Physics of magnetic resonance imaging
Certain atomic nuclei are able to absorb and emit radio frequency energy when placed in an external magnetic field. In clinical and research MRI, hydrogen atoms are most often used to generate a detectable radio-frequency signal that is received by antennas close to the anatomy being examined. Hydrogen atoms are naturally abundant in people and other biological organisms, particularly in water and fat. For this reason, most MRI scans essentially map the location of water and fat in the body. Pulses of radio waves excite the nuclear spin energy transition, and magnetic field gradients localize the signal in space. By varying the parameters of the pulse sequence, different contrasts may be generated between tissues based on the relaxation properties of the hydrogen atoms therein.
When inside the magnetic field (B0) of the scanner, the
History
The MRI scanner was developed from 1975 to 1977 at the University of Nottingham by Prof Raymond Andrew FRS FRSE following from his research into nuclear magnetic resonance. The full body scanner was created in 1978.[1]
Nuclear magnetism
Subatomic particles have the
When these spins are placed in a strong external magnetic field they precess around an axis along the direction of the field. Protons align in two energy eigenstates (the Zeeman effect): one low-energy and one high-energy, which are separated by a very small splitting energy.
Resonance and relaxation
Quantum mechanics is required to accurately model the behaviour of a single proton. However, classical mechanics can be used to describe the behaviour of an ensemble of protons adequately. As with other spin particles, whenever the spin of a single proton is measured it can only have one of two results commonly called parallel and anti-parallel. When we discuss the state of a proton or protons we are referring to the wave function of that proton which is a linear combination of the parallel and anti-parallel states.[3]
In the presence of the magnetic field, B0, the protons will appear to precess at the
The net longitudinal magnetization in thermodynamic equilibrium is due to a tiny excess of protons in the lower energy state. This gives a net polarization that is parallel to the external field. Application of an RF pulse can tip this net polarization vector sideways (with, i.e., a so-called 90° pulse), or even reverse it (with a so-called 180° pulse). The protons will come into phase with the RF pulse and therefore each other.[citation needed]
The recovery of longitudinal magnetization is called longitudinal or T1 relaxation and occurs exponentially with a time constant T1. The loss of phase coherence in the transverse plane is called transverse or T2 relaxation. T1 is thus associated with the enthalpy of the spin system, or the number of nuclei with parallel versus anti-parallel spin. T2 on the other hand is associated with the entropy of the system, or the number of nuclei in phase.
When the radio frequency pulse is turned off, the transverse vector component produces an oscillating magnetic field which induces a small current in the receiver coil. This signal is called the free induction decay (FID). In an idealized nuclear magnetic resonance experiment, the FID decays approximately exponentially with a time constant T2. However, in practical MRI there are small differences in the static magnetic field at different spatial locations ("inhomogeneities") that cause the Larmor frequency to vary across the body. This creates destructive interference, which shortens the FID. The time constant for the observed decay of the FID is called the T*
2 relaxation time, and is always shorter than T2. At the same time, the longitudinal magnetization starts to recover exponentially with a time constant T1 which is much larger than T2 (see below).
In MRI, the static magnetic field is augmented by a
Typically, in soft tissues T1 is around one second while T2 and T*
2 are a few tens of milliseconds. However, these values can vary widely between different tissues, as well as between different external magnetic fields. This behavior is one factor giving MRI its tremendous soft tissue contrast.
MRI contrast agents, such as those containing Gadolinium(III) work by altering (shortening) the relaxation parameters, especially T1.
Imaging
Imaging schemes
A number of schemes have been devised for combining field gradients and radio frequency excitation to create an image:
- computed tomography.
- Building the image point-by-point or line-by-line.
- Gradients in the RF field rather than the static field.
Although each of these schemes is occasionally used in specialist applications, the majority of MR Images today are created either by the two-dimensional Fourier transform (2DFT) technique with slice selection, or by the three-dimensional Fourier transform (3DFT) technique. Another name for 2DFT is spin-warp. What follows here is a description of the 2DFT technique with slice selection.
