Steatosis
| Steatosis | |
|---|---|
| Other names | Fatty change, fatty degeneration, fatty atrophy, adipose degeneration, fatty infiltration, fatty replacement |
non-alcoholic fatty liver disease. Masson's trichrome stain. | |
| Specialty | Gastroenterology |
| Complications | Fatty liver disease |
Steatosis, also called fatty change, is abnormal retention of fat (lipids) within a cell or organ.[1] Steatosis most often affects the liver – the primary organ of lipid metabolism – where the condition is commonly referred to as fatty liver disease. Steatosis can also occur in other organs, including the kidneys, heart, and muscle.[2] When the term is not further specified (as, for example, in 'cardiac steatosis'), it is assumed to refer to the liver.[3]
Risk factors associated with steatosis are varied, and may include
Steatosis reflects an impairment of the normal processes of
, the condition is known as macrovesicular steatosis; otherwise, the condition is known as microvesicular steatosis. While not particularly detrimental to the cell in mild cases, large accumulations can disrupt cell constituents, and in severe cases the cell may even burst.Pathogenesis
No single mechanism leading to steatosis exists; rather, a varied multitude of pathologies disrupt normal lipid movement through the cell and cause accumulation.[7] These mechanisms can be separated based on whether they ultimately cause an oversupply of lipid which can not be removed quickly enough (i.e., too much in), or whether they cause a failure in lipid breakdown (i.e., not enough used).[citation needed]
Failure of lipid metabolism can also lead to the mechanisms which would normally utilise or remove lipids becoming impaired, resulting in the accumulation of unused lipids in the cell. Certain toxins, such as alcohols, carbon tetrachloride, aspirin, and diphtheria toxin, interfere with cellular machinery involved in lipid metabolism. In those with Gaucher's disease, the lysosomes fail to degrade lipids and steatosis arises from the accumulation of glycolipids. Protein malnutrition, such as that seen in kwashiorkor, results in a lack of precursor apoproteins within the cell, therefore unused lipids which would normally participate in lipoprotein synthesis begin to accumulate.[citation needed]
Macrovesicular steatosis
Macrovesicular steatosis is the more common form of fatty degeneration and may be caused by oversupply of lipids due to obesity,
Microvesicular steatosis

Microvesicular steatosis is characterized by small intracytoplasmic fat vacuoles (liposomes) which accumulate within hepatocytes.
Histology
Histologically, steatosis is physically apparent as lipid within membrane bound liposomes of parenchymal cells.[2] When this tissue is fixed and stained to be better viewed under a microscope, the lipid is usually dissolved by the solvents used to prepare the sample. As such, samples prepared this way will appear to have empty holes (or vacuoles) within the cells where the lipid has been cleared. Special lipid stains, such as Sudan stains and osmium tetroxide are able to retain and show up lipid droplets, hence more conclusively indicating the presence of lipids. Other intracellular accumulations, such as water or glycogen, can also appear as clear vacuoles, therefore it becomes necessary to use stains to better determine what substance is accumulating.
Grossly, steatosis causes organ enlargement and lightening in colour.[2] This is due to the high lipid content increasing the organ's volume and becoming visible to the unaided eye. In severe cases, the organ may become vastly enlarged, greasy, and yellow in appearance.
-
Histological section of a mouse's liver showing severe steatosis. The clear vacuoles contained lipid in life; however, histological fixation caused it to be dissolved and hence only empty/clear spaces are seen.
-
Micrograph of fatty liver showing lipid steatosis.H&Estain.
-
Steatosis with a centrilobular pattern, which is the general tendency for steatosis in adults.[11]
Medical imaging
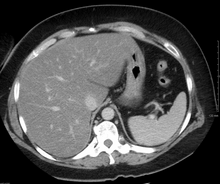
On
On magnetic resonance imaging, multiecho gradient echo images can be used to determine the percent fat fraction of the liver.[13] The different resonance frequencies between water and fat make this technique very sensitive and accurate. Acquisition of echoes in "in phase" and "out phase" conditions (pertaining to the relative phases of the fat and water proton contingents) enables to obtain a signal proportional to the water and fat contingent, or a signal proportional to the water minus the fat contingent. These signal intensities are then algebraically combined into a percent fat. More recent techniques take into account experimental noise, signal decay and spectroscopic properties of fat. Numerous validation studies have demonstrated excellent correlations between the steatosis level quantified at MRI and the steatosis levels semi-quantitatively and quantitatively determined on liver biopsies (reference methods).[citation needed] Several MRI vendors offer automated calculation of percent fat with acquisition sequences no longer than a single breath hold.[citation needed]
On abdominal ultrasonography, steatosis is seen as a hyperechoic liver as compared to the normal kidney.
-
Liver steatosis (fatty liver disease) as seen on MRI. Multiecho MR sequence in a healthy liver (top row) and a liver with severe steatosis (bottom row) are shown. In the healthy liver, the signal does not vary much in the different echoes. In the steatotic liver, the signal varies greatly between in and out phase echoes. Algebraic combination of these images can be used to accurately quantify liver steatosis.
-
Abdominal ultrasonography with the liver and kidney side by side (left image) may give a false impression of hyperechogenic liver, so it's preferably done with the organ borders facing the ultrasound probe (right image, of the same case).
-
hepatic vein.
Incidence
In
See also
- Fatty liver disease
- Lipid metabolism
- Non-alcoholic fatty liver disease
- Visceral fat
- Fat globules
References
- ^ "steatosis". Farlex Dictionary. Retrieved 2019-01-03.
- ^ ISBN 0-7216-7335-X.
- ^ "steatosis". Oxford dictionaries. Archived from the original on January 3, 2019. Retrieved 2019-01-03.
- PMID 17273155.
- S2CID 9057820.
- PMID 20818807.
- PMID 25422863.
- PMID 20857857.
- PMID 21172393.
- PMID 2177300.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 28652891.
- ISBN 978-0-7817-6135-2.[page needed]
- PMID 22025886.
- ^ Sarah Boseley (12 April 2019). "Experts warn of fatty liver disease 'epidemic' in young people". The Guardian.



![Steatosis with a centrilobular pattern, which is the general tendency for steatosis in adults.[11]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/d2/Histopathology_of_centrilobular_steatosis_of_the_liver.jpg/120px-Histopathology_of_centrilobular_steatosis_of_the_liver.jpg)


