BRCA2
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 13: 32.32 – 32.4 Mb | Chr 5: 150.45 – 150.49 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
BRCA2 and BRCA2 (
BRCA2 and BRCA1 are normally expressed in the cells of breast and other tissue, where they help repair damaged DNA or destroy cells if DNA cannot be repaired. They are involved in the repair of chromosomal damage with an important role in the error-free repair of DNA double strand breaks.[11][12] If BRCA1 or BRCA2 itself is damaged by a BRCA mutation, damaged DNA is not repaired properly, and this increases the risk for breast cancer.[13][14] BRCA1 and BRCA2 have been described as "breast cancer susceptibility genes" and "breast cancer susceptibility proteins". The predominant allele has a normal tumor suppressive function whereas high penetrance mutations in these genes cause a loss of tumor suppressive function, which correlates with an increased risk of breast cancer.[15]
The BRCA2 gene is located on the long (q) arm of
Function
Although the structures of the BRCA1 and BRCA2 genes are very different, at least some functions are interrelated. The proteins made by both genes are essential for repairing damaged DNA (see Figure of recombinational repair steps). BRCA2 binds the single strand DNA and directly interacts with the recombinase RAD51 to stimulate[27] and maintain [28] strand invasion, a vital step of homologous recombination. The localization of RAD51 to the DNA double-strand break requires the formation of the BRCA1-PALB2-BRCA2 complex. PALB2 (Partner and localizer of BRCA2)[29] can function synergistically with a BRCA2 chimera (termed piccolo, or piBRCA2) to further promote strand invasion.[30] These breaks can be caused by natural and medical radiation or other environmental exposures, but also occur when chromosomes exchange genetic material during a special type of cell division that creates sperm and eggs (meiosis). Double strand breaks are also generated during repair of DNA cross links. By repairing DNA, these proteins play a role in maintaining the stability of the human genome and prevent dangerous gene rearrangements that can lead to hematologic and other cancers.
BRCA2 has been shown to possess a crucial role in protection from the
Like BRCA1, BRCA2 probably regulates the activity of other genes and plays a critical role in embryo development.
Clinical significance
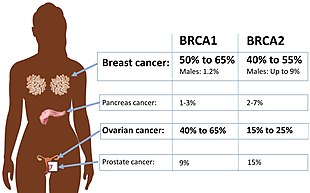
Certain variations of the BRCA2 gene increase risks for breast cancer as part of a hereditary breast–ovarian cancer syndrome. Researchers have identified hundreds of mutations in the BRCA2 gene, many of which cause an increased risk of cancer. BRCA2 mutations are usually insertions or deletions of a small number of DNA base pairs in the gene. As a result of these mutations, the protein product of the BRCA2 gene is abnormal, and does not function properly. Researchers believe that the defective BRCA2 protein is unable to fix DNA damage that occurs throughout the genome. As a result, there is an increase in mutations due to error-prone translesion synthesis past un-repaired DNA damage, and some of these mutations can cause cells to divide in an uncontrolled way and form a tumor.
People who have two mutated copies of the BRCA2 gene have one type of Fanconi anemia. This condition is caused by extremely reduced levels of the BRCA2 protein in cells, which allows the accumulation of damaged DNA. Patients with Fanconi anemia are prone to several types of leukemia (a type of blood cell cancer); solid tumors, particularly of the head, neck, skin, and reproductive organs; and bone marrow suppression (reduced blood cell production that leads to anemia). Women having inherited a defective BRCA1 or BRCA2 gene have risks for breast and ovarian cancer that are so high and seem so selective that many mutation carriers choose to have prophylactic surgery. There has been much conjecture to explain such apparently striking tissue specificity. Major determinants of where BRCA1- and BRCA2-associated hereditary cancers occur are related to tissue specificity of the cancer pathogen, the agent that causes chronic inflammation, or the carcinogen. The target tissue may have receptors for the pathogen, become selectively exposed to carcinogens and an infectious process. An innate genomic deficit impairs normal responses and exacerbates the susceptibility to disease in organ targets. This theory also fits data for several tumor suppressors beyond BRCA1 or BRCA2. A major advantage of this model is that it suggests there are some options in addition to prophylactic surgery.[33]
In addition to breast cancer in men and women, mutations in BRCA2 also lead to an increased risk of ovarian, uterine tube, prostate and pancreatic cancer. In some studies, mutations in the central part of the gene have been associated with a higher risk of ovarian cancer and a lower risk of prostate cancer than mutations in other parts of the gene. Several other types of cancer have also been seen in certain families with BRCA2 mutations.
