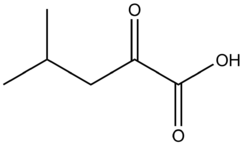
α-Ketoisocaproic acid
(Redirected from
Alpha-Ketoisocaproic acid
)
| |
| Names | |
|---|---|
| IUPAC name
4-Methyl-2-oxopentanoic acid
| |
| Systematic IUPAC name
4-Methyl-2-oxopentanoic acid[1] | |
| Other names
4-Methyl-2-oxovaleric acid
2-Ketoisocaproic acid 2-Oxo-4-methylpentanoic acid 2-Oxo-4-methylvaleric acid 2-Oxoisocaproic acid 2-Oxoleucine Isobutylglyoxylic acid Ketoleucine α-Ketoisocapronic acid α-Oxoisocaproic acid | |
| Identifiers | |
3D model (
JSmol ) |
|
| 3DMet | |
| 1701823 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.011.304 |
| EC Number |
|
IUPHAR/BPS |
|
| KEGG | |
| MeSH | Alpha-ketoisocaproic+acid |
PubChem CID
|
|
| UNII | |
| UN number | 3265 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H10O3 | |
| Molar mass | 130.143 g·mol−1 |
| Density | 1.055 g cm−3 (at 20 °C) |
| Melting point | 8 to 10 °C (46 to 50 °F; 281 to 283 K) |
| Boiling point | 85 °C (185 °F; 358 K) at 13 mmHg |
| log P | 0.133 |
| Acidity (pKa) | 2.651 |
| Basicity (pKb) | 11.346 |
| Hazards | |
| GHS labelling: | |

| |
| Danger | |
| H314 | |
| P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
α-Ketoisocaproic acid (α-KIC), also known as 4-methyl-2-oxovaleric acid, and its
resistance exercises in people who do not usually perform those exercises.[9]
Biological activity
Supplements
α-KIC has been studied as a
beta-hydroxy beta-methylbutyrate (HMB), participants reported delayed onset of muscle soreness, as well as other positive effects such as increased muscle girth.[9] It is important to note that studies have also suggested that ɑ-KIC taken alone did not have any significant positive impacts on physical performance, so it should be taken in conjunction with other ergogenic substances.[10] ɑ-KIC is not available as a supplement on its own, but its decarboxylated form HMB is available in calcium salt capsules or powder.[4]
Applications
The biochemical implications of α-KIC are largely connected to other
Protein Synthesis, skeletal muscle regeneration, and skeletal muscle proteolysis have all been noted to change when ɑ-KIC is taken. There is not much research into the specific mechanisms taking part in these processes, but there is a noticeable correlation between ɑ-KIC ingestion and increased skeletal muscle protein synthesis, regeneration, and proteolysis.[4]
Toxicity
Multiple studies have demonstrated that there have been no adverse effects on humans nor animals that ingested α-KIC or HMB.[11][12]
In patients with maple syrup urine disease, who are unable to metabolize the branched chain alpha keto acids, α-KIC is believed to be one of the key mediators of neurotoxicity.[13]
Medical use
Branched-chain alpha-keto acids such as α-KIC are found in high concentrations in the urine of people who suffer from Maple Syrup Urine Disease. This is disease is caused by a partial
excreted in the urine as α-KIC, HMB, and many other similar keto acids. Flare-ups in people who have this condition are caused due to poor diet.[7] Symptoms of Maple Syrup Urine Disease include sweet smelling urine, irritability, lethargy, and in serious cases edema of the brain, apnea, coma, or respiratory failure.[14][7] Treatment includes lowering leucine intake and a specialized diet to make up for the lack of leucine ingestion.[7]
Leucine metabolism
References
- ^ CID 70 from PubChem
- ^ PMID 23374455.
- ^ "Leucine".
- ^ PMID 23374455.
- ^ S2CID 11120110.
- ^ ISBN 978-0-12-387784-0.
- ^ a b c d Strauss, Kevin A; Puffenberger, Erik G; Carson, Vincent J (1993). "Maple Syrup Urine Disease". GeneReviews. University of Washington, Seattle.
- ^ ISBN 978-0-12-396454-0.
- ^ PMID 16286672.
- PMID 17908285.
- PMID 10917905.
- PMID 16006030.
- S2CID 1253267.
- ^ PMID 20061171.
- ^ ISBN 978-0-12-387784-0. Retrieved 6 June 2016.
Energy fuel: Eventually, most Leu is broken down, providing about 6.0kcal/g. About 60% of ingested Leu is oxidized within a few hours ... Ketogenesis: A significant proportion (40% of an ingested dose) is converted into acetyl-CoA and thereby contributes to the synthesis of ketones, steroids, fatty acids, and other compounds
Figure 8.57: Metabolism of L-leucine