The 3DFT technique is rather similar except that there is no slice selection and phase-encoding is performed in two separate directions.
Echo-planar imaging
Another scheme which is sometimes used, especially in brain scanning or where images are needed very rapidly, is called echo-planar imaging (EPI):[5] In this case, each RF excitation is followed by a train of gradient echoes with different spatial encoding. Multiplexed-EPI is even faster, e.g., for whole brain functional MRI (fMRI) or diffusion MRI.[6]
Image contrast and contrast enhancement
Image
2 and so can be encoded with suitable pulse sequences.
In some situations it is not possible to generate enough image contrast to adequately show the
) is given. Gadolinium-enhanced tissues and fluids appear extremely bright on T1-weighted images. This provides high sensitivity for detection of vascular tissues (e.g., tumors) and permits assessment of brain perfusion (e.g., in stroke). There have been concerns raised recently regarding the toxicity of gadolinium-based contrast agents and their impact on persons with impaired kidney function. (See Safety/Contrast agents below.)More recently, superparamagnetic contrast agents, e.g., iron oxide nanoparticles,[9][10] have become available. These agents appear very dark on T*
2-weighted images and may be used for liver imaging, as normal liver tissue retains the agent, but abnormal areas (e.g., scars, tumors) do not. They can also be taken orally, to improve visualization of the gastrointestinal tract, and to prevent water in the gastrointestinal tract from obscuring other organs (e.g., the pancreas). Diamagnetic agents such as barium sulfate have also been studied for potential use in the gastrointestinal tract, but are less frequently used.
k-space
In 1983, Ljunggren[11] and Twieg[12] independently introduced the k-space formalism, a technique that proved invaluable in unifying different MR imaging techniques. They showed that the demodulated MR signal S(t) generated by the interaction between an ensemble of freely precessing nuclear spins in the presence of a linear magnetic field gradient G and a receiver-coil equals the Fourier transform of the effective spin density, . Fundamentally, the signal is derived from Faraday's law of induction:
where:
In other words, as time progresses the signal traces out a trajectory in k-space with the
From the basic k-space formula, it follows immediately that we reconstruct an image by taking the
Using the k-space formalism, a number of seemingly complex ideas became simple. For example, it becomes very easy (for physicists, in particular) to understand the role of phase encoding (the so-called spin-warp method). In a standard spin echo or gradient echo scan, where the readout (or view) gradient is constant (e.g., G), a single line of k-space is scanned per RF excitation. When the phase encoding gradient is zero, the line scanned is the kx axis. When a non-zero phase-encoding pulse is added in between the RF excitation and the commencement of the readout gradient, this line moves up or down in k-space, i.e., we scan the line ky = constant.
The k-space formalism also makes it very easy to compare different scanning techniques. In single-shot
The importance of the center of k-space in determining image contrast can be exploited in more advanced imaging techniques. One such technique is spiral acquisition—a rotating magnetic field gradient is applied, causing the trajectory in k-space to spiral out from the center to the edge. Due to T2 and T*
2 decay the signal is greatest at the start of the acquisition, hence acquiring the center of k-space first improves
Since and are conjugate variables (with respect to the Fourier transform) we can use the
(These relationships apply to each axis independently.)
Example of a pulse sequence

In the
The first part of the pulse sequence, SS, achieves "slice selection". A shaped pulse (shown here with a
The longitudinal magnetisation is then allowed to recover somewhat and after a time TR the whole sequence is repeated nPE times, but with the phase-encoding gradient incremented (indicated by the horizontal hatching in the green gradient block). Typically nPE of between 128 and 512 repetitions are made.
The negative-going lobes in GX and GZ are imposed to ensure that, at time TE (the spin echo maximum), phase only encodes spatial location in the Y direction.
Typically TE is between 5 ms and 100 ms, while TR is between 100 ms and 2000 ms.