In general, strongly inherited gene mutations (including mutations in BRCA2) account for only 5-10% of breast cancer cases; the specific risk of getting breast or other cancer for anyone carrying a BRCA2 mutation depends on many factors.[34]
History
The BRCA2 gene was discovered in 1994.
The gene was first cloned by scientists at Myriad Genetics, Endo Recherche, Inc., HSC Research & Development Limited Partnership, and the University of Pennsylvania.[41]
Methods to diagnose the likelihood of a patient with mutations in
Germline mutations and founder effect
All germline BRCA2 mutations identified to date have been inherited, suggesting the possibility of a large "founder" effect in which a certain mutation is common to a well-defined population group and can theoretically be traced back to a common ancestor. Given the complexity of mutation screening for BRCA2, these common mutations may simplify the methods required for mutation screening in certain populations. Analysis of mutations that occur with high frequency also permits the study of their clinical expression.[46] A striking example of a founder mutation is found in Iceland, where a single BRCA2 (999del5) mutation accounts for virtually all breast/ovarian cancer families.[47][48] This frame-shift mutation leads to a highly truncated protein product. In a large study examining hundreds of cancer and control individuals, this 999del5 mutation was found in 0.6% of the general population. Of note, while 72% of patients who were found to be carriers had a moderate or strong family history of breast cancer, 28% had little or no family history of the disease. This strongly suggests the presence of modifying genes that affect the phenotypic expression of this mutation, or possibly the interaction of the BRCA2 mutation with environmental factors. Additional examples of founder mutations in BRCA2 are given in the table below.
| Population or subgroup | BRCA2 mutation(s)[46][49] | Reference(s) |
|---|---|---|
| Ashkenazi Jewish | 6174delT | [50] |
| Dutch | 5579insA | [51] |
| Finns | 8555T>G, 999del5, IVS23-2A>G | [52][53] |
| French Canadians | 8765delAG, 3398delAAAAG | [54][55][56] |
| Hungarians | 9326insA | [57] |
| Icelanders | 999del5 | [47][48] |
| Italians | 8765delAG | [58] |
| Northern Irish | 6503delTT | [59] |
| Pakistanis | 3337C>T | [60] |
| Scottish | 6503delTT | [59] |
| Slovenians | IVS16-2A>G | [61] |
| Spanish | 3034delAAAC(codon936), 9254del5 | [62] |
| Swedish | 4486delG | [63] |
Meiosis
In the plant Arabidopsis thaliana, loss of the BRCA2 homolog AtBRCA2 causes severe defects in both male meiosis and in the development of the female gametocyte.[64] AtBRCA2 protein is required for proper localization of the synaptonemal complex protein AtZYP1 and the recombinases AtRAD51 and AtDMC1. Furthermore, AtBRCA2 is required for proper meiotic synapsis. Thus AtBRCA2 is likely important for meiotic recombination. It appears that AtBRCA2 acts during meiosis to control the single-strand invasion steps mediated by AtRAD51 and AtDMC1 occurring during meiotic homologous recombinational repair of DNA damages.[64]
Homologs of BRCA2 are also essential for meiosis in the fungus
Mice that produce truncated versions of BRCA2 are viable but sterile.[69] BRCA2 mutant rats have a phenotype of growth inhibition and sterility in both sexes.[70] Aspermatogenesis in these mutant rats is due to a failure of homologous chromosome synapsis during meiosis.
