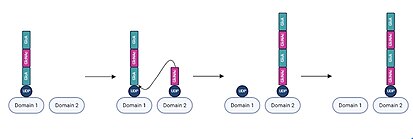
In each iteration of chain elongation, one active site domain is occupied by the existing UDP-hyaluronan chain. A UDP-sugar monomer corresponding to the next unit then binds to the unoccupied active site domain.
HAS1, HAS2, and HAS3 perform functionally equivalent hyaluronan biosyntheses but demonstrate differences in
Role in cancer metastasis
HAS can play roles in all of the stages of cancer metastasis. By producing anti-adhesive HA, HAS can allow tumor cells to release from the primary tumor mass and if HA associates with receptors such as CD44, the activation of Rho GTPases can promote
Increased HA production by cancer cells increases invasive capacity. HA's interaction with CD44 activates focal adhesion kinase (FAK), an important molecule in the process of cell motility by coordinating dissolution of the focal adhesions at the leading edge of the cell and formation at the lagging edge.[26] Another signaling pathway activated by HA's interaction with CD44 is the Akt pathway which leads to expression of osteopontin, a molecule which can stimulate cell migration.[27] The HA produced by HAS also has been suggested to protect the cancer cell from physical damage while in the circulatory or lymphatic systems. This role of HA has been shown in other cell types, but has not yet been researched in cancer cells.[28] The HA produced by HAS up-regulates secretion of various MMPs, proteolytic enzymes that are involved in many stages of the metastatic cascade.[29] Research has shown that the different HASs may impact the metastatic steps in different ways based on the molecular weight and amount of HA they produce.
References
- PMID 35355017.
- PMID 9169154.
- PMID 12512858.
- PMID 10455188.
- PMID 16822580.
- PMID 10930438.
- PMID 15790861.
- PMID 26722395.
- S2CID 202745517.
- PMID 28987865.
- ^ PMID 35355017.
- PMID 12799342.
- ^ PMID 10988250.
- PMID 20507985.
- PMID 35671866.
- PMID 9083017.
- ^ PMID 9206724.
- PMID 22343360.
- S2CID 21344879.
- PMID 6870820.
- S2CID 25537143.
- PMID 10455188.
- PMID 32623953.
- S2CID 53217005.
- PMID 19218337.
- PMID 12297287.
- PMID 19262113.
- S2CID 11765495.
- PMID 19231585.
External links
- EC 2.4.1.212
- hyaluronan+synthase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
