Glyceraldehyde 3-phosphate dehydrogenase
Ensembl |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) |
| ||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 12: 6.53 – 6.54 Mb | n/a | |||||||
| PubMed search | [2] | [3] | |||||||
| View/Edit Human | View/Edit Mouse |
| Glyceraldehyde 3-phosphate dehydrogenase, NAD binding domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
| Glyceraldehyde 3-phosphate dehydrogenase, C-terminal domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
Glyceraldehyde 3-phosphate dehydrogenase (abbreviated GAPDH) (
Structure
Under normal cellular conditions,
GAPDH is encoded by a single gene that produces a single mRNA transcript with 8 splice variants, though an isoform does exist as a separate gene that is expressed only in
Reaction
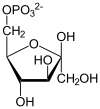
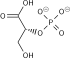
| glyceraldehyde 3-phosphate | glyceraldehyde phosphate dehydrogenase
|
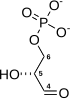
D- glycerate 1,3-bisphosphate
| |

|
| ||
| NAD+ +Pi | NADH + H+ | ||
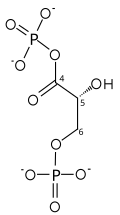
| |||
| NAD+ +Pi | NADH + H+ | ||
Compound C00118 at KEGG Pathway Database. Enzyme 1.2.1.12 at KEGG Pathway Database. Reaction R01063 at KEGG Pathway Database. Compound C00236 at KEGG Pathway Database.
Two-step conversion of G3P
The first reaction is the oxidation of glyceraldehyde 3-phosphate (G3P) at the position-1 (in the diagram it is shown as the 4th carbon from glycolysis), in which an aldehyde is converted into a carboxylic acid (ΔG°'=-50 kJ/mol (−12kcal/mol)) and NAD+ is simultaneously reduced endergonically to NADH.
The energy released by this highly
This is an example of phosphorylation coupled to oxidation, and the overall reaction is somewhat endergonic (ΔG°'=+6.3 kJ/mol (+1.5)). Energy coupling here is made possible by GAPDH.
Mechanism
GAPDH uses covalent catalysis and general base catalysis to decrease the very large activation energy of the second step (phosphorylation) of this reaction.
1: Oxidation
First, a cysteine residue in the active site of GAPDH attacks the carbonyl group of G3P, creating a hemithioacetal intermediate (covalent catalysis).
The hemithioacetal is deprotonated by a
Next, an adjacent, tightly bound molecule of
This thioester species is much higher in energy (less stable) than the carboxylic acid species that would result if G3P were oxidized in the absence of GAPDH (the carboxylic acid species is so low in energy that the energy barrier for the second step of the reaction (phosphorylation) would be too high, and the reaction, therefore, too slow and unfavorable for a living organism).
2: Phosphorylation
NADH leaves the active site and is replaced by another molecule of NAD+, the positive charge of which stabilizes the negatively charged carbonyl oxygen in the transition state of the next and ultimate step. Finally, a molecule of
Regulation
This protein may use the morpheein model of allosteric regulation.[8]
Function
Metabolic
As its name indicates, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) catalyses the conversion of
Adhesion
One of the GAPDH moonlighting functions is its role in adhesion and binding to other partners. Bacterial GAPDH from Mycoplasma and Streptococcus and fungal GAPDH from Paracoccidioides brasiliensis are known to bind with the human extracellular matrix component and act in adhesion.[9][10][11] GAPDH is found to be surface bound contributing in adhesion and also in competitive exclusion of harmful pathogens.[12] GAPDH from Candida albicans is found to cell-wall associated and binds to Fibronectin and Laminin.[13] GAPDH from probiotics species are known to bind human colonic mucin and ECM, resulting in enhanced colonization of probiotics in the human gut.[14][15][16] Patel D. et al., showed that Lactobacillus acidophilus GAPDH binds with mucin, acting in adhesion.[17]
Transcription and apoptosis
GAPDH can itself activate
In 2005, Hara et al. showed that GAPDH initiates
Metabolic switch
GAPDH acts as a reversible metabolic switch under oxidative stress.
ER-to-Golgi transport
GAPDH also appears to be involved in the
Additional functions
GAPDH, like many other enzymes, has multiple functions. In addition to catalysing the 6th step of glycolysis, recent evidence implicates GAPDH in other cellular processes. GAPDH has been described to exhibit higher order multifunctionality in the context of maintaining cellular iron homeostasis,[24] specifically as a chaperone protein for labile heme within cells.[25] This came as a surprise to researchers but it makes evolutionary sense to re-use and adapt existing proteins instead of evolving a novel protein from scratch.