BRC repeat sequences
DMC1 (DNA meiotic recombinase 1) is a meiosis specific homolog of RAD51 that mediates strand exchange during homologous recombinational repair. DMC1 promotes the formation of DNA strand invasion products (joint molecules) between homologous DNA molecules. Human DMC1 interacts directly with each of a series of repeat sequences in the BRCA2 protein (called BRC repeats) that stimulate joint molecule formation by DMC1.[71] BRC repeats conform to a motif consisting of a sequence of about 35 highly conserved amino acids that are present at least once in all BRCA2-like proteins. The BRCA2 BRC repeats stimulate joint molecule formation by promoting the interaction of single-stranded DNA (ssDNA) with DMC1.[71] The ssDNA complexed with DMC1 can pair with homologous ssDNA from another chromosome during the synopsis stage of meiosis to form a joint molecule, a central step in homologous recombination. Thus the BRC repeat sequences of BRCA2 appear to play a key role in recombinational repair of DNA damages during meiotic recombination.
Overall, it appears that homologous recombination during meiosis functions to repair DNA damages,[citation needed] and that BRCA2 plays a key role in performing this function.
Neurogenesis
BRCA2 is required in the mouse for neurogenesis and suppression of medulloblastoma.[72] ‘’BRCA2’’ loss profoundly affects neurogenesis, particularly during embryonic and postnatal neural development. These neurological defects arise from DNA damage.[72]
Epigenetic control
Epigenetic alterations in expression of BRCA2 (causing over-expression or under-expression) are very frequent in sporadic cancers (see Table below) while mutations in BRCA2 are rarely found.[73][74][75]
In non-small cell lung cancer, BRCA2 is epigenetically repressed by hypermethylation of the promoter.[76] In this case, promoter hypermethylation is significantly associated with low mRNA expression and low protein expression but not with loss of heterozygosity of the gene.
In sporadic ovarian cancer, an opposite effect is found. BRCA2 promoter and 5'-UTR regions have relatively few or no methylated CpG dinucleotides in the tumor DNA compared with that of non-tumor DNA, and a significant correlation is found between hypomethylation and a >3-fold over-expression of BRCA2.[77] This indicates that hypomethylation of the BRCA2 promoter and 5'-UTR regions leads to over-expression of BRCA2 mRNA.
One report indicated some epigenetic control of BRCA2 expression by the microRNAs miR-146a and miR-148a.[78]
BRCA2 expression in cancer
In eukaryotes, BRCA2 protein has an important role in homologous recombinational repair. In mice and humans, BRCA2 primarily mediates orderly assembly of RAD51 on single-stranded (ss) DNA, the form that is active for homologous pairing and strand invasion.[79] BRCA2 also redirects RAD51 from double-stranded DNA and prevents dissociation from ssDNA.[79] In addition, the four paralogs of RAD51, consisting of RAD51B (RAD51L1), RAD51C (RAD51L2), RAD51D (RAD51L3), XRCC2 form a complex called the BCDX2 complex (see Figure: Recombinational repair of DNA). This complex participates in RAD51 recruitment or stabilization at damage sites.[26] The BCDX2 complex appears to act by facilitating the assembly or stability of the RAD51 nucleoprotein filament. RAD51 catalyses strand transfer between a broken sequence and its undamaged homologue to allow re-synthesis of the damaged region (see homologous recombination models).
Some studies of cancers report over-expressed BRCA2 whereas other studies report under-expression of BRCA2. At least two reports found over-expression in some sporadic breast tumors and under-expression in other sporadic breast tumors.[80][81] (see Table).
Many cancers have epigenetic deficiencies in various DNA repair genes (see Frequencies of epimutations in DNA repair genes in cancers). These repair deficiencies likely cause increased unrepaired DNA damages. The over-expression of BRCA2 seen in many cancers may reflect compensatory BRCA2 over-expression and increased homologous recombinational repair to at least partially deal with such excess DNA damages. Egawa et al.[82] suggest that increased expression of BRCA2 can be explained by the genomic instability frequently seen in cancers, which induces BRCA2 mRNA expression due to an increased need for BRCA2 for DNA repair.