After the two-dimensional matrix (typical dimension between 128 × 128 and 512 × 512) has been acquired, producing the so-called k-space data, a two-dimensional inverse Fourier transform is performed to provide the familiar MR image. Either the magnitude or phase of the Fourier transform can be taken, the former being far more common.
Overview of main sequences
This table does not include
| Group | Sequence | Abbr. | Physics | Main clinical distinctions | Example |
|---|---|---|---|---|---|
| Spin echo | T1 weighted | T1 | Measuring echo time (TE).
|
Standard foundation and comparison for other sequences |

|
| T2 weighted | T2 | Measuring spin–spin relaxation by using long TR and TE times |
Standard foundation and comparison for other sequences |

| |
Proton density weighted |
PD | Long TE (to minimize T2).[16]
|
Joint disease and injury.[17]
|

| |
| Gradient echo (GRE) | Steady-state free precession | SSFP | Maintenance of a steady, residual transverse magnetisation over successive cycles.[19] | Creation of cardiac MRI videos (pictured).[19] | 
|
| Effective T2 or "T2-star" |
T2* | Spoiled gradient recalled echo (GRE) with a long echo time and small flip angle[20] | Low signal from hemosiderin deposits (pictured) and hemorrhages.[20] | 
| |
| Susceptibility-weighted | SWI | Spoiled gradient recalled echo (GRE), fully flow compensated, long echo time, combines phase image with magnitude image[21] | Detecting small amounts of hemorrhage (diffuse axonal injury pictured) or calcium.[21] | 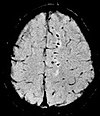
| |
| Inversion recovery | Short tau inversion recovery | STIR | Fat suppression by setting an inversion time where the signal of fat is zero.[22]
|
High signal in edema, such as in more severe stress fracture.[23] Shin splints pictured: | 
|
| Fluid-attenuated inversion recovery | FLAIR | Fluid suppression by setting an inversion time that nulls fluids | High signal in lacunar infarction, multiple sclerosis (MS) plaques, subarachnoid haemorrhage and meningitis (pictured).[24] | 
| |
| Double inversion recovery | DIR | Simultaneous suppression of cerebrospinal fluid and white matter by two inversion times.[25] | High signal of multiple sclerosis plaques (pictured).[25] | 
| |
Diffusion weighted (DWI) |
Conventional | DWI | Measure of Brownian motion of water molecules.[26] | High signal within minutes of cerebral infarction (pictured).[27] | 
|
Apparent diffusion coefficient |
ADC | Reduced T2 weighting by taking multiple conventional DWI images with different DWI weighting, and the change corresponds to diffusion.[28] | Low signal minutes after cerebral infarction (pictured).[29] | 
| |
| Diffusion tensor | DTI | Mainly tractography (pictured) by an overall greater Brownian motion of water molecules in the directions of nerve fibers.[30] |
|
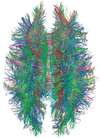
| |
| Perfusion weighted (PWI) | Dynamic susceptibility contrast |
DSC | Measures changes over time in susceptibility-induced signal loss due to gadolinium contrast injection.[32]
|
|

|
| Arterial spin labelling | ASL | Magnetic labeling of arterial blood below the imaging slab, which subsequently enters the region of interest.[34] It does not need gadolinium contrast.[35] | |||
Dynamic contrast enhanced |
DCE | Measures changes over time in the shortening of the gadolinium contrast bolus.[36]
|
Faster Gd contrast uptake along with other features is suggestive of malignancy (pictured).[37] | 
| |
| Functional MRI (fMRI) | Blood-oxygen-level dependent imaging |
BOLD | Changes in oxygen saturation-dependent magnetism of hemoglobin reflects tissue activity.[38] | Localizing brain activity from performing an assigned task (e.g. talking, moving fingers) before surgery, also used in research of cognition.[39] | 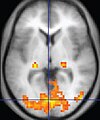
|
| Magnetic resonance angiography (MRA) and venography | Time-of-flight | TOF | Blood entering the imaged area is not yet magnetically saturated, giving it a much higher signal when using short echo time and flow compensation. | Detection of dissection[40]
|

|
| Phase-contrast magnetic resonance imaging | PC-MRA | Two gradients with equal magnitude, but opposite direction, are used to encode a phase shift, which is proportional to the velocity of spins.[41]
|
Detection of dissection (pictured).[40]
|
VIPR )
|
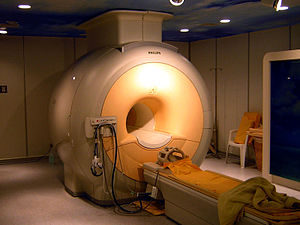
MRI scanner
Construction and operation

The major components of an MRI scanner are: the main magnet, which polarizes the sample, the shim coils for correcting inhomogeneities in the main magnetic field, the gradient system which is used to localize the MR signal and the RF system, which excites the sample and detects the resulting NMR signal. The whole system is controlled by one or more computers.
