Glycogen phosphorylase
This article needs additional citations for verification. (January 2009) |
| Phosphorylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Glycogen phosphorylase is one of the
Mechanism
Glycogen phosphorylase breaks up glycogen into glucose subunits (see also figure below):
(α-1,4 glycogen chain)n + Pi ⇌ (α-1,4 glycogen chain)n-1 + α-D-glucose-1-phosphate.[2]
Glycogen is left with one fewer glucose
Although the reaction is reversible in vitro, within the cell the enzyme only works in the forward direction as shown below because the concentration of inorganic phosphate is much higher than that of glucose-1-phosphate.[2]
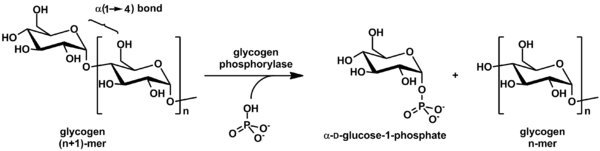
Glycogen phosphorylase can act only on
Glycogen phosphorylase has a pyridoxal phosphate (PLP, derived from Vitamin B6) at each catalytic site. Pyridoxal phosphate links with basic residues (in this case Lys680) and covalently forms a Schiff base. Once the Schiff base linkage is formed, holding the PLP molecule in the active site, the phosphate group on the PLP readily donates a proton to an inorganic phosphate molecule, allowing the inorganic phosphate to in turn be deprotonated by the oxygen forming the α-1,4 glycosidic linkage. PLP is readily deprotonated because its negative charge is not only stabilized within the phosphate group, but also in the pyridine ring, thus the conjugate base resulting from the deprotonation of PLP is quite stable. The protonated oxygen now represents a good leaving group, and the glycogen chain is separated from the terminal glycogen in an SN1 fashion, resulting in the formation of a glucose molecule with a secondary carbocation at the 1 position. Finally, the deprotonated inorganic phosphate acts as a nucleophile and bonds with the carbocation, resulting in the formation of glucose-1-phosphate and a glycogen chain shortened by one glucose molecule.
There is also an alternative proposed mechanism involving a positively charged oxygen in a half-chair conformation.[3]

Structure
The glycogen phosphorylase monomer is a large protein, composed of 842 amino acids with a mass of 97.434

In mammals, the major isozymes of glycogen phosphorylase are found in muscle, liver, and brain. The brain type is predominant in adult brain and embryonic tissues, whereas the liver and muscle types are predominant in adult liver and skeletal muscle, respectively.[5]
The glycogen phosphorylase dimer has many regions of biological significance, including
Perhaps the most important
The allosteric site of AMP binding on muscle isoforms of glycogen phosphorylase are close to the subunit interface just like Ser14. Binding of AMP at this site, corresponding in a change from the T state of the enzyme to the R state, results in small changes in tertiary structure at the subunit interface leading to large changes in quaternary structure.[8] AMP binding rotates the tower helices (residues 262-278) of the two subunits 50˚ relative to one another through greater organization and intersubunit interactions. This rotation of the tower helices leads to a rotation of the two subunits by 10˚ relative to one another, and more importantly disorders residues 282-286 (the 280s loop) that block access to the catalytic site in the T state but do not in the R state.[6]
The final, perhaps most curious site on the glycogen phosphorylase protein is the so-called glycogen storage site. Residues 397-437 form this structure, which allows the protein to covalently bind to the glycogen chain a full 30 Å from the catalytic site . This site is most likely the site at which the enzyme binds to glycogen granules before initiating cleavage of terminal glucose molecules. In fact, 70% of dimeric phosphorylase in the cell exists as bound to glycogen granules rather than free floating.[9]
Clinical significance
| phosphorylase, glycogen; muscle (McArdle syndrome, glycogen storage disease type V) | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | PYGM | ||||||
Chr. 11 q12-q13.2 | |||||||
| |||||||
| phosphorylase, glycogen; liver (Hers disease, glycogen storage disease type VI) | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | PYGL | ||||||
Chr. 14 q11.2-24.3 | |||||||
| |||||||
| phosphorylase, glycogen; brain | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | PYGB | ||||||
Chr. 20 p11.2-p11.1 | |||||||
| |||||||
The inhibition of glycogen phosphorylase has been proposed as one method for treating
Mutations in the muscle isoform of glycogen phosphorylase (PYGM) are associated with glycogen storage disease type V (GSD V, McArdle's Disease). More than 65 mutations in the PYGM gene that lead to McArdle disease have been identified to date.[15][16] Symptoms of McArdle disease include muscle weakness, myalgia, and lack of endurance, all stemming from low glucose levels in muscle tissue.[17]
Mutations in the liver isoform of glycogen phosphorylase (PYGL) are associated with
The brain isoform of glycogen phosphorylase (PYGB) has been proposed as a biomarker for gastric cancer.[21]
Regulation
Glycogen phosphorylase is regulated through
Hormones such as
In the liver, glucagon also activates another GPCR that triggers a different cascade, resulting in the activation of phospholipase C (PLC). PLC indirectly causes the release of calcium from the hepatocytes' endoplasmic reticulum into the cytosol. The increased calcium availability binds to the calmodulin subunit and activates glycogen phosphorylase kinase. Glycogen phosphorylase kinase activates glycogen phosphorylase in the same manner mentioned previously.
