Phosphoglycerate mutase

| Phosphoglycerate mutase family | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||
| Symbol | PGAM | ||||||||||
SCOP2 | 3pgm / SCOPe / SUPFAM | ||||||||||
| |||||||||||
| phosphoglycerate mutase 1 (brain) | |||||||
|---|---|---|---|---|---|---|---|
Chr. 10 q25.3 | |||||||
| |||||||
| phosphoglycerate mutase 2 (muscle) | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | PGAM2 | ||||||
Chr. 7 p13-p12 | |||||||
| |||||||
- This enzyme is not to be confused with 2,3-bisphosphoglycerate.
Phosphoglycerate mutase (PGM) is any
Mechanism
PGM is an
The catalyzed mutase reaction involves two separate phosphoryl groups and the ending phosphate on the 2-carbon is not the same phosphate removed from the 3-carbon.
In the cofactor-dependent
Reaction summary
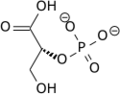
3PG + P-Enzyme → 2,3BPG + Enzyme → 2PG + P-Enzyme
3-phosphoglycerate intermediate 2-phosphoglycerate
ΔG°′=+1.1kcal/mol
-
3PG
-
2,3BPG
-
2PG
Isozymes
Phosphoglycerate mutase exists primarily as a dimer of two either identical or closely related subunits of about 32kDa. The enzyme is found in organisms as simple as yeast through Homo sapiens and its structure is highly conserved throughout. (Yeast PGM≈74% conserved vs mammal form).
In mammals, the enzyme subunits appear to be either a muscle-derived form (m-type) or other tissue (b-type for brain where the b-isozyme was originally isolated). Existing as a dimer, the enzyme then has 3 isozymes depending on which subunit forms makeup the whole molecule (mm, bb or mb). The mm-type is found mainly in smooth muscle almost exclusively. The mb-isozyme is found in cardiac and skeletal muscle and the bb-type is found in the rest of tissues.[11] While all three isozymes may be found in any tissue, the above distributions are based on prevalence in each.
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Regulation
Phosphoglycerate mutase has a small positive Gibbs free energy and this reaction proceeds easily in both directions. Since it is a reversible reaction, it is not the site of major regulation mechanisms or regulation schemes for the glycolytic pathway.
Clinical significance
In humans the PGAM2 gene which encodes this enzyme is located on the short arm of chromosome 7.
Deficiency of phosphoglycerate mutase causes
Onset is generally noted as childhood to early adult though some who may be mildly affected by the disorder may not know they have it. Patients with PGAM deficiency are usually asymptomatic, except when they engage in brief, strenuous efforts which may trigger myalgias, cramps, muscle necrosis and myoglobinuria.[15] An unusual pathologic feature of PGAM deficiency is the association with tubular aggregates. The symptoms are an intolerance to physical exertion or activity, cramps and muscle pain. Permanent weakness is rare. The disease is not progressive and has an excellent prognosis.[citation needed]
Human proteins containing this domain
References
External links
- Phosphoglycerate+Mutase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Phosphoglycerate mutase 1