Magnet

The magnet is the largest and most expensive component of the scanner, and the remainder of the scanner is built around it. The strength of the magnet is measured in teslas (T). Clinical magnets generally have a field strength in the range 0.1–3.0 T, with research systems available up to 9.4 T for human use and 21 T for animal systems.[42] In the United States, field strengths up to 7 T have been approved by the FDA for clinical use.[43]
Just as important as the strength of the main magnet is its precision. The straightness of the magnetic lines within the center (or, as it is technically known, the iso-center) of the magnet needs to be near-perfect. This is known as homogeneity. Fluctuations (inhomogeneities in the field strength) within the scan region should be less than three parts per million (3 ppm). Three types of magnets have been used:
- Permanent magnet: Conventional magnets made from ferromagnetic materials (e.g., steel alloys containing rare-earth elements such as neodymium) can be used to provide the static magnetic field. A permanent magnet that is powerful enough to be used in an MRI will be extremely large and bulky; they can weigh over 100 tonnes. Permanent magnet MRIs are very inexpensive to maintain; this cannot be said of the other types of MRI magnets, but there are significant drawbacks to using permanent magnets. They are only capable of achieving weak field strengths compared to other MRI magnets (usually less than 0.4 T) and they are of limited precision and stability. Permanent magnets also present special safety issues; since their magnetic fields cannot be "turned off," ferromagnetic objects are virtually impossible to remove from them once they come into direct contact. Permanent magnets also require special care when they are being brought to their site of installation.
- Resistive electromagnet: A solenoid wound from copper wire is an alternative to a permanent magnet. An advantage is low initial cost, but field strength and stability are limited. The electromagnet requires considerable electrical energy during operation which can make it expensive to operate. This design is essentially obsolete.
- superconductor, losing resistance to flow of electric current. An electromagnet constructed with superconductors can have extremely high field strengths, with very high stability. The construction of such magnets is extremely costly, and the cryogenic helium is expensive and difficult to handle. However, despite their cost, helium cooled superconducting magnets are the most common type found in MRI scanners today.
Most superconducting magnets have their coils of superconductive wire immersed in liquid helium, inside a vessel called a
Magnets are available in a variety of shapes. However, permanent magnets are most frequently C-shaped, and superconducting magnets most frequently cylindrical. C-shaped superconducting magnets and box-shaped permanent magnets have also been used.
Magnetic field strength is an important factor in determining image quality. Higher magnetic fields increase signal-to-noise ratio, permitting higher resolution or faster scanning. However, higher field strengths require more costly magnets with higher maintenance costs, and have increased safety concerns. A field strength of 1.0–1.5 T is a good compromise between cost and performance for general medical use. However, for certain specialist uses (e.g., brain imaging) higher field strengths are desirable, with some hospitals now using 3.0 T scanners.


Shims
When the MR scanner is placed in the hospital or clinic, its main magnetic field is far from being homogeneous enough to be used for scanning. That is why before doing fine tuning of the field using a sample, the magnetic field of the magnet must be measured and shimmed.