Use as loading control
Because the GAPDH gene is often stably and constitutively expressed at high levels in most tissues and cells, it is considered a
Cellular distribution
All steps of glycolysis take place in the
Clinical significance
Cancer
GAPDH is overexpressed in multiple human cancers, such as cutaneous
Neurodegeneration
GAPDH has been implicated in several neurodegenerative diseases and disorders, largely through interactions with other proteins specific to that disease or disorder. These interactions may affect not only energy metabolism but also other GAPDH functions.
Interactions
Protein binding partners
GAPDH participates in a number of biological functions through its
- tubulin to facilitate microtubule bundling;[6]
- actin to facilitate actin polymerization;[6]
- membrane permeabilization (MMP) and apoptosis;[6]
- Inositol 1,4,5-trisphosphate receptor to regulate intracellular
- coactivator complex OCA-S, which is required for histone H2B synthesis during S phase of the cell cycle;[7]
- p22 to aid microtubule organization;[7]
- Rab2 to facilitate endoplasmic reticulum (ER)–golgi transport;[7]
- Transferrin on the surface of diverse cells and in extracellular fluid;[7][31][37]
- Lactate dehydrogenase;[7]
- Lactoferrin;[38]
- Apurinic/apyrimidinic endonuclease (APE1), thus converting oxidized APE1 to its reduced form, to restart its endonuclease activity;[7]
- Promyelocytic leukaemia protein (PML) in an RNA-dependent fashion;[7]
- Siah1 to form a complex that translocates to the nucleus, where it ubiquitinates and degrades nuclear proteins during nitrosative stress conditions;[7]
- GAPDH's competitor of Siah protein enhances life (GOSPEL) to block GAPDH interaction with Siah1 and, thus, cell death in response to oxidative stress;[7]
- p300/acetylates GAPDH and, in turn, enhances the acetylation of additional apoptotic targets;[7]
- skeletal muscle-specific Ca2+/calmodulin-dependent protein kinase;[7]
- Akt;[7]
- Beta-amyloid precursor protein (betaAPP);[35]
- Huntingtin.[35]
- GAPDHcan self-associate into homotypic oligomers/aggregates
Nucleic acid binding partners
GAPDH binds to single-stranded RNA [39] and DNA and a number of nucleic acid binding partners have been identified:[7]
- tRNA,
- Hepatitis A viral RNA,
- Hepatitis B viral RNA,
- Hepatitis C viral RNA,
- HPIV3,
- lymphokine mRNA,
- IFN-γmRNA,
- JEVmRNA, and
- telomeric DNA.
Inhibitors
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000111640 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- S2CID 20291542.
- PMID 23374344.
- ^ PMID 20727968.
- ^ S2CID 23499684.
- PMID 22182754.
- PMID 21546586.
- PMID 15288930.
- PMID 16368993.
- PMID 18619532.
- PMID 9573088.
- PMID 33371288.
- S2CID 22346488.
- PMID 18790050.
- PMID 27180300.
- S2CID 5543647.
- S2CID 1922911.
- PMID 16505364.
- PMID 23064950.
- PMID 18154684.
- PMID 17488287.
- PMID 25399609.
- PMID 30012884.
- PMID 15769908.
- PMID 25065746.
- S2CID 85327763.
- PMID 15701694.
- PMID 25286305.
- ^ PMID 22062951.
- PMID 25550585.
- PMID 23620736.
- PMID 21749859.
- ^ PMID 12428732.
- PMID 20864222.
- PMID 17121833.
- PMID 26672975.
- PMID 25451934.
Further reading
- Voet D, Voet JG (2010). Biochemistry. New York: Wiley. ISBN 978-0-470-57095-1.
- Stryer L, Berg JM, Tymoczko JL (2002). Biochemistry, Fifth Edition & Lecture Notebook. San Francisco: W. H. Freeman. ISBN 978-0-7167-9804-0.
- diagram of the GAPDH reaction mechanism from Lodish MCB at NCBI bookshelf
- similar diagram from Alberts The Cell at NCBI bookshelf
External links
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Glyceraldehyde-3-phosphate dehydrogenase