Under-expression of BRCA2 would itself lead to increased unrepaired DNA damages. Replication errors past these damages (see translesion synthesis) would lead to increased mutations and cancer.
| Cancer | Over or Under expression | Frequency of altered expression | Evaluation method | Ref. |
|---|---|---|---|---|
| Sporadic ovarian cancer | Over-expression | 80% | messenger RNA | [77] |
| Sporadic ovarian cancer | Under-expression | 42% | immunohistochemistry | [83] |
| (recurrent cancer in study above) | Increased-expression | 71% | immunohistochemistry | [83] |
| Non-small cell lung cancer | Under-expression | 34% | immunohistochemistry | [76] |
| Breast cancer | Over-expression | 66% | messenger RNA | [82] |
| Breast cancer | Over-expression | 20% | messenger RNA | [80] |
| (same study as above) | Under-expression | 11% | messenger RNA | [80] |
| Breast cancer | Over-expression | 30% | immunohistochemistry | [81] |
| (same study as above) | Under-expression | 30% | immunohistochemistry | [81] |
| Triple negative breast cancer | Under-expression | 90% | immunohistochemistry | [84] |
Interactions
BRCA2 has been shown to interact with
- BRE,[85]
- BARD1,[85][86]
- BCCIP,[87]
- BRCA1,[85][88][89][90]
- BRCC3,[85]
- BUB1B,[91]
- CREBBP,[92]
- C11orf30,[93]
- FANCD2,[94][95][96]
- FANCG,[97]
- FLNA,[98]
- HMG20B,[99][100]
- P53,[85][101]
- PALB2,[29][102]
- PCAF,[103][104]
- PLK1,[103][105]
- RAD51,[85][88][103][106][107][108][109][110][111][112][87][89][101]
- RPA1,[113]
- SHFM1[114][115] and
- SMAD3.[116]
Domain architecture
| BRCA2 repeat | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
BRCA2 contains a number of 39 amino acid repeats that are critical for binding to RAD51 (a key protein in DNA recombinational repair) and resistance to methyl methanesulphonate treatment.[101][108][109][117]
The BRCA2 helical domain adopts a
The BRCA OB1 domain assumes an OB fold, which consists of a highly curved five-stranded
The BRCA OB3 domain assumes an OB fold, which consists of a highly curved five-stranded
The Tower domain adopts a
Patents, enforcement, litigation, and controversy
A patent application for the isolated
The patents begin to expire in 2014.Peter Meldrum, CEO of Myriad Genetics, has acknowledged that Myriad has "other competitive advantages that may make such [patent] enforcement unnecessary" in Europe.[120]
Legal decisions surrounding the BRCA1 and BRCA2 patents will affect the field of genetic testing in general.[121] In June 2013, in Association for Molecular Pathology v. Myriad Genetics (No. 12-398), the US Supreme Court unanimously ruled that, "A naturally occurring DNA segment is a product of nature and not patent eligible merely because it has been isolated," invalidating Myriad's patents on the BRCA1 and BRCA2 genes. However, the Court also held that manipulation of a gene to create something not found in nature could still be eligible for patent protection.[122] The Federal Court of Australia came to the opposite conclusion, upholding the validity of an Australian Myriad Genetics patent over the BRCA1 gene in February 2013,[123] but this decision is being appealed and the appeal will include consideration of the US Supreme Court ruling.[124]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000139618 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000041147 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Hamel PJ (2007-05-29). "BRCA1 and BRCA2: No Longer the Only Troublesome Genes Out There". HealthCentral. Retrieved 2010-07-02.
- ^ "OrthoMaM phylogenetic marker: BRCA2 coding sequence". Archived from the original on 2016-03-03. Retrieved 2010-02-19.
- ^ "BRCA2 gene tree". Ensembl. May 2021
- PMID 10193517.