After a sample is placed into the scanner, the main magnetic field is distorted by
After placing the sample in the scanner, the
Gradients
Gradient coils are used to spatially encode the positions of protons by varying the magnetic field linearly across the imaging volume. The Larmor frequency will then vary as a function of position in the x, y and z-axes.
Gradient coils are usually resistive electromagnets powered by sophisticated
Scan speed is dependent on performance of the gradient system. Stronger gradients allow for faster imaging, or for higher resolution; similarly, gradient systems capable of faster switching can also permit faster scanning. However, gradient performance is limited by safety concerns over nerve stimulation.
Some important characteristics of gradient amplifiers and gradient coils are slew rate and gradient strength. As mentioned earlier, a gradient coil will create an additional, linearly varying magnetic field that adds or subtracts from the main magnetic field. This additional magnetic field will have components in all 3 directions, viz. x, y and z; however, only the component along the magnetic field (usually called the z-axis, hence denoted Gz) is useful for imaging. Along any given axis, the gradient will add to the magnetic field on one side of the zero position and subtract from it on the other side. Since the additional field is a gradient, it has units of gauss per centimeter or millitesla per meter (mT/m). High performance gradient coils used in MRI are typically capable of producing a gradient magnetic field of approximate 30 mT/m or higher for a 1.5 T MRI. The slew rate of a gradient system is a measure of how quickly the gradients can be ramped on or off. Typical higher performance gradients have a slew rate of up to 100–200 T·m−1·s−1. The slew rate depends both on the gradient coil (it takes more time to ramp up or down a large coil than a small coil) and on the performance of the gradient amplifier (it takes a lot of voltage to overcome the inductance of the coil) and has significant influence on image quality.
Radio frequency system
The
The receiver consists of the coil, pre-amplifier and signal processing system. The RF electromagnetic radiation produced by nuclear relaxation inside the subject is true EM radiation (radio waves), and these leave the subject as RF radiation, but they are of such low power as to also not cause appreciable RF interference that can be picked up by nearby radio tuners (in addition, MRI scanners are generally situated in metal mesh lined rooms which act as Faraday cages.)
While it is possible to scan using the integrated coil for RF transmission and MR signal reception, if a small region is being imaged, then better image quality (i.e., higher signal-to-noise ratio) is obtained by using a close-fitting smaller coil. A variety of coils are available which fit closely around parts of the body such as the head, knee, wrist, breast, or internally, e.g., the rectum.
A recent development in MRI technology has been the development of sophisticated multi-element phased array[52] coils which are capable of acquiring multiple channels of data in parallel. This 'parallel imaging' technique uses unique acquisition schemes that allow for accelerated imaging, by replacing some of the spatial coding originating from the magnetic gradients with the spatial sensitivity of the different coil elements. However, the increased acceleration also reduces the signal-to-noise ratio and can create residual artifacts in the image reconstruction. Two frequently used parallel acquisition and reconstruction schemes are known as SENSE[53] and GRAPPA.[54] A detailed review of parallel imaging techniques can be found here:[55]
References
- ^ Independent (newspaper) obituary of R Edward 20 July 2001
- ^
Callaghan P (1994). Principles of Nuclear Magnetic Resonance Microscopy. ISBN 978-0-19-853997-1.
- ^ "Quantum philosophy". Questions and Answers in MRI. Retrieved 1 June 2019.
- ISBN 978-3-662-07805-1.
- PMID 11353123.
- PMID 21187930.
- ^
Weinmann HJ, Brasch RC, Press WR, Wesbey GE (March 1984). "Characteristics of gadolinium-DTPA complex: a potential NMR contrast agent". AJR. American Journal of Roentgenology. 142 (3): 619–24. PMID 6607655.
- ^
Laniado M, Weinmann HJ, Schörner W, Felix R, Speck U (1984). "First use of GdDTPA/dimeglumine in man". Physiological Chemistry and Physics and Medical NMR. 16 (2): 157–65. PMID 6505042.