Glycogen phosphorylase b is not always inactive in muscle, as it can be activated allosterically by AMP.[6][9] An increase in AMP concentration, which occurs during strenuous exercise, signals energy demand. AMP activates glycogen phosphorylase b by changing its conformation from a tense to a relaxed form. This relaxed form has similar enzymatic properties as the phosphorylated enzyme. An increase in ATP concentration opposes this activation by displacing AMP from the nucleotide binding site, indicating sufficient energy stores.
Upon eating a meal, there is a release of insulin, signaling glucose availability in the blood. Insulin indirectly activates protein phosphatase 1 (PP1) and phosphodiesterase via a signal transduction cascade. PP1 dephosphorylates glycogen phosphorylase a, reforming the inactive glycogen phosphorylase b. The phosphodiesterase converts cAMP to AMP. Together, they decrease the concentration of cAMP and inhibit PKA. As a result, PKA can no longer initiate the phosphorylation cascade that ends with formation of (active) glycogen phosphorylase a. Overall, insulin signaling decreases glycogenolysis to preserve glycogen stores in the cell and triggers glycogenesis.[22]
Historical significance
Glycogen phosphorylase was the first allosteric enzyme to be discovered. Arda Green and Gerty Cori crystallized it for the first time in 1943[25] and illustrated that glycogen phosphorylase existed in either the a or b forms depending on its phosphorylation state, as well as in the R or T states based on the presence of AMP.[26]
See also
- AMP deaminase deficiency (MADD)
- Glycogenolysis
- McArdle disease (GSD-V)
- Metabolic myopathies
- Purine nucleotide cycle § Pathology
References
- ^ PDB: 3E3N
- ^ S2CID 12036788.
- PMID 2182117.
- PMID 1566331.
- PMID 3510670.
- ^ S2CID 25954545.
- PMID 2667896.
- ^ PMID 2137445.
- ^ PMID 4320610.
- PMID 12769745.
- S2CID 4426975.
- PMID 1874749.
- S2CID 11168623.
- .
- S2CID 39626196.
- PMID 17915571.
- PMID 4502558.
- PMID 9529348.
- PMID 9536091.
- PMID 12809646.
- S2CID 25602637.
- PMID 3007140.
- PMID 17731092.
- .
- .
- .
Further reading
- Voet JG, Voet D (1995). "Chapter 17: Glycogen Metabolism". Biochemistry (2nd ed.). New York: J. Wiley & Sons. ISBN 978-0-471-58651-7.
- Voet JG, Voet D (2004). "Chapter 18: Glycogen Metabolism". Biochemistry (3rd ed.). New York: J. Wiley & Sons. ISBN 978-0-471-19350-0.
- Goodsell DS (2001-12-01). "Glycogen Phosphorylase". Molecule of the Month. RCSB Protein Data Bank. Archived from the original on 2010-07-21. Retrieved 2009-01-10.
- Diwan JJ. "Glycogen Metabolism". Molecular Biochemistry I. Rensselaer Polytechnic Institute. Archived from the original on 2009-01-25. Retrieved 2009-01-10.
External links
- GeneReviews/NCBI/NIH/UW entry on Glycogen Storage Disease Type VI - Hers disease
- Glycogen+phosphorylase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P11217 (Human muscle Glycogen phosphorylase) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: P06737 (Human liver Glycogen phosphorylase) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: P11216 (Human brain Glycogen phosphorylase) at the PDBe-KB.