- S2CID 24297965.
- ^ Check W (2006-09-01). "BRCA: What we know now". College of American Pathologists. Retrieved 2010-08-23.
- PMID 17683622.
- ^ Friedenson B (2008-06-08). "Breast cancer genes protect against some leukemias and lymphomas" (video). SciVee.
- ^ "Breast and Ovarian Cancer Genetic Screening". Palo Alto Medical Foundation. Archived from the original on 4 October 2008. Retrieved 2008-10-11.
- PMID 17683622.
- PMID 20400477.
- ^ PMID 8091231.
- ^ "BRCA2 breast cancer 2, early onset [Homo sapiens]". EntrezGene. National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Breast cancer type 2 susceptibility protein - Homo sapiens (Human)". P51587. UniProt.
- ^ Williams-Jones B (2002). Genetic testing for sale: Implications of commercial brca testing in Canada (Ph.D.). The University of British Columbia.
- PMID 20484397.
- PMID 19633289.
- PMID 21466974.
- PMID 21088254.
- PMID 12239151.
- PMID 24998779.
- ^ PMID 23149936.
- PMID 20729832.
- PMID 30872704.
- ^ PMID 16793542.
- PMID 20871615.
- PMID 29038466.
- PMID 20301425.
- PMID 22972572.
- ^ "High-Penetrance Breast and/or Ovarian Cancer Susceptibility Genes". National Cancer Institute. Retrieved 7 December 2012.
- S2CID 4346791.
- ^ High-Impact Science: Tracking down the BRCA genes (Part 2) Archived 2012-03-03 at the Wayback Machine - Cancer Research UK science blog, 2012
- ^ "Kenneth Offit | Breast Cancer Research Foundation | BCRF". Bcrfcure.org. 23 June 2014. Retrieved 2015-07-16.
- ISSN 0362-4331. Retrieved 2015-07-02.
- ^ "20 Years of Progress in Understanding Breast Cancer" (JPG). Mskcc.org. Retrieved 2015-07-17.
- ISSN 0362-4331. Retrieved 2015-07-07.
- ^ a b US patent 5837492, Tavtigian SV, Kamb A, Simard J, Couch F, Rommens JM, Weber BL, "Chromosome 13-linked breast cancer susceptibility gene", issued 1998-11-17, assigned to Myriad Genetics, Inc., Endo Recherche, Inc., HSC Research & Development Limited Partnership, Trustees of the University of Pennsylvania
- ^ a b US patent 5747282, Skolnick HS, Goldgar DE, Miki Y, Swenson J, Kamb A, Harshman KD, Shattuck-Eidens DM, Tavtigian SV, Wiseman RW, Futreal PA, "7Q-linked breast and ovarian cancer susceptibility gene", issued 1998-05-05, assigned to Myriad Genetics, Inc., The United States of America as represented by the Secretary of Health and Human Services, and University of Utah Research Foundation
- ^ US patent 5837492, Tavtigian SV, Kamb A, Simard J, Couch F, Rommens JM, Weber BL, "Chromosome 13-linked breast cancer susceptibility gene", issued 1998-11-17, assigned to Myriad Genetics, Inc., Endo Recherche, Inc., HSC Research & Development Limited Partnership, Trustees of the University of Pennsylvania
- ^ a b Myriad Investor Page—see "Myriad at a glance" Archived 2012-10-18 at the Wayback Machine accessed October 2012
- ^ a b c Schwartz J (2009-05-12). "Cancer Patients Challenge the Patenting of a Gene". Health. New York Times.
- ^ S2CID 23327569.
- ^ S2CID 8443452.
- ^ PMID 9150155.
- PMID 10612815.
- S2CID 11909356.
- PMID 11597388.
- PMID 9585608.
- S2CID 41804152.
- S2CID 23931343.
- PMID 16539696.
- PMID 16980226.
- S2CID 25394976.
- PMID 10682665.
- ^ PMID 12698193.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - PMID 12181777.
- PMID 12461697.