- ^
Widder DJ, Greif WL, Widder KJ, Edelman RR, Brady TJ (February 1987). "Magnetite albumin microspheres: a new MR contrast material". AJR. American Journal of Roentgenology. 148 (2): 399–404. PMID 3492120.
- ^
Weissleder R, Elizondo G, Wittenberg J, Rabito CA, Bengele HH, Josephson L (May 1990). "Ultrasmall superparamagnetic iron oxide: characterization of a new class of contrast agents for MR imaging". Radiology. 175 (2): 489–93. PMID 2326474.
- .
- PMID 6646065.
- ^ University of Wisconsin. Archived from the originalon 10 May 2017. Retrieved 14 March 2016.
- ^ a b c d Johnson KA. "Basic proton MR imaging. Tissue Signal Characteristics". Harvard Medical School. Archived from the original on 5 March 2016. Retrieved 14 March 2016.
- ^ "MRI Questions, Fast Spin Echo". MRIQuestions.com. Retrieved 18 May 2021.
- ISBN 978-0-7020-4614-8.}
- ^ du Plessis V, Jones J. "MRI sequences (overview)". Radiopaedia. Retrieved 13 January 2017.
- PMID 27057352.
- ^ a b Luijkx T, Weerakkody Y. "Steady-state free precession MRI". Radiopaedia. Retrieved 13 October 2017.
- ^ PMID 19755604.
- ^ a b Di Muzio B, Gaillard F. "Susceptibility weighted imaging". Retrieved 15 October 2017.
- ^ Sharma R, Taghi Niknejad M. "Short tau inversion recovery". Radiopaedia. Retrieved 13 October 2017.
- ^ Berger F, de Jonge M, Smithuis R, Maas M. "Stress fractures". Radiology Assistant. Radiology Society of the Netherlands. Retrieved 13 October 2017.
- ^ Hacking C, Taghi Niknejad M, et al. "Fluid attenuation inversion recoveryg". radiopaedia.org. Retrieved 3 December 2015.
- ^ a b Di Muzio B, Abd Rabou A. "Double inversion recovery sequence". Radiopaedia. Retrieved 13 October 2017.
- ^ Lee M, Bashir U. "Diffusion weighted imaging". Radiopaedia. Retrieved 13 October 2017.
- ^ Weerakkody Y, Gaillard F. "Ischaemic stroke". Radiopaedia. Retrieved 15 October 2017.
- ^ Hammer M. "MRI Physics: Diffusion-Weighted Imaging". XRayPhysics. Retrieved 15 October 2017.
- PMID 21454821.
- ^ a b Smith D, Bashir U. "Diffusion tensor imaging". Radiopaedia. Retrieved 13 October 2017.
- S2CID 24731783.
- ^ Gaillard F. "Dynamic susceptibility contrast (DSC) MR perfusion". Radiopaedia. Retrieved 14 October 2017.
- PMID 22468186.
- ^ "Arterial spin labeling". University of Michigan. Retrieved 27 October 2017.
- ^ Gaillard F. "Arterial spin labelling (ASL) MR perfusion". Radiopaedia. Retrieved 15 October 2017.
- ^ Gaillard F. "Dynamic contrast enhanced (DCE) MR perfusion". Radiopaedia. Retrieved 15 October 2017.
- S2CID 5305422.
- ^ Chou Ih. "Milestone 19: (1990) Functional MRI". Nature. Retrieved 9 August 2013.
- ^ Luijkx T, Gaillard F. "Functional MRI". Radiopaedia. Retrieved 16 October 2017.
- ^ a b "Magnetic Resonance Angiography (MRA)". Johns Hopkins Hospital. Retrieved 15 October 2017.
- ^ Keshavamurthy J, Ballinger R et al. "Phase contrast imaging". Radiopaedia. Retrieved 15 October 2017.