- S2CID 30388365.
- PMID 11250694.
- ^ PMID 22077663.
- PMID 12408834.
- PMID 18779660.
- PMID 15798199.
- PMID 18266476.
- S2CID 42462448.
- PMID 16964288.
- ^ PMID 26976601.
- ^ PMID 17476307.
- S2CID 9831745.
- S2CID 3203046.
- S2CID 26808443.
- ^ PMID 17289874.
- ^ PMID 12124354.
- PMID 25537514.
- ^ PMID 21731065.
- ^ PMID 10498873.
- ^ PMID 25879355.
- ^ S2CID 9083282.
- ^ PMID 19602291.
- S2CID 3566545.
- ^ PMID 14636569.
- PMID 19176389.
- ^ PMID 11313963.
- ^ PMID 11477095.
- ^ PMID 9774970.
- PMID 14499622.
- PMID 10749118.
- PMID 19578754.
- S2CID 18911371.
- PMID 15199141.
- PMID 15115758.
- PMID 18263878.
- PMID 12915460.
- PMID 11602572.
- S2CID 5822368.
- PMID 12032298.
- ^ PMID 9811893.
- ^ "Entrez Gene: PALB2 partner and localizer of BRCA2".
- ^ PMID 12815053.
- PMID 9824164.
- PMID 14647413.
- S2CID 4238943.
- PMID 14580352.
- ^ PMID 9560268.
- ^ PMID 9405383.
- S2CID 45954246.
- S2CID 4359383.
- PMID 15065660.
- PMID 12527904.
- PMID 10373512.
- ^ PMID 12228710.
- PMID 12165866.
- S2CID 2312211.
- S2CID 222165754.
- ^ "ACLU sues over patents on breast cancer genes". CNN. Archived from the original on 15 May 2009. Retrieved 2009-05-14.
- ^ Conley J, Vorhous D, Cook-Deegan J (2011-03-01). "How Will Myriad Respond to the Next Generation of BRCA Testing?". Robinson, Bradshaw, and Hinson. Retrieved 2012-12-09.
- ^ "Genetics and Patenting". Human Genome Project Information. U.S. Department of Energy Genome Programs. 2010-07-07.
- ^ Liptak A (13 June 2013). "Supreme Court Rules Human Genes May Not Be Patented". New York Times. Retrieved 13 June 2013.
- Sydney Morning Herald. Retrieved June 14, 2013.
- Sydney Morning Herald. Retrieved June 14, 2013.
Further reading
- Zou JP, Hirose Y, Siddique H, Rao VN, Reddy ES (1999). "Structure and expression of variant BRCA2a lacking the transactivation domain". PMID 10023017.
- Venkitaraman AR (2001). "Chromosome stability, DNA recombination and the BRCA2 tumour suppressor". PMID 11343905.
- Orelli BJ, Bishop DK (2001). "BRCA2 and homologous recombination". PMID 11597317.
- Daniel DC (2002). "Highlight: BRCA1 and BRCA2 proteins in breast cancer". S2CID 30091586.
- Tutt A, Ashworth A (2003). "The relationship between the roles of BRCA genes in DNA repair and cancer predisposition". PMID 12470990.
- Gonçalves A, Viens P, Sobol H, Maraninchi D, Bertucci F (2005). "[Molecular alterations in breast cancer: clinical implications and new analytical tools]". PMID 15936476.
- Hay T, Clarke AR (2005). "DNA damage hypersensitivity in cells lacking BRCA2: a review of in vitro and in vivo data". PMID 16042582.
- Domchek SM, Weber BL (2006). "Clinical management of BRCA1 and BRCA2 mutation carriers". PMID 16998496.
- Honrado E, Osorio A, Palacios J, Benitez J (2006). "Pathology and gene expression of hereditary breast tumors associated with BRCA1, BRCA2 and CHEK2 gene mutations". Oncogene. 25 (43): 5837–45. S2CID 20960561.
External links
- BRCA2 Protein at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
|}