- ^ Schepkin, Victor D.; Grant, Samuel C.; Cross, Timothy A. "In vivo MR Imaging at 21.1 T" (PDF). Archived from the original (PDF) on 24 April 2008.
- ^ "FDA clears first 7T magnetic resonance imaging device" (Press release). U.S. Food and Drug Administration. 12 October 2017. Retrieved 7 December 2023.
- ISBN 978-0-306-45300-7.
- ^ "Philips Helium-Free MRI System Combines Productivity with High Quality Imaging | Medgadget". 12 September 2018.
- ^ Wang, Brian (8 January 2017). "Japan makes progress toward realization of MRI magnets using high temperature superconducting wire materials | NextBigFuture.com". Next Big Future.
- ^ "High-temperature superconducting coils tested for future NMR magnet - MagLab".
- ISBN 978-0-85274-118-4.
- ^
Gruetter R (June 1993). "Automatic, localized in vivo adjustment of all first- and second-order shim coils" (PDF). Magnetic Resonance in Medicine. 29 (6): 804–11. S2CID 41112243.
- ^ This unrealistically assumes that the gradient is linear out to the end of the magnet bore. While this assumption is fine for pedagogical purposes, in most commercial MRI systems the gradient droops significantly after a much smaller distance; indeed, the decrease in the gradient field is the main delimiter of the useful field of view of a modern commercial MRI system.
- ^
Oppelt A (2006). Imaging Systems for Medical Diagnostics: Fundamentals, Technical Solutions and Applications for Systems Applying Ionizing Radiation, Nuclear Magnetic Resonance and Ultrasound. ISBN 978-3-89578-226-8.
- ^
S2CID 9482029.
- S2CID 16046989.
- S2CID 14724155.
- S2CID 110429.
Further reading
- Pykett IL (1 May 1982). "NMR Imaging in Medicine" (PDF). PMID 7079720. Archived from the original(PDF) on 10 March 2016.
- Sprawls P (2000). Magnetic Resonance Imaging: Principles, Methods, and Techniques. Medical Physics Publishing. ISBN 978-0-944838-97-6.
- Haacke EM, Brown RF, Thompson M, Venkatesan R (1999). Magnetic resonance imaging: Physical principles and sequence design. New York: J. Wiley & Sons. ISBN 978-0-471-35128-3.
- Mansfield P (1982). NMR Imaging in Biomedicine: Supplement 2 Advances in Magnetic Resonance. Elsevier. ISBN 978-0-323-15406-2.
- Fukushima E (1989). NMR in Biomedicine: The Physical Basis. Springer Science & Business Media. ISBN 978-0-88318-609-1.
- Blümich B, Kuhn W (1992). Magnetic Resonance Microscopy: Methods and Applications in Materials Science, Agriculture and Biomedicine. Wiley. ISBN 978-3-527-28403-0.
- Blümer P (1998). Blümler P, Blümich B, Botto RE, Fukushima E (eds.). Spatially Resolved Magnetic Resonance: Methods, Materials, Medicine, Biology, Rheology, Geology, Ecology, Hardware. Wiley-VCH. ISBN 978-3-527-29637-8.
- Liang ZP, Lauterbur PC (1999). Principles of Magnetic Resonance Imaging: A Signal Processing Perspective. Wiley. ISBN 978-0-7803-4723-6.
- Schmitt F, Stehling MK, Turner R (1998). Echo-Planar Imaging: Theory, Technique and Application. Springer Berlin Heidelberg. ISBN 978-3-540-63194-1.
- Kuperman V (2000). Magnetic Resonance Imaging: Physical Principles and Applications. Academic Press. ISBN 978-0-08-053570-8.
- Blümich B (2000). NMR Imaging of Materials. Clarendon Press. ISBN 978-0-19-850683-6.
- Jin J (1998). Electromagnetic Analysis and Design in Magnetic Resonance Imaging. CRC Press. ISBN 978-0-8493-9693-9.










